Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
July 2023
Nomenclature
Bigflower agoseris (Agoseris grandiflora [Nutt.] Greene) belongs to the Cichorieae tribe and Microseridinae subtribe within the Asteraceae family (Hemleben et al. 1978; Baird 2006).
Family
Asteraceae – Aster family
Genus
Agoseris
Species
grandiflora
NRCS Plant Code
AGGR (USDA NRCS 2023).
Subtaxa
The Flora of North America (Baird 2006) recognizes two varieties, grandiflora (Nutt.) Greene and leptophylla G.I. Baird.
Synonyms
Agoseris cinerea Greene, A. grandiflora var. intermedia (Greene) Jepson, A. grandiflora var. plebeia (Greene) G. L. Wittrock, A. intermedia Greene, A. marshallii (Greene) Greene, A. obtusifolia (Suksdorf) Rydberg, A. plebeia (Greene) Greene, and Stylopappus grandiflorus Nutt. (Baird 2006).
Common Names
Bigflower agoseris, grassland agoseris, large-flowered agoseris, California dandelion, goat chicory (Tilley 2013; LBJWC 2018), and Puget sound agoseris (variety leptophylla) (Baird 2004).
Chromosome Number
2n = 18 (Munz and Keck 1973; Baird 2006).
Hybridization
Throughout most of the species’ range, varieties of bigflower agoseris are parapatric, meaning they evolved in spatially segregated but contiguous habitats and intermediate forms are limited to areas where they remain in contact (Baird 2004). In the Selkirk-Clearwater Mountains, Columbia River Gorge, southern Willamette Valley, and Siskiyou-Klamath Mountains, however, varieties are considered sympatric. In these areas, varieties co-occur, but evolved separately. Varieties appear to be introgressive and intermediates are common (Baird 2006).
Intermediates between variety grandiflora and other species are rare, but hybridization with A. apargioides is suspected (Baird 2006). Variety grandiflora is one of the suspected parents of A. × elata, especially populations in the Sierra Nevada Range. Variety leptophylla is a suspected parent of A. × elata, especially of the Puget Sound-Willamette Valley populations (Baird 2006).
Distribution
Bigflower agoseris occurs in locations west of central Utah. It occurs in southern British Columbia, Washington, Oregon, California, Nevada, Utah, Idaho, and Montana, and is most common in those states bordering the Pacific Ocean (Baird 2006). Variety grandiflora is most common east of the Cascade Mountains and southward on both the east and west side of the Sierra Nevada Mountains to southern California (Fig. 1). Variety leptophylla is most common west of the Cascade Mountains from Vancouver Island through Puget Sound and the Willamette Valley to the Siskiyou-Klamath Mountains of southwestern Oregon and northwestern California. Variety leptophylla also occurs sporadically in mesic forests on the eastern slopes of the Cascade Mountains with disjunct populations in the Selkirk-Clearwater Mountains of British Columbia and northern Idaho (Baird 2004, 2006).
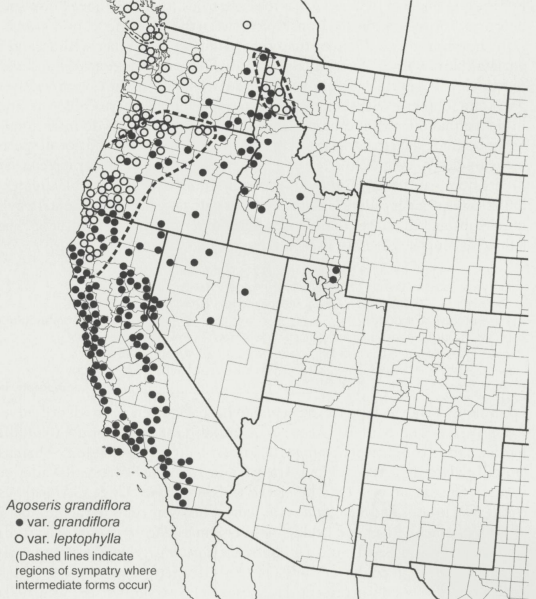
Figure 1. Distribution of bigflower agoseris varieties (grandiflora [filled circles], leptophylla [open circles]). Dashed lines indicate regions of sympatry where the varieties form intermediates (Baird 2004).
Habitat And Plant Associations
Bigflower agoseris grows in low densities on dry to mesic sites where the average annual precipitation ranges from 12 to 20 in (300-500 mm) (Baird 2006;Tilley 2013). It occurs in grassland (Fig. 2), steppe (Fig. 3), chaparral, woodland, and open forest communities (Baird 2006). Variety grandiflora occurs primarily in grassland, steppe, or chaparral communities, and variety leptophylla typically occurs in lowland prairies, coastal forests, and open woodlands and forests (Baird 2006).
Leger and Barga (2017) modeled potential habitat for bigflower agoseris and nine other forb species using herbarium records together with bioclimatic variables obtained from PRISM data for last 64 years. Soil water balance contributed 52.5% and summer precipitation contributed 37.7% to defining the ecological niche for bigflower agoseris. Findings suggested that this species grows in areas with soils that retain water and sites receiving high amounts of annual rainfall, but little summer rain.

Figure 2. Bigflower agoseris setting seed (dandelion-like seed heads) in an Idaho grassland. Photo: USDI Bureau of Land Management (BLM) ID 931 Seeds of Success (SOS).
Bigflower agoseris is often associated with open and disturbed sites (Pavek et al. 2012). In California, it grows in many grassland, shrubland, and woodland communities (Munz and Keck 1973; Hickman 1993). In Utah and Nevada, it grows in sagebrush (Artemisia spp.), pinyon-juniper (Pinus–Juniperus spp.), and ponderosa pine (P. ponderosa) vegetation (Tidestrom et al. 1925). In valleys and montane zones of western Montana, it occurs in meadow forest openings (Lesica 2012).
In surveys conducted from the 1980s until 2004 in Oregon’s Blue and Ochoco Mountains (Johnson and Swanson 2005), bigflower agoseris was common in bluebunch wheatgrass (Pseudoroegneria spicata) communities. In the bluebunch wheatgrass-Sandberg bluegrass-arrowleaf balsamroot (Poa secunda–Balsamorhiza sagittata) vegetation types, constancy of bigflower agoseris increased with increasing ungulate use that coincided with a loss of bunchgrasses. Cover of bigflower agoseris was greatest (3%) in relatively undisturbed bluebunch wheatgrass-Sandberg bluegrass-narrowleaf skullcap (Scutellaria angustifolia) vegetation types (Johnson and Swanson 2005). Tisdale (1976) considered bigflower agoseris a common perennial forb in the bluebunch wheatgrass-Sandberg bluegrass and bluebunch wheatgrass-Idaho fescue (Festuca idahoensis) grasslands growing north of the Salmon River in Idaho (Tisdale 1976).
A Humboldt Redwoods State Park in northern coastal California study evaluated the vegetation associated with environmental gradients. Bigflower agoseris was associated with the second lowest moisture, lowest nutrient, and highest light conditions (Waring and Major 1964).

Figure 3. Bigflower agoseris setting seed in a big sagebrush grassland in Oregon. Photo: USDI BLM OR010 SOS.
Elevation
Bigflower agoseris occupies an elevation range of 30 to 8,200 ft (10-2,500 m). The elevation range for variety grandiflora is 1,000 to 8,200 ft (300–2,500 m), broader than that of variety leptophylla (30–5,900 ft [10–1,800 m]) (Baird 2006). Plants typically occur below 6,600 ft (2,000 m) in California (Hickman 1993; Munz and Keck 1973) and in the Intermountain West (Cronquist et al. 1994).
Soils
Bigflower agoseris tolerates a broad range of soil types (Tilley 2013) and pH of 5.8 to 7.3 is reported for Nevada habitats (Eldredge et al. 2013). In relatively undisturbed, late seral Wyoming big sagebrush (Artemisia tridentata subsp. wyomingensis)-grassland habitats of southeastern Oregon and northern Nevada, bigflower agoseris was moderately positively correlated with clay in the upper 6 in (15 cm) of the soil profile (Davies et al. 2007). At Mima Mounds in Turnbull National Wildlife Refuge in Cheney, Washington, bigflower agoseris grew on alluvial mounds, basalt mounds, and basalt intermound areas. Soils between mounds were shallow (0-7.6 in [19.5 cm] deep) and experienced extremes of wet and dry conditions, including potential inundation in the spring. Mounds were up to 7 ft (2 m) tall and 100 ft (30 m) in diameter, and soils were aggregations of fine loess over alluvial gravel or basalt bedrock substrates (Anicito 2013). At the Camp Roberts Army National Guard Training area in San Luis Obispo County, California, bigflower agoseris grew with the threatened purple amole (Chlorogalum purpureum) in small fescue (Vulpia microstachys) grasslands. At the training area, bigflower agoseris and yellowflower tarweed (Holocarpha virgata) comprised 75% of total plant cover in survey plots with purple amole. These plots occurred where soils were gravelly and overlaid hard-packed clay (Kofron et al. 2013).
Description
Bigflower agoseris is a robust scapose perennial (Fig. 4) from a branched root crown and strong, deep, simple or branched taproot (U.S. Department of Agriculture 1937; Munz and Keck 1973; Tilley 2013). Stems elongate after flowering, and plants can be up to 33 in (85 cm) tall when flowering and producing seed (Beidleman and Kozloff 2003). Stems are few to several and woolly when plants are young (Pavek et al. 2012). Plants release a milky juice when herbage is clipped or damaged (U.S. Department of Agriculture 1937).

Figure 4. Bigflower agoseris plant with fully opened seed head. Photo: USDI BLM ID931 SOS.
Basal leaves are erect to ascending with purplish petioles (Baird 2006; Lesica 2012; Hitchcock and Cronquist 2018). Leaf blades are usually linear to oblanceolate and 4 to 16 in (10-40 cm) long and 0.4 to 1.2 in (1-3 cm) wide (Fig. 5) (Munz and Keck 1973; Cronquist et al. 1994; Baird 2006; Pavek et al. 2012). Leaf margins are ciliate to tomentose and range from entire to dentate, lobed, or deeply pinnatifid with lobes in three to five pairs (Hickman 1993; Baird 2006; Lesica 2012).

Figure 5. Upright stems and linear basal leaves of bigflower agoseris growing amongst grasses in Oregon. Photo: USDI BLM OR010 SOS.
Plants produce solitary heads of 40 to 500 perfect, yellow, ligulate flowers (Fig. 6) (Hitchcock and Cronquist 2018). Flowers turn pink with age and drying (Pavek et al. 2012). Involucres are campanulate to hemispheric and 0.8 to 2 in (2-5 cm) tall in fruit (Baird 2006; Hitchcock and Cronquist 2018). Phyllaries in four or five series are green or medially rosy purple, and more or less hairy with prominent tips (Hickman 1993; Baird 2006; Lesica 2012). Inner phyllaries are elongate and narrow and about twice as long as the outer phyllaries, which are short and broad (Tidestrom et al. 1925; Munz and Keck 1973; Cronquist et al. 1994; Beidleman and Kozloff 2003).

Figure 6. Solitary bigflower agoseris flower head. Photo: USDI BLM OR101 SOS.

Figure 7. Involucre of bigflower agoseris flower. Photo: USDI BLM OR010 SOS.
Bigflower agoseris fruits (cypselas) are often referred to as achenes (Baird 2006; LBJWC 2018) and from this point forward will be called seeds. The fruit body (up to 7 mm long) is slender, tapered at the apex, and 10-ribbed with deep, straight, longitudinal grooves (Tidestrom et al. 1925). Fruits taper into a long, nerveless beak that is three to four times as long as the body and attached to a cottony pappus (7-15 mm) with bristles in two or three series (Tidestrom et al. 1925; Cronquist et al. 1994; Baird 2006; Hitchcock and Cronquist 2018).
Distinguishing varieties. In general, variety leptophylla differs from grandiflora by its smaller size, slender, laciniate lobed leaves, slender peduncles, smaller heads with fewer florets and outer phyllaries that are subequal to rather than longer than the inner phyllaries at anthesis (Baird 2004). Leaves of variety leptophylla range from 4 to 14 in (10-36 cm) long and 0.8 to 3 in (2-8 cm) wide (not including lobes). Involucres of variety leptophylla are campanulate and 0.8 to 1.5 in (2-4 cm) tall in fruit. Flower heads typically have 40 to 60 or more ray florets (Baird 2006; Hitchcock and Cronquist 2018). Leaves of variety grandiflora are oblanceolate to spatulate, 4 to 20 in (10–50 cm) long and 0.4 to 1.4 in (1–3.5 cm) wide (excluding lobes). Involucres of variety grandiflora are campanulate to hemispheric and 1.2 to 2.2 in (3–5.5 cm) tall in fruit. Flower heads typically have 150 to 300 (500) ray florets (Baird 2006; Hitchcock and Cronquist 2018).
Reproduction
Bigflower agoseris reproduces from seed. Flowers require cross pollination by insects (Bartow 2015). Flowering may occur from March through October (Baird 2006), but it is most common in May, June, or July (Munz and Keck 1973; Cronquist et al. 1994).
Ecology
Bigflower agoseris is a short-lived, colonizing species (Pavek et al. 2012; Tilley 2013). It established in areas representing primary succession following the eruption of the Mt. St. Helens Volcano in the Cascade Mountains of southwestern Washington in 1980. Before the blast, the area was a patchwork of forests and clearcuts. In vegetation surveys conducted in 1993 and 1994, bigflower agoseris occurred although infrequently at sites where existing vegetation was essentially eliminated and revegetation resulted from offsite propagules (Titus et al. 1998).
Seed And Seedling Ecology
Bigflower agoseris seed is wind dispersed (Fig. 8), and seedlings are quick to establish (Pavek et al. 2012).

Figure 8. Bigflower agoseris seed head with 10 to 20 seeds with pappi still attached. Photo: USDI BLM OR101 SOS.
Disturbance Ecology
Bigflower agoseris capitalizes on openings associated with soil disturbance or reduced plant competition caused by disturbance. In big sagebrush-grassland communities ranging from low to high seral condition in northern Nevada, the frequency of bigflower agoseris increased with decreasing seral conditions resulting from increases in grazing pressure (Tueller and Platou 1991). In north-central California, the density of bigflower agoseris was high four years after a clearcut logging operation in a ponderosa pine forest. Plants originated from off-site seed, and in the fifth year following logging, the clearcut was dominated by bigflower agoseris and smooth cat’s ear (Hypochaeris glabra) (McDonald 1999).
Several studies report early colonization or increases in abundance of bigflower agoseris following fires and fuel treatments in shrublands and forests (Swanson 1970; Stickney 1986). Bigflower agoseris was an early colonizer of burned grand fir (Abies grandis) forests in north-central Washington. Sites had well-drained medium-textured soils, burned in August or September, and were seeded with nonnative grass cultivars (Roche et al. 2008). It was a minor colonizer reaching its greatest abundance 3 to 5 years after clearcutting and slash burning of old-growth Douglas-fir (Pseudotsuga menziesii) in Oregon’s western Cascade Mountains (Halpern 1989). Plants were restricted to sites that supported more xeric communities before clearcutting and occurred in lightly burned but not severely burned plots (Dyrness 1973; Zouhar 2013). Similarly, frequency of bigflower agoseris increased after thinning and burning treatments in dry mixed-conifer forests in the northeastern Cascade Mountains of Washington. Frequency of bigflower agoseris before treatments was 0%, increasing to 2% after 2 to 3 years and to 10% after 10 to 13 years after treatments (Rossman et al. 2018).
At locations in Oregon and California, bigflower agoseris was an indicator species for treatments on burned sites. In buckbrush (Ceanothus cuneatus) chaparral in southwestern Oregon, it was an indicator species on 2-year-old plots that were part of hand cut-pile and burn fuel treatments. The other fuel treatment, shrub mastication, did not support bigflower agoseris, at least not at the two-year post-treatment mark (Sikes and Muir 2009). Bigflower agoseris was also an indicator species on 22-year-old sites that were part of an active post-fire reforestation effort involving herbicide treatments and tree planting in the Sierra Nevada Mountains in California. It also occurred, but was uncommon, on plots allowed to regenerate without intervention following stand-replacing fires in mixed-conifer forests (Bohlman et al. 2016).
Wildlife And Livestock Use
Bigflower agoseris taproots are eaten by rodents (Skinner et al. 2005), plants and associated invertebrates are important to greater sage-grouse (Martin et al. 1951), and aboveground plant parts are eaten by elk (Cervus canadensis) and livestock (Roche et al. 2008). Agoseris species are important to greater sage-grouse (Centrocercus urophasianus). Leaves are eaten in late spring and early summer during brood rearing (Luna et al. 2018). An aggregate mass (mass of bigflower agoseris/total food mass) of up to 3.1% and a frequency of up to 9% was found in the diets of greater sage-grouse chicks at Hart Mountain, Lake County, Oregon. These findings were based on the evaluation of the crops of 44 chicks age 1 to 10 weeks collected in June and July of 1988 and 1989 (Pyle 1992).
Bigflower agoseris was reported as valuable elk forage by Cook (2002 cited in Roche et al. 2008). Plants are also eaten by livestock and reportedly were closely grazed by cattle and horses in the Payette National Forest in Idaho and used in early summer by livestock on the Ochoco National Forest in Oregon (Hermann 1966).
Ethnobotany
Indigenous use of bigflower agoseris, specifically, was not reported in the reviewed literature. Use of other Agoseris species has been reported, please review the related taxa at WesternForbs.org.
Horticulture
Although bigflower agoseris is not readily available as a horticultural species, it does have potential for use in low-maintenance landscaping along roadsides and at rest areas and campgrounds. It produces attractive yellow flowers in early summer and spreads by wind-dispersed seeds. It is easily grown from seed and is a good fit for wild-looking garden, but not for more formal landscaping. Seed does not require stratification and can be sown in spring or fall, although better establishment is reported from fall seeding (see Wildland Seeding and Planting section) (Skinner et al. 2005).
Revegetation Use
Bigflower agoseris tolerates a broad range of soil types, pH from 5.8 to 7.3, grows where annual precipitation averages 12 to 20 in (300-500 mm), and it is adapted to disturbed conditions (Tilley 2013).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among all partners to select appropriate species and seed sources; these are determined by current or projected revegetation requirements and goals (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals as well as through consultation with land managers and stakeholders. Collected seed is distributed for restoration or first increased in seed fields to provide a larger supply of seed (PCA 2015). Production of nursery stock requires less seed than large-scale seeding operations. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for bigflower agoseris, generalized provisional seed zones developed by Bower et al. (2014) may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat to moisture index). In Figure 9, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seed zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USDA FS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Restoration Tool (Richardson et al. 2020) can also guide revegetation planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
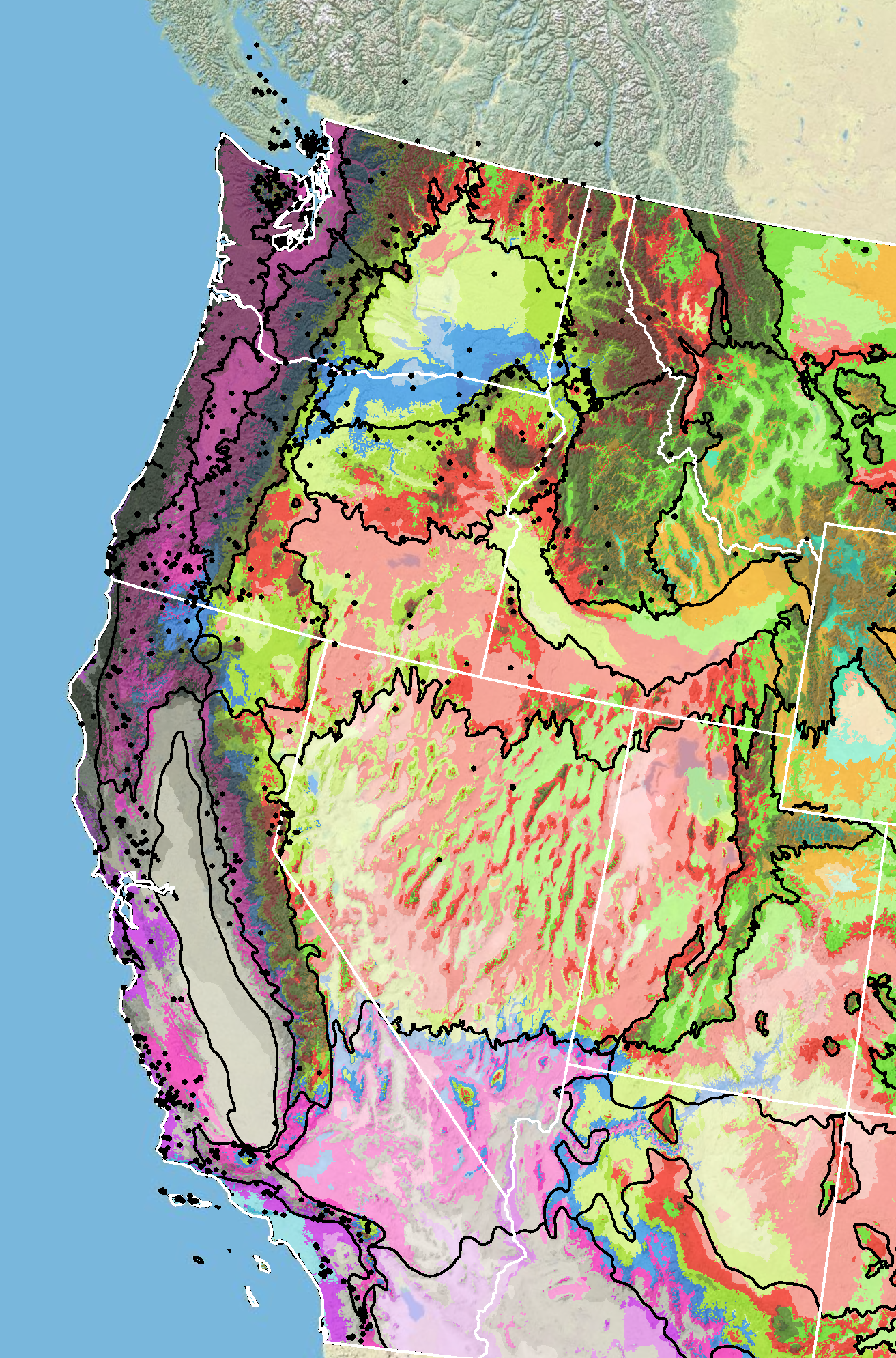
Figure 9. Distribution of bigflower agoseris (black circles) based on geo-referenced herbarium specimens and observational data from 1880–2023 (CPNWH 2017; SEINet 2017; USDI USGS 2020). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USDA FS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USDI USGS.
Releases
As of 2023, there were no bigflower agoseris germplasm releases.
Wildland Seed Collection
Harvesting wildland seed from bigflower agoseris is challenging and requires careful monitoring due to low density plant populations, indeterminate seed maturation, and wind dispersal of ripe seed (Jensen 2007a; Bartow 2015). Jensen (2007a), however, notes that seed retention for bigflower agoseris is much better than for related taxa (Fig. 10).

Figure 10. Closed bigflower agoseris seed head. Photo: USDI BLM OR035 SOS.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (UCIA 2015; Young et al. 2020). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding the collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Seed (Fig. 11) is typically collected in midsummer (early June-late July). Seed maturation is indicated by the spreading of the drying pappus that begins to open the seed head. Mature seed is dark brown to black with an expanded white pappus and detaches easily from the head (Young 2001; Jensen 2007a; Tilley 2013).


Figure 11. Bigflower agoseris seeds with pappi and scale for size (top) and seed body enlarged to see detail (bottom). Photos: USDI BLM ID931 SOS (top) and USDI BLM Or035 SOS (bottom).
Wildland bigflower agoseris seed was commonly collected in July by USDI Bureau of Land Management’s Seeds of Success crews. The earliest collection date for bigflower agoseris was June 30 and the latest was August 1. This was based on eight harvests made from 2009 to 2013 in Oregon, California, and Idaho. The earliest collection date (June 30) occurred in 2006 at an elevation of 1,190 feet (360 m) in Monterey County, California, and in 2010 at an elevation of 3,000 ft (910 m) in Jackson County, Oregon. The latest collection (August 1) was made at 2,500 feet (760 m) in Linn County, Oregon. More than one collection was made at several of the SOS collection sites (BLM SOS). Seed is also typically collected in July at elevations of 3,200 to 4,800 ft (970-1,460 m) in the northern Great Basin (Jensen 2007a).
Collection Methods
Because of the low-density populations and single flowering stems produced by bigflower agoseris, wildland seed is hand collected. Harvests made by SOS field crews were made by removing seeds from the heads while being careful to not let them blow away (DeBolt and Barrash 2013). Farm staff at the USDA ARS Plant Materials Center in Pullman, Washington (WAPMC), clipped seed heads once the pappus had expanded (Skinner 2004). Jensen (2007a) notes that minimizing the amount of inert material collected at the time of harvest makes for easier and quicker seed cleaning.
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of bigflower agoseris.
Post-Collection Management
The milky juice released by damaged or clipped herbage adds moisture to seed collections, which causes leaves, chaff, and seeds to stick together. Careful drying of collected seed lots is important for maintaining seed viability and makes for easier, faster cleaning. The amount of moisture in harvested seed lots depends on the collection method used. Several protocols reported that seed collections were stored in paper bags at room temperature to dry before cleaning (Skinner 2004; DeBolt and Barrash 2013). Bartow (2015) dried seed collections on a tarp. Seed was stirred to reduce the drying time required. No-Pest strips can be placed in breathable collection containers for 2 to 3 days to reduce insect predation.
Seed Cleaning
There are various methods for removing the pappus from the seed, although this step is not necessary for greenhouse propagation (Tilley 2013). For small collections, larger chaff can be removed by hand, and the pappus can be removed by rubbing the seed between rubber mats or rolling it between leather boards (Jensen 2007; Wall and MacDonald 2009). Removal of the pappus by hand, is slow and wearing a mask is recommended (Wall and MacDonald 2009). For large collections, pappus removal can be achieved using a hammermill, brush machine, or Missoula dewinger (Jensen 2007a; DeBolt and Barrash 2013; Tilley 2013; Bartow 2015). Seed cleaning is then finished using a blower, air column separator, or other air screening equipment (Skinner 2004; Jensen 2007a; Wall and MacDonald 2009).
The Bend Seed Extractory described the following procedure for cleaning a small lot of bigflower agoseris seed. First the seed was processed through a small brush machine with a #32 mantle, speed of one, and medium bristles. Then the collection was hand sieved using a #40 USGS screen. Finish cleaning was done using an Office Clipper but screen size and air flow level were not reported (DeBolt and Barrash 2013).
Seed Storage
Bigflower agoseris seed is orthodox (RBG Kew 2023). Clean seed stored at 40 °F (4 °C) and 40% relative humidity by the WAPMC germinated at a very high percentage after 5 years of storage (Skinner 2004).
Seed Testing
There is no Association of Official Seed Analysts (AOSA) rule for testing germination or protocol for examining the viability of bigflower agoseris seed (AOSA 2010; 2016). Tetrazolium chloride testing (TZ) should follow that suggested for other Asteraceae species, which prescribes cutting seeds longitudinally, leaving the distal end intact and soaking for 6 to 12 hours in 0.1% TZ at (86–95 °F [30–35 °C]). The embryos of viable seed will be entirely stained red (AOSA 2010).
Germination Biology
Bigflower agoseris is considered a generalist germinator (Barga et al. 2017). Seed is nondormant, does not require prechilling or other treatments to germinate, and typically germinates within 14 days of exposure to warm temperatures (Tilley 2013; Bartow 2015).
High germination percentages were reported in several studies using controlled conditions. Barga et al. (2017) labeled bigflower agoseris a generalist after evaluating a battery of seed germination treatments. Seed was collected at various times from three populations in northern Nevada to adequately sample the reproductive window. Seed was stored in the dark at 70 °F (21 °C) until testing. Viability of the wild-collected seed was 95 to 100%. Germination was nearly 100% for all treatments, which included 4 weeks of cool (70 °F [21 °C]) or hot (104 °F [40 °C]) afterripening, 0 to 6 weeks of cold moist stratification 36 °F (2 °C), and incubation at 59 °F (15 °C). Rates of germination differed by population but total germination did not, and hot afterripening stimulated faster germination but did not affect total germination (Barga et al. 2017).
Testing by RBG Kew (2023) achieved 100% germination after 14 days for seed on 1% agar exposed to a temperature of 50 °F (10 °C) and an 8:16 hour light-dark cycle and after 28 days at 41 °F (5 °C). Maguire and Overland (1959 cited in Buhler and Hoffman 1999) reported 84% germination for seed kept in the dark at a constant temperature of 59 °F (15 °C) or 68 °F (20 °C), and 68% germination for seed exposed to light and dark at 59 °F (15 °C). Germination was lower at fluctuating temperatures of 68/86 °F (20/30 °C), reaching 20% in dark and 60% with light and dark exposure. Tests were conducted on filter paper in petri dishes within a year of seed collection.
Wildland Seed Yield And Quality
Post-cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 1 (USDA FS BSE 2017). The results indicate that bigflower agoseris seed can generally be cleaned to high levels of purity and seed fill and that viability of fresh seed is generally high. Seed weights (394,361 seeds/lb [869,403 seeds/kg]) reported by RBG Kew (2023) fall within those reported in Table 1, but lower seed weights (274,000 seeds/lb [604,056 seeds/kg]) have been reported elsewhere (Tilley 2013; Bartow 2015).
Table 1. Seed yield and quality of bigflower agoseris seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USDA Forest Service National Seed Laboratory (USDA FS BSE 2017).
| Seed lot characteristic | Mean | Range | Samples (no.) |
| Bulk weight (lbs) | 0.61 | 0.06–2.8 | 9 |
| Clean weight (lbs) | 0.22 | 0.02–0.88 | 9 |
| Clean-out ratio | 0.39 | 0.29–0.57 | 9 |
| Purity (%) | 98 | 95–99 | 9 |
| Fill (%)¹ | 93 | 87–99 | 9 |
| Viability (%)² | 95 | 85–98 | 8 |
| Seeds/lb | 506,834 | 348,112–761,073 | 9 |
| Pure live seeds/lb | 486,438 | 330,915–723,476 | 8 |
¹ 100 seed X-ray test
² Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well.
Agricultural Seed Production
Seed increase plots of bigflower agoseris were established from seed at the USDA Forest Service (USFS) Lucky Peak Nursery in Boise, Idaho (Shaw et al. 2008) and from plugs at the USFS Rocky Mountain Research Station, Provo Shrub Sciences Laboratory (PSSL) in Utah (Jensen 2007b) and the USDA Natural Resources Conservation Service (NRCS), Plant Materials Center in Corvallis, Oregon (ORPMC). Seeding at Lucky Peak occurred in fall 2006 and resulted in good stand emergence. Seed was harvested in the next year (Shaw et al. 2008). Plugs were planted into weed barrier fabric in Provo using a tree planter (Jensen 2007b).


Figure 12. Bigflower agoseris grown for seed in an agricultural setting at USDA Forest Service Lucky Peak Nursery. Photo: USDA Forest Service, Rocky Mountain Research Station.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (UCIA 2015; Young et al. 2020).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Seeding
Preliminary results from direct seeding trials conducted at WAPMC suggest that bigflower agoseris can be seeded in spring or fall. Although stand emergence was much higher with fall seeding, spring seeded stands were acceptable (Skinner 2004). Seed should be planted 0.12 to 0. 5 in (0.3–1.3 cm) deep into a firm weed-free seed bed. The full stand seeding rate is 4 lbs PLS/ac (4.5 kg/ha) (Scheinost et al. 2010; Tilley 2013).
Pest Management
Bigflower agoseris is susceptible to leaf rust and powdery mildew. These diseases can severely stunt plant growth and reduce seed yields (Bartow 2015). Gophers and voles feed on bigflower agoseris roots. Rodent management should be planned for seed production fields (Tilley 2013).
Seed Harvesting
Seed production plots grown at ORPMC (Bartow 2015), were established by planting plugs into weed barrier fabric. Plugs were started in a greenhouse in the winter and transplanted at 1 × 1-ft (0.3 × 0.3 m) spacings in early spring. This system provided weed control and allowed for the recovery of wind-dispersed seed. At ORPMC, seeds ripened from late June through early September. Small fields were harvested as seed heads opened by hand stripping or vacuuming. Larger fields were harvested using a flail vacuum harvester. Multiple harvests were required to maximize seed yields (Bartow 2015).
Seed Yields And Stand Life
Seed production plots at ORPMC established from spring-planted plugs were harvested in the first year and often produced another harvestable crop in year 2. Plants rarely lived more than 2 years, and 40 lbs (18 kg) of seed were harvested from a 0.1-ac (0.04 ha) plot (Bartow 2015).
Nursery Practice
Protocols for growing bigflower agoseris plants in a greenhouse or nursery were provided by several organizations. Removal of the pappus from the seed is not necessary for greenhouse propagation, and container plants are typically ready for outplanting after about 60 days (Tilley 2013).
A nursery in San Francisco, California, grew plants using the following procedure. Seed (2 g) was sown into flats filled with Sunshine mix #4 by mixing the seed with the media and spreading it over the surface. Seed sown on June 1 resulted in just 10% germination within 14 days. After 14 days of growth, seedlings were transplanted into 2 × 7-in (5 × 18 cm) tubes filled with a standard potting mix of peat moss, fir bark, perlite, and sand. Transplant survival was 95%. Once established (1–2 months after transplanting), seedlings were dressed with a balanced fertilizer (NPK 13-13-13 [1 g]). Flower removal was required for plants kept in the greenhouse longer than 3 months (Young 2001).
The WAPMC provided the following protocol for producing root-tight plugs. Seed was sown in 10 in3 (164 cm3) Ray Leach Super cell cone-tainers filled with Sunshine #4 media. Seed was covered lightly with soil and a thin layer of pea gravel to keep seeds from floating. Cone-tainers were watered thoroughly and kept moist until seeds germinated. Germination started at day 5 and was complete after 2 weeks. Seedlings developed quickly. Cone-tainers were watered deeply every other day and fertilized weekly with a complete water-soluble fertilizer with micronutrients. The establishment phase was 2 weeks, and the active growth phase was 60 days. Plants were moved to a cold frame in late March or early April and hardened for 2 to 4 weeks (Skinner 2004).
At the Idaho Botanical Garden in Boise, Idaho, bigflower agoseris seed was sown directly into containers filled with 2:1:5 mix of lava fines, perlite, and Sunshine mix #4. Five seeds were sown shallowly into the 3 × 5.5-in (7.4 × 14 cm) containers on March 22 and covered with 0.1 in (0.3 cm) of chicken grit. Containers were watered and put outside. At 12 days, 90% of the seeds had germinated. Seedlings were divided and transplanted into individual containers 3 weeks later. Transplant survival was 99%. The establishment phase was about 1 month. Containers were allowed to dry slightly between waterings, and a weak fish emulsion fertilizer was applied twice a month from April through June. Plants remained outdoors until outplanting in early October. Flower stalks were removed as they appeared (DeBolt and Barrash 2013).
The PSSL planted seeds vertically to the depth of the seed body into q-plugs (7.5 in3 [123 cm3]). Germination was rapid at greenhouse temperatures (70 °F [20 °C] days, 50 °F [10 °C] nights). Most seed germinated within 1 week. Seedlings were considered established after 1 to 2 weeks. Plants were watered twice a week. The active growth period was 5 weeks or more. A single seedling transplanted into a large pot (6-in [15 cm] diameter, 36 in [91 cm] tall) that was kept moist in the greenhouse produced 50 seed heads before senescing (Jensen 2007b).
Wildland Seeding And Planting
Bigflower agoseris should be seeded 0.12 to 0.5 in (0.3-1.2 cm) deep into a weed-free seed bed (Scheinost et al. 2010; Tilley 2013). Seeding can be done in the spring, but emergence is improved with fall seeding (Scheinost et al. 2010). The recommended full stand seeding rate is 4 lbs PLS/ac (4.5 kg/ha), so this amount can be adjusted in accordance with the amount of bigflower agoseris in the seed mix, which would be minimal. Post-seeding management should be based on those key species in the establishing community, but grazing should be deferred for at least two growing seasons (Tilley 2013). Increasing the seeding rate can improve establishment, but if costs or seed supply prohibit a high seeding rate consider concentrating forb seed in several small areas. These islands of establishment can provide a seed source to allow spread into the adjacent areas (Scheinost et al. 2010).
Establishment of bigflower agoseris was mixed in field studies. Initial establishment was high, but persistence was low in a field study conducted at WAPMC where annual precipitation averages 20 in (510 mm) (Scheinost et al. 2010). Study plots were prepared by chiseling and disking existing grain stubble. Plots were cultivated with a Glencoe cultivator and packed with Brillion roller immediately prior to seeding. Seed was collected from a seed increase plot established from locally collected seed. Seed was planted 0.25 to 0.5 in (0.6-1.3 cm) deep in 20 ft (6 m) long rows at 20 or 40 seeds/linear foot (66 or 131/m) using a Hege 1000 plot seeder. Plots were seeded in early fall (mid-Sept) 2002, late fall (late Oct) 2002, and early spring (mid- to late April). Emergence was significantly higher with fall seeding than spring. Initial year seedling counts were exceptional with 80% of seed emergence. Plants were short lived, and almost no plants remained in year 2 or 3. Researchers suggested that early-seral bigflower agoseris could be included in seeding mixes to provide initial cover (Scheinost et al. 2010).
Emergence was much lower in field studies in Nevada. Increasing the seeding depth reduced emergence and adding row cover (N-sulate (DeWitt Co, Sikeston, MO) sometimes increased emergence at sites in Nevada. The Nevada field study took place in Orovada and Wells. The 10-year annual precipitation average in Orovada was 13 in (320 mm) where soils were very fine sandy loams over silt loams. Wells precipitation averaged 12 in (305 mm), and soils were silt loams. Bigflower agoseris seed was planted at depths of 0.5, 1, 1.4, and 1.6 in (1.4, 2.6, 3.6 and 4.0 cm) in late fall using a tractor drawn Hege seeder. Increasing the seeding depth from 0.5 (1.4 cm) to 1 in (2.6 cm) reduced emergence in 2013 by 82% at Orovada and by 12.4% at Wells. In 2014, increasing the seeding depth from 0.5 (1.4 cm) to 1 in (2.6 cm) increased emergence by 13.1% at Orovada. Overall emergence was low for bigflower agoseris, averaging 0.6% across sites, years, seeding depths, and with or without row cover. The use of row cover increased emergence at Orovada over control plots in both years but not at Wells. Row cover was installed immediately after seeding and remained in place until mid-April. Growing conditions were more favorable in 2013 than in 2014. Mean emergence at Orovada in 2013 was 3.85% with row cover and 0% without and in 2014 was 0.85% with row cover and 0% without. At Wells in 2013, emergence was 4.9% with row cover and 4.05% without (Jensen et al. 2022).
Use of row cover to establish islands of native forbs in big sagebrush communities was field tested at four sites in Utah (three in Wyoming big sagebrush [Ecoregions 13 and 19] and one in mountain big sagebrush [Ecoregion 20]) (Landeen et al. 2021). Emergence was often greater for Agoseris (heterophylla and grandiflora species) with row cover, but this benefit was short lived. Agoseris species were seeded in a mix with many other forbs. The seeding rate for bigflower agoseris was 2.95 PLS/ft2 (31.75 PLS/m2), which is higher than typical. Sites were cleared of vegetation using a Dixie Harrow, seed was broadcast using a hand-crank seeder, and a Brillion imprinter and lightweight chain was used to incorporate the seed into the soil. Across all sites, Agoseris species emergence was significantly greater in covered than noncovered plots (P < 0.001). Agoseris species persisted through the second year on all sites, but density dropped significantly (P < 0.001) in the second year (Table 2). The benefits of ground cover were ephemeral and did not result in persistence. In control plots (unseeded with and without row cover), undesirable species increased with row cover treatments suggesting that increases in competition could accompany row cover treatments (Landeen et al. 2021).
Table 2. Mean density of Agoseris species (heterophylla and grandiflora) with and without row cover (N-sulate) (DeWitt Co, Sikeston, MO) treatments following seeding at four sites in Utah (Landeen et al. 2021).
| Year | ARTRW (Ecoregion 19) – Fountain Green | ARTRV (Ecoregion 20) – Gordon Creek | ARTRW (Ecoregion 13) – Hatch Ranch | ARTRW (Ecoregion 13) – Lookout Pass | ||||
| C | NC | C | NC | C | NC | C | NC | |
| 1 | 6.68 | 1.56 | 6.91 | 3.34 | 14.78 | 2.37 | 8.47 | 1.78 |
| 2 | 0.28 | 0.17 | 4.05 | 2.94 | 2.40 | 1.12 | 0.85 | 1.24 |
| 5 | 0 | 0 | 0.07 | 0 | 0.20 | 0.27 | 0 | 0.07 |
Bold values within a site show significantly different plant densities between covered and noncovered plots (P < 0.001). C = covered, NC = not covered. ARTRW = Artemisia tridentata subsp. wyomingensis, ARTRV = A. t. subsp. vaseyana.
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Sarah Barga, Research Botanist, USDA Forest Service, Rocky Mountain Research Station and Amy Bartow, Seed and Plant Production Manager at USDA Natural Resources Conservation Service Corvallis Plant Materials Center.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Abella, S.R.; Springer, J.D. 2006. Canopy-tree influences along a soil parent material gradient in Pinus ponderosa–Quercus gambelii forests, northern Arizona. Journal of the Torrey Botanical Society. 135(1): 26-36.
Anicito, K.R. 2013. A holistic approach to Mima mound prairie restoration. Cheney, WA: Eastern Washington University. Thesis. 150 p.
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Baird, G.I. 2004. Two new varieties of Agoseris (Asteraceae: Lactuceae). SIDA, Contributions to Botany. 21(1): 267-274.
Baird, G.I. 2006. Agoseris. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 19 Magnoliophyta: Asteridae (in part): Asteraceae, part 1. New York, NY: Oxford University Press: 323-334.
Barga, S.; Dilts, T.E.; Leger, E.A. 2017. Climate variability affects the germination strategies exhibited by arid land plants. Oecologia. 185: 437-452.
Bartow, A. 2015. Native seed production manual for the Pacific Northwest. Corvallis, OR: U.S. Department of Agriculture, Natural Resources Conservation Service, Corvallis Plant Materials Center. 191 p.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Beidleman, L.H.; Kozloff, E.N. 2003. Flowering plant species. In: Plants of the San Francisco Bay Region, 3rd ed. Berkeley, CA: University of California Press: 68-400.
Bohlman, G.N.; North, M.; Safford, H.D. 2016. Shrub removal in reforested post-fire areas increases native plant species richness. Forest Ecology and Management. 374: 195-210.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Buhler, D.D.; Hoffman, M.L. 1999. Andersen’s guide to practical methods of propagating weeds and other plants. Lawrence, KS: Allen Press. 248 p.
Chirco, E.; Turner, T. 1986. Species without AOSA testing procedures. The Newsletter of the Association of Official Seed Analysts. 60(2): 2-66. Updated April 2007.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2020. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K. 1994. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Volume 5: Asterales. Bronx, NY: The New York Botanic Garden. 496 p.
Davies, K.W.; Bates, J.D.; Miller, R.F. 2007. Environmental and vegetation relationships of the Artemisia tridentata spp. wyomingensis alliance. Journal of Arid Environments. 70(3): 478-494.
Debolt, A.M.; Barrash, K. 2013. Propagation protocol for production of container (2.875 x 5.5 in plant band) Agoseris grandiflora (Nutt.) Greene plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2023 March 6].
Dyrness, C.T. 1973. Early stages of plant succession following logging and burning in the western Cascades of Oregon. Ecology. 54(1): 57-69.
Eldredge, E.; Novak-Echenique, P.; Heater, T.; Mulder, A.; Jasmine, J. 2013. Plants for pollinator habitat in Nevada. Tech. Note NV 57. Reno, NV: U.S. Department of Agriculture, Natural Resources Conservation Service. 65 p.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Halpern, C.B. 1989. Early successional patterns of forest species: Interactions of life history traits and disturbance. Ecology. 70(3): 704-720.
Hemleben, V.; Bachmann, K.; Price, H.J. 1978. Ribosomal gene numbers in Microseridinae (Compositae: Cichorieae). Experientia. 34(11): 1452-1432.
Hermann, F. 1966. Notes on western range forbs: Cruciferae through Compositae. Agric. Handb. 293. Washington, DC: U.S. Department of Agriculture, Forest Service. 365 p.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
Hitchcock, C.L.; Cronquist, A. 2018. Flora of the Pacific Northwest: An illustrated manual. Second Ed. Giblin, D.E.; Legler, B.S.; Zika, P.F.; Olmstead, R.G., eds. Seattle, WA: University of Washington Press. 882 p.
Jensen, S. 2007a. Propagation protocol for production of plug (7.5 cu. In Qplug [International Horticultural Technologies LLC. Hollister, CA) Agoseris grandiflora (Nutt.) Greene plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2023 March 6].
Jensen, S.; Christensen, W.F.; Roundy, B.; Anderson, V.J.; Kitchen, S.G.; Allphin, L. 2022. Temporal and spatial factors influence native forb emergence more than sowing depth. Rangeland Ecology and Management. 83: 41-49.
Johnson, C.G.; Swanson, D.K. 2005. Bunchgrass plant communities of the Blue and Ochoco Mountains: A guide for managers. R6-NR-ECOL-TP-06-98. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. 128 p.
Kofron, C.P.; Rutherford, C.; Clark, R.E.; Woodbury, D.; Olson, J.; Holland, R.F. 2013. Review of the purple amole (Chlorogalum purpureum (Agavaceae): A threatened plant in the Coast Ranges of central California. Bulletin, Southern California Academy of Sciences. 112(1): 38-48.
Lady Bird Johnson Wildflower Center [LBJWC]. 2020. Agoseris grandiflora (Nutt.) Greene. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants-main [Accessed 2023 March 6].
Landeen, M.; Jones, C.; Jensen, S.; Whittaker, A.; Summers, D.D.; Eggett, D.; Petersen, S.L. 2021. Establishing seed islands for native forb species on rangelands using N-Sulate ground cover fabric. Native Plants Journal. 22(1): 51-63.
Leger, E.; Barga, S. 2017. Climate niche comparisons of cold desert forbs using herbarium records. In: Kilkenny, F; Edwards, F.; Irwin, J., Barga, S. eds. Great Basin Native Plant Project: 2016 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 47-57.
Lesica, P. 2012. Manual of Montana vascular plants. Fort Worth, TX: Botanical Research Institute of Texas (BRIT) Press. 779 p.
Luna, T. 2005. Propagation protocol for production of container (plug) Erigeron speciosus (Lindl.) DC. plants 116 ml (7.0 cu. in). Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2018 January 3].
Luna, T.; Mousseaux, M.R.; Dumroese, R.K. 2018. Common native forbs of the northern Great Basin important for greater sage-grouse. Gen. Tech. Rep. RMRS-GTR-387. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station; Portland, OR: U.S. Department of the Interior, Bureau of Land Management, Oregon-Washington Region. 76 p.
Martin, A.C.; Zim, H.S.; Nelson, A.L. 1951. American wildlife and plants: A guide to wildlife food habits. New York, NY: Dover Publications. 500 p.
McDonald, P.M. 1999. Diversity, density, and development of early vegetation in a small clear-cut environment. Res. Paper PSW-RP-239. Albany, CA: U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station. 22 p.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Pavek, P.; Erhardt, B.; Heekin, T.; Old, R. 2012. Forb seedling identification guide for the Inland Northwest: Native, introduced, invasive, and noxious species. Pullman, WA: U.S. Department of Agriculture, Natural Resources Conservation Service, Pullman Plant Materials Center. 144 p.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Pyle, W.H. 1992. Response of brood-rearing habitat of sage grouse to prescribed burning in Oregon. Corvallis, OR: Oregon State University. Thesis. 47 p.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Roche, C.T.; Sheley, R.L.; Korfhage, R.C. 2008. Native species replace introduced grass cultivars seeded following wildfire. Ecological Restoration. 26(4): 321-330.
Royal Botanic Gardens Kew [RBG Kew]. 2017. Seed Information Database (SID). Version 7.1. http://data.kew.org/sid/
Scheinost, P.L.; Skinner, D.M.; Stannard, M.E. 2010. Evaluation of planting time and survivability of 16 forb and 2 grass species native to the Inland Northwest. Plant Materials Tech Note Note 20. Pullman, WA: U.S. Department of Agriculture, Natural Resources Conservation Service. 20 p.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2020. SEINet Regional Networks of North American Herbaria. https://Symbiota.org/docs/seinet
Shaw, N.; Cox, R.; Flegee, C.; Sloan, J.; St. John, L. 2008. Native forb selection, seedling establishment, species interactions, and post-fire seedings. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2007 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 145146.
Sikes, K.G.; Muir, P.S. 2009. A comparison of the short-term effects of two fuel treatments on chaparral communities in southwest Oregon. Madroño. 56(1): 8-22.
Skinner, D.M. 2004. Propagation protocol for production of container (plug) Agoseris grandiflora (Nutt.) Greene plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2023 March 6].
Stickney, P.F. 1986. First decade plant succession following the Sundance forest fire, northern Idaho. Tech. Rep. INT-GTR-197. Ogden, UT: USDA Forest Service, Intermountain Research Station. 26 p.
Swanson, D.O. 1970. Roosevelt elk-forest relationships in the Douglas-fir region of the southern Oregon Coast Range. Ann Arbor, MI: University of Michigan. Dissertation. 173 p.
Tidestrom, I.; Shantz, H.L.; Sampson, A.W. 1925. Flora of Utah and Nevada. Contributions from the United States National Herbarium. 25: 1-665.
Tilley, D. 2013. Plant guide: Bigflower agoseris (Agoseris grandiflora [Nutt.] Greene). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 2 p.
Tisdale, E.W. 1976. Vegetational responses following biological control of Hypericum perforatum in Idaho. Northwest Science. 50(2): 61-75.
Titus, J.H.; Moore, S.; Arnot, M.; Titus, P.J. 1998. Inventory of the vascular flora of the blast zone, Mount St. Helens, Washington. Madroño. 45(2): 146-161.
Tueller, P.T.; Platou, K.A. 1991. A plant succession gradient in a big sagebrush/grass ecosystem. Vegetatio. 94(1): 57-68.
U.S. Department of Agriculture, Forest Service [USFS]. 1937. Range Plant Handbook. Washington, DC: U.S. Department of Agriculture, Forest Service. 816 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2021. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDA Natural Resources Conservation Service [USDA NRCS]. 2023. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2023. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 45 p.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2020. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Wall, M.; MacDonald, J. 2009. Processing seeds of California native plants for conservation, storage, and restoration. Claremont, CA: Rancho Santa Ana Botanic Garden. 157 p.
Waring, R.H.; Major, J. 1964. Some vegetation of the California coastal redwood region in relation to gradients of moisture, nutrients, light, and temperature. Ecological Monographs. 34(2): 167-215.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Young, B. 2001. Propagation protocol for production of container plants (Deepot 16) Agoseris grandiflora (Nutt.) Greene plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2023 March 6].
Young, S.A.; Schrumpf, B.; Bouck, M.; Moore, M. 2020. How the Association of Official Seed Certifying Agencies (AOSCA) tracks wildland sourced seed and other plant propagating materials. Logan, UT: Utah State University, Utah Agricultural Experiment Station. 36 p.
Zouhar, K. 2013. Plant succession following clearcutting and slash burning in the western Cascade Range, Oregon. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer), Missoula, MT. Available: www.fs.usda.gov/database/feis/research_project_summaries/HJ_Andrews/all.html [2023, April 20].
How to Cite
Gucker, C.L.; Shaw, N.L. 2023. Bigflower agoseris (Agoseris grandiflora [Pursh] Raf.). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. https://westernforbs.org/species/bigflower-agoseris-agoseris-grandiflora/