Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
February 2024
Nomenclature
Cutleaf daisy (Erigeron compositus) belongs to the Astereae tribe, Conyzinae subtribe, and Osteocaulis section of the Asteraceae family (Nesom 2008). Cutleaf daisy was first collected in 1805 by Lewis and Clark from a site along the Clearwater River near modern-day Lewiston, Idaho (Reveal et al. 1999).
Family
Asteraceae – Aster family
Genus
Erigeron
Species
compositus
NRCS Plant Code
ERCO4 (USDA NRCS 2023)
Subtaxa
The Flora of North America does not recognize any subtaxa (Nesom 2006).
Synonyms
Erigeron compositus var. compositus Pursh, E. c. var. discoideus A. Gray, E. c. var. glabratus Macoun, E. c. var. multifidus (Rydberg) J. F. Macbride & Payson, E. c. var. pinnatisectus A. Gray, E. c. var. typicus Hook, and E. gormanii Greene (Nesom 2006; ITIS 2023).
Common Names
Cutleaf daisy, alpine daisy, compound fleabane, daisy fleabane, dwarf mountain fleabane, fernleaf fleabane (Currah et al. 1983; Mee et al. 2003; Nesom 2008; LBJWC 2022; USDA NRCS 2023).
Chromosome Number
There are sexual and apomictic forms of cutleaf daisy. Sexual forms are primarily diploid (2n = 18) (Beaman 1977; Noyes et al. 1995). Chromosome numbers of 2n = 36 and 2n = 35 were also found in sexual plants and more polyploids are likely to be discovered as only a fraction of the range occupied by sexual forms was evaluated by Noyes et al. (1995). Apomictic forms are usually hexaploid (2n = 54), but counts range from 2n = 36 to 2n = 80 (Beaman 1977; Noyes and Soltis 1996).
Hybridization
Although hybridization is considered likely between closely related Erigeron taxa (Noyes et al. 1995), hybrids were not identified in the reviewed literature.
Distribution
Cutleaf daisy has a broad North American distribution (Nesom 2006; USDA 2023). In Canada, it occurs in all territories but Ontario. In the United States, populations occur in Alaska and states from Washington east to the Dakotas and south to Colorado, New Mexico, and California. This species is also thought to occur in Greenland, the Arctic bridge gap (at elevations as low as 30-660 ft [10-200 m]), and in far eastern Russia (Nesom 2006; USDA 2023).
Populations of apomictic and sexual plants are segregated, and sexual forms are more common and more broadly distributed than apomictic forms (Beaman 1977; Noyes and Soltis 1996). Apomictic populations are usually located more than 60 miles (100 km) from the nearest sexual populations. In places where sexual and asexual population ranges overlap, strong ecological separation exists. In most cases, apomictic plants grow at higher elevations and tolerate more extreme climate and soil conditions than sexual plants (Noyes and Soltis 1996). Based on chromosome counts, embryological studies, and examination of pollen of many herbarium specimens, the greatest diversity of both forms of the species centers in southwestern Montana (Beaman 1977).
Habitat And Plant Associations
Cutleaf daisy has been reported in a variety of habitats and plant associations ranging from low-elevation grasslands and shrublands to montane woodlands and forests and high-elevation alpine communities (Cronquist et al. 1994; Nesom 2006). Plants often occur in open, rocky or sandy sites and tolerate dry conditions and very cold temperatures (Rydberg 1914; Andersen and Holmgren 1996; Mee et al. 2003; Blackwell 2006). Recorded plant associates include sagebrush (Artemisia spp.), rabbitbrush (Chrysothamnus and Ericameria spp.), quaking aspen (Populus tremuloides), and conifers (Nesom 2006; Lesica 2012). Cutleaf daisy has been identified as an important forb in the black sagebrush/Idaho fescue (A. nova/Festuca idahoensis) rangeland cover type in the northern Rocky Mountains west of the Continental Divide at elevations of 6,200 to 9,100 ft (1,900-2,800 m). In these habitats, temperatures may range from -40 °F (-40 °C) to 100 °F (40 °C), and annual precipitation averages 14 to 16 in (360-410 mm) (Shiflet 1994).
Cutleaf daisy has been described as a component of several grassland (Fig. 1) and shrubland communities from Alaska to Colorado (Vestal 1917; Ramaley 1919; Tisdale 1947; Mueggler and Stewart 1980; Lloyd et al. 1994; Vetter 2000). On the western ridge of Kathul Mountain in eastern interior Alaska, cutleaf daisy was found growing in both grass-dominated steppe (820-3,100 ft [250-940 m]) and shrub-dominated tundra (>3,120 ft [950 m]) vegetation but more commonly in the steppe. In this part of interior Alaska, the climate is subarctic continental with an average January temperature of -9 °F (-23 °C), an average July temperature of 59 °F (15 °C), and an average annual precipitation of 12 in (308 mm) (Lloyd et al. 1994).
On Eagle Bluff 1,440 ft (440 m) above the Yukon River in interior Alaska, cutleaf daisy was documented in steppe but not forested vegetation. In this vegetation survey, steppe vegetation was described as being dominated by prairie sagewort (A. frigida), bluebunch wheatgrasss (Pseudoroegneria spicata), and xerophytic herbs. Forests were dominated by quaking aspen, balsam poplar (Populus balsamifera), and white spruce (Picea glauca) (Wesser and Armbruster 1991). In a vegetation survey of the Alaska highway corridor in the Yukon Territory, cutleaf daisy was typically documented in purple reedgrass (Calamagrostis purpurascens) grasslands. These grasslands were described as free of permafrost in the top 3 ft (1 m) of soil and occupying steep south slopes with moderate to high erosion potential (Orlóci and Stanek 1980).
In southern Saskatchewan where the climate was considered semi-arid continental, cutleaf daisy was associated with blue grama-tufted milkvetch (Bouteloua gracilis–Astragalus spatulatus) mixed-grass prairie (Looman and Campbell 1960). In southern interior British Columbia, it was found in bluebunch wheatgrass-fescues (Festuca spp.) and Sandberg bluegrass-cheatgrass (Poa secunda–Bromus tectorum) grasslands (Tisdale 1947). In western Montana, cutleaf daisy was commonly associated with low sagebrush (Artemisia arbuscula)/Idaho fescue and big sagebrush (A. tridentata)/Idaho fescue and grew in grassland types dominated by fescues or needle and thread (Hesperostipa comata) (Mueggler and Stewart 1980, 1981). In Colorado, it was found growing in xerophytic grasslands in the upper foothills, montane, and subalpine zones (Ramaley 1919).

Figure 1. Cutleaf daisy growing in a grassland in Oregon. Photo: USDI Bureau of Land Management (BLM) OR931 Seeds of Success (SOS).
Cutleaf daisy was noted in forest understories from California to Colorado. It was recorded in the montane zone of Lee Vining Canyon near Yosemite National Park (9,475 ft [2,888 m]) alongside granite prickly phlox (Leptodactylon pungens) and scattered Sierra lodgepole pine (Pinus contorta var. murryana) and limber pine (P. flexilis) (Kimball et al. 2008). It was also found growing in the understory of a pure subalpine larch (Larix lyalli) stand at 9,200 ft (2,800 m) elevation in Montana’s Anaconda-Pintlar range. Trees were described as slender and less than 20 ft (6 m) tall on a steep mesic site (Arno and Habeck 1972). In Rocky Mountain National Park, it was recorded in ponderosa pine (P. ponderosa) forests in the foothills and lower elevations of south and west slopes (7,000 to 9,000 ft [2,100-2,700 m) where crown cover was an estimated 35% (Sperry 1936).
Cutleaf daisy was reported in a variety of alpine habitats (Fig. 2) in the western United States (McNeal 1976; Douglas and Bliss 1977; Taye 1995; Lavin and Wojciechowski 2002; Aho and Weaver 2010). In high-elevation meadows in the Olympic Mountains of Washington, cutleaf daisy was reportedly associated with arid meadows on scree slopes and other exposed sites (Belsky and del Moral 1982). In the Sweetwater Mountains of California and adjacent western Nevada, cutleaf daisy was widespread in high-elevation sagebrush-grasslands in alpine tundra (Lavin and Wojciechowski 2002). Cutleaf daisy was documented in many alpine communities on Mount Washburn in Yellowstone National Park, Wyoming, but constancy and cover were highest in woolly groundsel-spiny milkvetch (Packera cana–Astragalus kentrophyta) communities on south-facing slopes (Aho and Weaver 2010). Frequency of cutleaf daisy was 7.9% on moraine and 13.6% in dry meadow vegetation in the alpine zone near Schoolroom Glacier in Wyoming’s Teton Range. The moraine sites were described as continually disturbed and unstable with very loose deposits and small erosion channels from snowmelt (Spence and Shaw 1983). In Utah, cutleaf daisy was reported in cushion plant communities that occupied windswept ridges with substrates that commonly experienced frost heaving. The species was also found in gravelly, barren bedrock and talus/scree habitats and dry, rocky meadows where pocket gopher disturbances were common (Taye 1995).

Figure 2. Cutleaf daisy growing at a high-elevation site in Oregon. Photo: USDI BLM OR090 SOS.
Elevation
Cutleaf daisy occurs across a broad elevation range of 5,900 to 14,100 ft (1,800-4,300 m) (Nesom 2006). It has been found at all elevations in Montana (Lesica 2012) and at elevations above 7,000 ft (2,100 m) throughout the Intermountain West, the Great Basin, and California (Munz and Keck 1973; Hickman 1993; Mee et al. 2003; Blackwell 2006).
Soils
The habitats and plant associations supporting cutleaf daisy occupy a variety of soil textures (fine to coarse) and parent substrates. Soils are often dry, gravelly or rocky (Fig. 3), and densely packed (St. John and Warren 1937; Craighead et al. 1963; Hermann 1966; Löve et al. 1971; Currah et al. 1983; del Moral 1983; Mee et al. 2003; Lesica 2012) but vary across the species range. Cutleaf daisy is commonly found growing in rangeland typified by shallow soils over limestone deposits (Shiflet 1994).

Figure 3. Cutleaf daisy growing in gravelly, rocky soil in Crater Lake National Park, Oregon. Photo: ©Keir Morse 2010, CalPhotos.
Cutleaf daisy has been documented in habitats ranging from excessively drained or dry to relatively moist. In a vegetation survey of the Alaska highway in the Yukon Territory, cutleaf daisy was recorded in purple reedgrass grasslands in excessively drained rocky soils composed of sand and silt (Orlóci and Stanek 1980). In a local ecology study of California’s Bishop Creek Watershed, cutleaf daisy occurred in 11 of 263 montane to alpine plots where it had a moisture preference for dry sites (Kimball et al. 2004). When water availability, soil texture, and disturbance were evaluated for 194 sagebrush sites in eastern Montana, cutleaf daisy was associated with relatively moist sites (≥4 in [11 cm]) water holding capacity above the Bk horizon) and slightly to heavily disturbed sites (Harvey 1990).
In southwestern Saskatchewan, cutleaf daisy has been found in mixed-grass prairie on calcareous, loams or sandy loams. Soils were often gravelly and lightly to moderately eroded with a pH of 8 and a moisture holding capacity of 61% (Looman and Campbell 1960). Along major rivers in eastern Washington and northern Idaho, cutleaf daisy was documented in dry, rocky, compacted soils, and in west-central Montana, it was found associated with flat, rocky, densely packed soils (Noyes et al. 1995). At Craters of the Moon National Monument in south-central Idaho, it was recorded on young or early seral lava flows supporting very little vegetation cover (Day and Wright 1985). In the montane zone of Lee Vining Canyon near Yosemite National Park, cutleaf daisy was found in mostly granitic gravel (Kimball et al. 2008). In a survey of dolomite, sandstone, and granite soils in the bristlecone pine (Pinus longaeva) zone in the White Mountains of California, cutleaf daisy was recorded on sandstone and granite soils but not on dolomite soils (Wright 1963). In foothills grassland vegetation in the Colorado Front Range, cutleaf daisy was restricted to places with packed granite-gravel soils (Vestal 1917) and was associated with xerophytic grasslands (>8,420 ft [2,570 m]) with coarse-grained soils of disintegrated rock where water content could be less than 7% (Ramaley 1919).
Plants may be adapted to soils at their place of origin. In a greenhouse study, seed from ultramafic habitats grew well in ultramafic soils, while seed from non-ultramafic soils grew poorly. The ultramafic soils used in the greenhouse study were olivine-rich rock or dunite collected near the Scheele mine in Skagit County, Washington (Kruckeberg 1967).
Description
Cutleaf daisy is a small, taprooted, caespitose perennial forb (Munz and Keck 1973; Currah et al. 1983; Hickman 1993; Cronquist et al. 1994; Mee et al. 2003). The species is highly variable, and variability is found within populations and without geographic patterns across its range (Cronquist et al. 1994). The caudex is simple or branched, covered by persistent leaves, and typically short and thick, but it can also be slender and rhizome-like (Munz and Keck 1973; Currah et al. 1983; Cronquist et al. 1994; Mee et al. 2003; Nesom 2006; Lesica 2012). Plants produce few to many erect stems (1-10 in [2.5-25 cm] tall) that are simple, often leafless, and terminate in a single flower head (Fig.4; Craighead et al. 1963; Hermann 1966; Taylor 1992; Nesom 2006; Dorn and Dorn 2007; Lesica 2012). Plants can occur as clumps up to 16 in (40 cm) in diameter (Currah et al. 1983). Herbage is glandular and hairy (hispido-pilose, hispid-hirsute, hirsute, or subglabrous) (Hermann 1966; Munz and Keck 1973; Hickman 1993; Cronquist et al. 1994; Nesom 2006; Lesica 2012). At the highest elevations, plants may be only 1 or 2 in (2.5-5 cm) tall and densely hairy with short bristly white hairs (Blackwell 2006).

Figure 4. Flowering cutleaf daisy growing in Utah. Photo: USDI BLM UT930 SOS.
Plants develop stout, well-branched taproots (Currah et al. 1983; Hickman 1993). Plants excavated from very stony soils on the east slope of the Medicine Bow Range in Wyoming produced slender taproots that divided into a system of fine laterals at depths of 4 in (10 cm) or more. Roots supported symbiotic endotrophic mycorrhizae (Daubenmire 1941).
Leaves are mostly basal and if present, stems leaves are alternate, mostly entire, and bract like (Currah et al. 1983; Hickman 1993; Cronquist et al. 1994; Mee et al. 2003; Nesom 2006). Leaf blades are spatulate, obovate, or obovate-spatulate, 0.4 to 2 in (1-5 cm) long, and 0.2 to 0.5 in (0.4-1.2 cm) wide with long petioles (Munz and Keck 1973; Nesom 2006; Lesica 2012). They are two to four-ternately lobed or dissected into crowded, narrow, linear lobes (Craighead et al. 1963; Hermann 1966; Munz and Keck 1973; Currah et al. 1983; Cronquist et al. 1994; Nesom 2006; Lesica 2012).
Flower heads are solitary, terminal, and radiate or disciform (Currah et al. 1983; Cronquist et al. 1994; Blackwell 2006; Nesom 2006; Lesica 2012). Heads are 0.5 or 1.5 in (1.3-3.8 cm) wide above a campanulate involucre (5-10 by 8-20 mm) with purple-tipped, hirsute phyllaries in an equal two or three series of whorls (Munz and Keck 1973; Andersen and Holmgren 1976; Hickman 1993; Cronquist et al. 1994; Mee et al. 2003; Blackwell 2006; Nesom 2006; Dorn and Dorn 2007; Lesica 2012). Disk florets are perfect, tubular, and yellow (Fig.5). Ray florets are pistillate, and when present, are crowded, white, blue, or pink and number 20 to 60 (Fig. 6; Craighead et al. 1963; Munz and Keck 1973; Andersen and Holmgren 1976; Currah et al. 1983; Hickman 1993; Cronquist et al. 1994; Blackwell 2006; Dorn and Dorn 2007; Lesica 2012). Ray florets are 6 to 12 mm long and the corollas of disk florets are 3 to 5 mm long (Mee et al. 2003; Nesom 2006; Lesica 2012). Fruits are two-nerved achenes with sparsely strigose-hirsute faces (Nesom 2006) and a pappus of 12 to 20 coarse bristles (Currah et al. 1983; Hickman 1993; Cronquist et al. 1994; Nesom 2006). The single seed is 1.6 to 2.7 mm long (Currah et al. 1983; Nesom 2006; Lesica 2012).

Figure 5. Cutleaf daisy flower with only disk florets, note the hirsute phyllaries in an equal series of whorls. Photo taken on August 1, 2009, in Mono County, CA. Photo: ©2009 Steve Matson, CalPhotos.

Figure 6. Cutleaf daisy flower with both disk and ray florets. Photo taken on August 22, 2006, in El Dorado County, CA. Photo: ©2006 Steve Matson, CalPhotos.
Cutleaf daisy is distinguished from other Erigeron species by its compact above-ground form, usually branching stout caudex, and dissected leaves (Noyes et al. 1995). Apomictic plants are notoriously variable in vegetative and floral morphology. Leaf size, degree of dissection, and pubescence vary, and ray florets range from absent to conspicuous. Sexual forms are more consistent morphologically and produce both disk and ray florets (Noyes and Soltis 1996).
Reproduction
Plants produce abundant seed in the field (Fig. 7) (Currah et al. 1983).

Figure 7. Cutleaf daisy seedhead. Photo taken on July 15, 2006, in Crater Lake National Park, Oregon. Photo: ©2010 Keir Morse, CalPhotos.
Phenology
Cutleaf daisy flowers are common from June to August (Nesom 2006) but can be found as early as April and as late as September (Craighead et al. 1963; Andersen and Holmgren 1976; Cronquist et al. 1994; Mee et al. 2003; Blackwell 2006; Dorn and Dorn 2007). in prairie habitats of southern Canada, plants typically emerge and produce new growth in late April, form flower buds in May, produce flowers in late May or early June, and mature seed in June or July (Currah et al. 1983).
Phenological timing varied with weather patterns in a 14-year study (1995-2008) of grassland vegetation in Missoula, Montana (Lesica and Kittelson 2010). Flowering of cutleaf daisy was earlier with decreased winter precipitation and increased spring temperature. The average date of earliest flowering for cutleaf daisy was April 27. Average March temperature and December and January precipitation were explanatory variables in the best-supported multiple linear regression model for average earliest flowering date. Over the course of the study, December plus January precipitation declined an average of 0.2 in (4.8 mm)/year, and March temperature increased 0.36 ˚F (0.2 ˚C)/year (Lesica and Kittelson 2010).
Breeding System
Cutleaf daisy is a complex of sexual and apomictic (agamospermous) forms. Sexual forms produce abundant and highly viable pollen and are strongly outcrossing. Apomictic forms either produce no pollen or extremely low-quality pollen (Noyes et al. 1995). Apomictic forms grown in a greenhouse without pollinators produced large numbers of filled seed and although viability was not rigorously tested, seed that fell to the soil germinated readily (Noyes and Soltis 1996).
Measures of genetic variation suggest that sexual forms of cutleaf daisy are strongly outcrossing (Noyes et al. 1995). Sexual populations include a large number of genotypes with relatively low heterozygosity. Genetic variation in apomictic populations is held within a limited number of highly heterozygous genotypes (Noyes and Soltis 1996).
Pollination
Pollination is required for good seed production in sexual forms. When grown in a greenhouse, sexual forms produced pollen but very few achenes (1-2/capitulum occasionally observed); in the field, full seed set was often observed. A lack of pollinators in the greenhouse was considered the reason for the low seed set there.
In a floral ecology study, Ostler and Harper (1978) reported that cutleaf daisy is animal pollinated and that access to nectaries and/or pollen is structurally restricted (Ostler and Harper 1978). Cutleaf daisy was preferred by bumble bees (Bombus spp.) in subalpine meadows in northeastern Utah’s Uinta Mountains (Bowers 1985).
Ecology
Cutleaf daisy is a colonizer of early seral habitats and disturbances from grassland to alpine zones (Luna et al. 2008), but also grows in late seral vegetation (see Habitat and Plant Associations section), although it is likely restricted to canopy openings when the overstory is dense. Established plants are also considered disturbance tolerant (USDA FS 1937; Riegel et al. 2006).
Cutleaf daisy can be an early colonizer in early seral habitats (Ellison 1954; McNeal 1976; Day and Wright 1985). It was reported in primary successional communities on talus slopes and rock fields at the base of limestone cliffs in the subalpine zone of the Wasatch Plateau, central Utah (Ellison 1954). It also occurred on mudflows in the alpine zone of Colorado’s Rocky Mountain National Park. Mudflows were described as actively moving, completely unstratified soil mixed with rock on steep slopes (McNeal 1976). In parts of Canada, plants establish readily and become abundant on limestone gravel (Marie-Victorin 1938).
Disturbance Ecology
Cutleaf daisy is disturbance tolerant. A survey of nearly 200 sagebrush sites in eastern Montana found that it occurred on slightly to heavily disturbed sites (Harvey 1990).
Fire.
Erigeron species common in sagebrush communities are not impacted negatively by fire (Riegel et al. 2006). Cutleaf daisy recovered soon after fire in a ponderosa pine-Douglas-fir (Pseudotsuga menziesii) forest in the Pike National Forest, Colorado. Presence-absence comparisons made between pre- and post-fire plots indicated a decrease in cutleaf daisy in the first postfire year but no differences 2 to 5 years after fire. The fire started on June 9 and was patchy in terms of severity (Fornwalt and Kaufmann 2014).
When 8-year-old burned and unburned sites were compared on Steep Mountain near Butte, Montana, cutleaf daisy did not occur on the burned site, while its frequency was about 30% on unburned sites. The prescribed fire occurred in early November in antelope bitterbrush (Pursia tridentata)-mountain big sagebrush (Artemisia tridentata subsp. vaseyana)/bluebunch wheatgrass vegetation (Fraas et al. 1992).
In a study to determine if historic fire regimes could be characterized from the composition of understory vegetation, cutleaf daisy was a significant indicator (P < 0.05) of high fire severity associated with a mean fire return interval of more than 40 years. The study evaluated 43 field sites in ponderosa pine-dominated forests in the montane zone of the Front Range in northern Colorado. Cutleaf daisy was one of about 30 species associated with specific fire regimes (Keith 2007; Keith et al. 2010).
Grazing.
Erigeron species and cutleaf daisy specifically often increase with grazing (USDA FS 1937; Mueggler and Stewart 1980). In southern British Columbia, grazing effects in rough fescue-parsnipflower buckwheat (Festuca campestris–Eriogonum heracleoides) vegetation were evaluated in exclosures that protected areas from livestock (Table 1). Cutleaf daisy abundance was lower on protected than grazed sites, but differences were not significant (McLean and Tisdale 1972).
Table 1. Frequency and cover of cutleaf daisy inside and outside of two livestock exclosures over an 8-year period in a rough fescue grassland in southern British Columbia. Exclosures were constructed in 1931 (McLean and Tisdale 1972).
| Year | Vegetation measurement | Exclosure 1 | Exclosure 2 | ||
| Inside | Outside | Inside | Outside | ||
| 1960 | Cover (%) | 1 | 7 | 0 | 6 |
| Frequency (%) | 8 | 52 | 0 | 74 | |
| 1968 | Cover (%) | T* | 9 | T | 4 |
| Frequency (%) | 2 | 74 | 6 | 20 | |
*T=trace.
In subalpine vegetation on Steens Mountain in eastern Oregon, the abundance of cutleaf daisy increased with erosion and loss of topsoil associated with heavy sheep grazing and decreases in the abundance of sheep fescue (Festuca ovina) (Maser and Strickler 1978).
Wildlife And Livestock Use
Studies documenting the use of cutleaf daisy by wildlife and livestock were limited in the available literature. Kufeld et al. (1973) reported it as a trace element of mule deer (Odocoileus hemionus) diets. In Montana it was eaten by mountain goats (Oreamnos americanus) in summer with an observed 35% frequency of use in an area where cutleaf daisy was rare (Saunders 1955; Chadwick 1974).
Generally, Erigeron species are considered more palatable to sheep and goats than to cattle and are not eaten by horses (USDA FS 1937). Cutleaf daisy is sometimes rated fair sheep forage but importance in livestock diets is negligible because of its generally sparse distribution (Hermann 1966). Cutleaf daisy is associated with and used as an indicator of livestock overgrazing (Craighead et al. 1963).
Due to its small stature, cutleaf daisy may be more important to insects and birds than to livestock and large ungulates. Erigeron species are host plants for northern checkerspot (Chlosyne palla) and field crescent (Phyciodes pulchella) butterflies (James and Nunnallee 2011).
In Sierra Nevada Mountain’s Virginia Canyon, California, cutleaf daisy seeds were eaten by gray-crowned rosy finches (Leucosticte tephrocotis) and thought to be an important winter food source for the species (Twining 1940). In the Great Basin, Erigeron species are important components of greater sage-grouse (Centrocercus urophasianus) habitats (Lambert 2005). They were noted as a food source for juvenile and adult greater sage-grouse in studies conducted in June and July in Montana and Oregon (Pennington et al. 2016).
Ethnobotany
Fleabane, the common name for the Erigeron genus, suggests its use as a flea repellent. Early reporting by the USDA FS (1937) notes that when burned, some fleabane species repel insects.
Thompson Indians used cutleaf daisy as an internal and external medicine. External use included chewing the plant, mixing it with saliva, and applying it to sores or pains. The plant was also toasted until brittle and dry, and then crushed and mixed with grease to make a salve for pain and swellings. It was often applied to swollen glands and sore throats. Methods for internal use are unknown (Turner et al. 1990).
Horticulture
Cutleaf daisy seed is easily grown from seed, which is available commercially (Dorn and Dorn 2007; LBJWC 2022]. The short-stature clumps are ideal for rock gardens or exposed slopes (Parkinson 2003; UW Ext 2017). Plants tolerate a variety of soils if drainage is good (Dorn and Dorn 2007), and there is a minimum rooting depth of 10 in (25 cm) (Mee et al. 2003). Cutleaf daisy plants are extremely cold tolerant, withstand hot sun and windy conditions, and are hardy to USDA Plant Hardiness Zones 3 or 4 (Parkinson 2003; UW Ext 2017). Seed should be surface sown (Dorn and Dorn 2007), and rooting occurs within 28 days of placing cuttings in moist sand or vermiculite.
Good plant vigor, hardiness, and attractiveness to pollinators and predatory or parasitoid insects that prey on pest insects make cutleaf daisy a good choice for use for anywhere native plants are desired (Currah et al. 1983; LBJWC 2022).
Revegetation Use
Cutleaf daisy’s importance to pollinators (Eldredge et al. 2013; Pollinator Partnership 1996-2023), broad distribution, tolerance of a range of dry soil types, and rapid colonization of disturbed sites suggests it could be useful in revegetation on adapted sites throughout the West (see sections on Distribution, Habitat and Plant Associations, Soils, and Ecology).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among all partners to select appropriate species and seed sources; these are determined by site conditions and current or projected revegetation requirements and goals (PCA 2015) and consultation with land managers and stakeholders.
Developing a seed supply begins with seed collection from native stands. Collected seed is distributed for restoration or production of nursery stock. Increase in agricultural seed fields may be required to provide a supply of seed (PCA 2015). Production of nursery stock requires less seed than large-scale seeding operations. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for cutleaf daisy, generalized provisional seed zones developed by Bower et al. (2014) may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat to moisture index). In Figure 8, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seed zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USDA FS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Restoration Tool (Richardson et al. 2020) can also guide revegetation planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
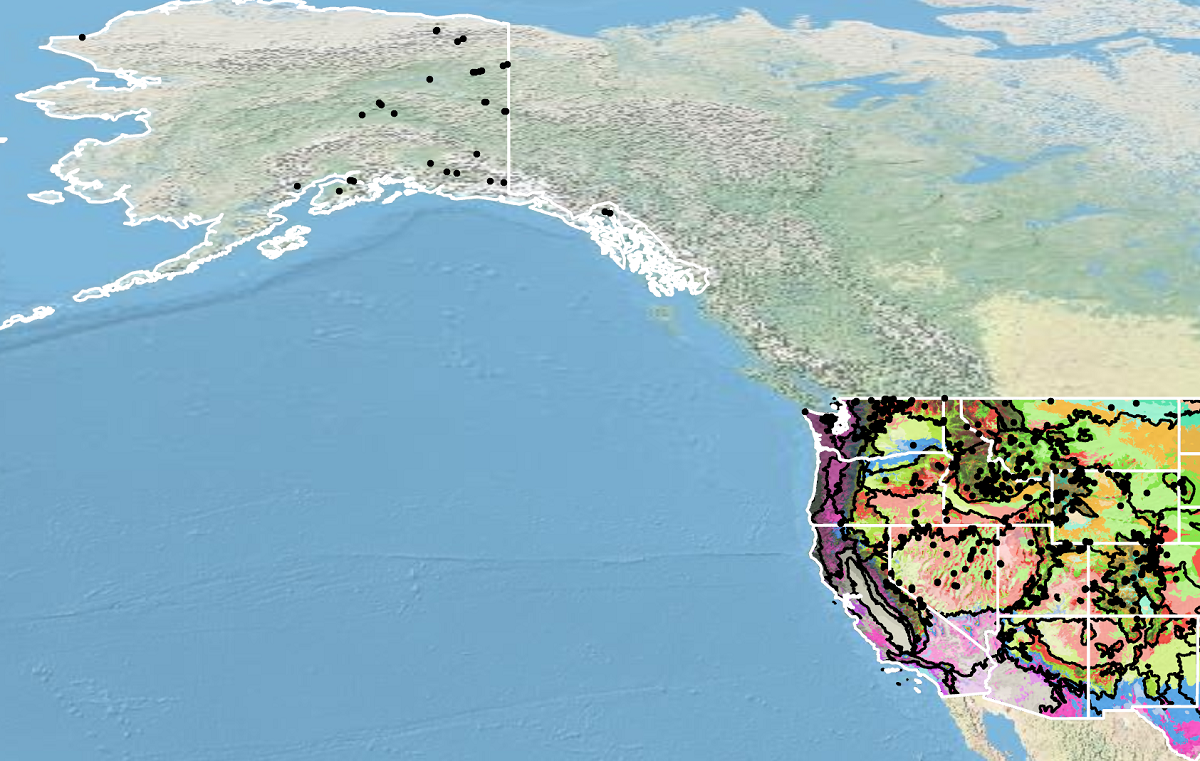
Distribution of cutleaf daisy based on geo-referenced herbarium specimens and observational data from 1861-2018 (CPNWH 2017; SEINet 2020; USDI USGS 2020). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USDA FS WWETAC 2017; https://www.fs.usda.gov/wwetac/Seedzonemapping.php). Map prepared by M. Fisk, USDI USGS.
Releases
As of 2023, there were no cutleaf daisy germplasm releases.
Wildland Seed Collection
Seed is generally hand-collected when achenes are tan and detach easily (Luna et al. 2008). Plants are very low statured, which suggests that seed collection may be labor intensive and time consuming. Ripening may be fairly uniform within seedheads but vary among seedheads of a single plant and within and among plants in a population (see Fig. 9).

Figure 9. Cutleaf daisy plant with multiple seedheads, several with seeds dispersed and one with seeds still maturing. Photo taken on July 30, 1981, in Mono County, California. Photo: ©2019 John Doyen, CalPhotos.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (UCIA 2015; Young et al. 2020). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding the collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
The BLM Seeds of Success (SOS) crews made six collections of cutleaf daisy seed over 5 years (USDI BLM SOS 2017). The earliest harvest date was June 4, 2012, in Albany County, Wyoming, at an elevation of 9,000 ft (2,700 m). The latest harvest date was August 15, 2005, in Salt Lake County, Utah, at an elevation of 10,760 ft (3,280 m) (USDI BLM SOS 2017). Seed (Fig. 10) was harvested in August from an elevation of 6,600 ft (2,000 m) in Glacier National Park, Montana (Luna et al. 2008).

Figure 10. Cutleaf daisy seed with pappi attached. Scale is 0.16 in2 (1 cm2). Photo: USDI BLM OR931 SOS.
Collection Methods
National Park Service staff at Glacier National Park collected seed by hand when seeds were tan and easily detached from the head. Seed was collected in paper bags (Luna et al. 2008).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of cutleaf daisy.
Post-Collection Management
Staff at Glacier National Park stored seed in paper bags in a ventilated drying shed prior to cleaning (Luna et al. 2008). If insects are suspected in the collections, insect strips can be used or seed can be stored at low temperatures to prevent substantial loss to insect damage. Seed should be stored in an area free of rodents.
Seed Cleaning
Seed collected in Glacier National Park was cleaned using a hammermill and office clipper (Luna et al. 2008).
Seed Storage
Cutleaf daisy seed is orthodox. There was no viability loss when dry seed was stored 33 days at -4 °F (-20 °C) (SER, INSR, RBGK 2023).
Seed Testing
Seed viability is evaluated using the general Tetrazolium testing procedures described for other Asteraceae genera as no specific AOSA procedures exist for Erigeron species (AOSA 2010). There is no AOSA rule for testing germination or purity of cutleaf daisy (AOSA 2016).
Germination Biology
Studies indicate that cutleaf daisy germinates readily with warm temperatures (70 °F [21 °C]) and light exposure (Young and Young 1986; Love and Akins 2019). An afterripening period (>1 mo) improved germination of seed collected from alpine sites (Kaye 1997), but it may not be necessary for all seed lots (Love and Akins 2019).
The Seed Information Database (SID) reported 100% germination after 21 days for cutleaf daisy seed on 1% agar exposed to an 8/16 hour light-dark cycle at constant temperatures of 59 °F (15 °C) or 77 °F (25 °C) (SER, INSR, RBGK 2023). Neither the age or origin of the seed tested was reported.
Germination of cutleaf daisy was much improved with a 9-month compared to a 1-month afterripening period in a study of seed dormancy (Table 2, Kaye 1997). Cutleaf daisy seed tested was collected from a single population on a south slope (5,600-5,900 ft [1,700-1,800 m]) in Olympic National Park, Washington. At harvest time, 30 to 70% of the seed was filled and only filled seed was used in the testing. Clean dry seed was stored 1 month and 9 months at 45 °F (7 °C). Seed was incubated at a constant 68 °F (20 °C) and an 8/16 hour light-dark cycle (Kaye 1997).
Table 2. Germination of cutleaf daisy seed with 1- and 9-month afterripening periods. Seed was collected from a single population on a south slope (5,600-5,900 ft [1,700-1,800 m]) in Olympic National Park, Washington (Kaye 1997).
| Afterripening period | Percent seed germination | Days to 50% germination | N |
| 1 month | 58.3 | 11 | 60 |
| 9 months in light | 89.0 | 13 | 75 |
| 9 months in dark | 90.7 | —– | 75 |
In a summary of Norman Deno’s research, Love and Akins (2019) reported that 100% of fresh cutleaf daisy seed germinated within 3 months at 70 °F (21 °C). Just 32% of fresh seed germinated within 3 months at 40 °F (4 °C), but at temperatures of 70 °F (21 °C), germination increased to 68%. Seed that was dry-stored for 6 months at either 40 °F (4 °C) or 70 °F (21 °C), germinated to 100% at an incubation temperature of 70 °F (21 °C) within 3 months. Just 18% of dry-stored seed germinated after 3 months at 40 °F (4 °C), but at an incubation temperature of 70 °F (21 °C), 82% germinated within 3 months (Love and Akins 2019).
Wildland Seed Yield And Quality
Post-cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 3 (USDA FS BSE 2017). The results indicate that when fresh cutleaf daisy seed is cleaned to a high level of purity, the fill and viability are likely to be high.
Table 3. Seed yield and quality of cutleaf daisy seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USDA Forest Service National Seed Laboratory (USDA FS BSE 2017).
| Seed lot characteristic | Mean | Range | Samples (no.) |
| Bulk weight (lbs) | 0.15 | 0.09-0.23 | 3 |
| Clean weight (lbs) | 0.02 | 0.01-0.03 | 3 |
| Clean-out ratio | 0.14 | 0.04-0.29 | 3 |
| Purity (%) | 97 | 96-98 | 3 |
| Fill (%)¹ | 98 | 96-99 | 3 |
| Viability (%)² | 95 | 90-98 | 3 |
| Seeds/lb | 1,919,082 | 1,778,784-2,040,000 | 3 |
| Pure live seeds/lb | 1,771,723 | 1,690,912-1,861,698 | 3 |
¹ 100 seed X-ray test
²Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well.
Agricultural Seed Production
Agricultural production of cutleaf daisy seed was not reported in the reviewed literature. Reports of abundant seed production and high levels of seed fill and viability suggest seed farming could be viable. Potential difficulties include the low stature and small size of plants and uneven seed ripening (see Fig. 9), which will impact methods for growing and harvesting. Although native forb seed is often seeded in the fall to allow for stratification of dormant seed, the Germination Biology section suggests and McManamen et al. (2018) reports that cutleaf daisy can be seeded in the spring.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (UCIA 2015; Young et al. 2020).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Nursery Practice
Researchers indicate that cutleaf daisy seedlings are easily grown from seed (Dorn and Dorn 2007). Seed does not need scarification and germinates well in a greenhouse (McManamen et al. 2018). Good drainage is important to seedling growth, and seedlings are easily transplanted (Dorn and Dorn 2007).
The following procedure was used at Glacier National Park (GNP) to grow cutleaf daisy in an outdoor nursery (Luna et al. 2008). Seed collected from the park was planted in the fall into soil filled containers. The soil was a mixture of peat moss, perlite, and vermiculite (70%) and sand (30%). Soil also included a controlled release fertilizer (13N:13P205:13K2O, 8-9 mo release at 70 °F [21 °C]). Containers were irrigated thoroughly and kept slightly moist. Germination was uniform and emergence occurred after several days with temperatures of 72 °F (22 °C) or more. Seedlings were thinned at the first true leaf stage. Once seedlings were established (about 4 wks), soil was allowed to dry between irrigations. Roots and shoots grew rapidly with four to six true leaves emerging 3 weeks following germination. During the growing season, plants were fertilized with a 20N:20P:20K liquid fertilizer (100 ppm). The active growth phase was 8 weeks. Plants were leached with water and fertilized (10N: 20P:20K at 200 ppm) once more before winterizing, which included a 4-week hardening period. Plants were overwintered under insulating foam cover and snow in an outdoor nursery. Harvesting occurred after 9 months (Luna et al. 2008).
Wildland Seeding And Planting
Cutleaf daisy has been established from seed in mine reclamation (Smyth 1997). In a controlled study, cutleaf daisy failed to germinate in soils treated with herbicide (picloram or aminopyralid, at recommended rates) (McManamen et al. 2018), which is important to consider in wildland revegetation site preparation for this species. The recommended pure stand seeding rate for Erigeron species ranges from 1 to 4 lbs/ac (1.1-4.5 kg/ha). This would be adjusted for the desired amount of cutleaf daisy in the seed mix (Ogle et al. 2012).
Cutleaf daisy established on amended and unamended mine spoils where it was seeded and where it was not purposely seeded (Smyth 1997). The site was a 3-year-old coal mine spoil at an alpine tundra and Engelmann spruce-subalpine fir (Picea engelmannii–Abies lasiocarpa) transition zone in southeastern British Columbia. Vegetation establishment was evaluated on seeded and unseeded plots with unamended spoils or spoils amended with native soil. Unamended spoils were primarily siltstone and carbonaceous shale. Amended spoils were covered with 2 in (5 cm) of native soil with vegetation and organic material removed and fertilizer added (171 lbs/ac [192 kg/ha] N, 17 lbs/ac [19 kg/ha] P and K). Seeded species were collected locally and stored at 36 to 41 °F (2-5 °C) until used. In germination tests for seeded species (68 °F [20 °C], 12/12 hour light-dark cycle), germination of cutleaf daisy seed was 73%. Spoil plots were seeded in May 1985 and vegetation establishment evaluated from 1986 to 1990. Cutleaf daisy occurred with low cover (<1%) on all plots. Establishment on unseeded plots was attributed to seed dispersal of 3 to 7 ft (1-2 m) from adjacent treatments (Smyth 1997).
In a year-long greenhouse study, cutleaf daisy germinated in untreated soils but not in herbicide-treated soils (picloram or aminopyralid, at recommended rates). Pots (4 in [10 cm] diameter by 3.8 in [9.75 cm] deep) were filled with soil (one part sand and two parts field-collected loamy topsoil). Filled pots were heat treated (360 °F [180 °C] for 1 hr) to kill any seeds. Half of the pots were sprayed with herbicide (picloram and aminopyralid, individually) at recommended rates. Cutleaf seed purchased from a vendor was then seeded into the pots. Pots were kept in a greenhouse that was unheated in the winter and in the summer was cooled with fans and had walls folded up for air circulation when temperatures exceeded 86 °F (30 °C). Emergence of cutleaf daisy per each untreated pot was 1.2 at 0 month, 1.0 at 3 months, 0.5 at 6 months, 0.3 at 9 months, and 0 at 11 months. No seedlings emerged in any herbicide-treated pots even at 11 months after treatment (McManamen et al. 2018).
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Paula Fornwalt, USDA, Forest Service, and Stephen Love, University of Idaho.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Aho, K.; Weaver, T. 2010. Alpine nodal ecology and ecosystem evolution in the north-central Rockies (Mount Washburn; Yellowstone National Park, Wyoming). Arctic, Antarctic, and Alpine Research. 42(2): 139-151.
Andersen, B.A.; Holmgren, A.H. 1996. Mountain plants of northeastern Utah. HG 506. Logan, UT: Utah State University. 124 p.
Arno, S.F.; Habeck, J.R. 1972. Ecology of alpine larch (Larix lyallii Parl.) in the Pacific Northwest. Ecological Monographs. 42(4): 417-450.
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Association of Official Seed Analysts [AOSA]. 2016. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Beaman, J.H. 1977. Apomixis and the systematics of Erigeron compositus. Botanical Society of America, Miscellaneous Series. 154: 57.
Belsky, J.; del Moral, R. 1982. Ecology of an alpine-subalpine meadow complex in the Olympic Mountains, Washington. Canadian Journal of Botany. 60(6): 779-788.
Blackwell, L.R. 2006. Great Basin wildflowers: A guide to common wildflowers of the high deserts of Nevada, Utah, and Oregon. Helena, MT: Morris Book Publishing. 288 p.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Bowers, M.A. 1985. Bumble bee colonization, extinction, and reproduction in subalpine meadows in northeastern Utah. Ecology. 66(3): 914-927.
Chadwick, D.H. 1974. Mountain goat ecology: Logging relationships in the Bunker Creek drainage of western Montana. Missoula, MT: University of Montana. Thesis. 262 p.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2020. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Craighead, J.J.; Craighead, F.C. Jr.; Davis, R.J. 1963. A field guide to Rocky Mountain wildflowers from northern Arizona and New Mexico to British Columbia. Boston, MA: Houghton Mifflin Company. 277 p.
Cronquist, A. 1994. Volume five: Asterales. In: Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K., eds. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Bronx, NY: The New York Botanic Garden. 496 p.
Currah, R.; Smreciu, A.; Van Dyk, M. 1983. Prairie wildflowers: An illustrated manual of species suitable for cultivation and grassland restoration. Edmonton, AB: University of Alberta, Friends of the Devonian Botanic Garden. 290 p.
Daubenmire, R.F. 1941. Some ecologic features of the subterranean organs of alpine plants. Ecology. 22(4): 370-378.
Day, T.; Wright, R.G. 1985. The vegetation types of Craters of the Moon National Monument. Bull. 38. Moscow, ID: University of Idaho, College of Forestry, Wildlife, and Range Sciences, Forest, Wildlife and Range Experiment Station. 6 p.
del Moral, R. 1983. Competition as a control mechanism in subalpine meadows. American Journal of Botany. 70(2): 232-245.
Dorn, R.D.; Dorn, J.L. 2007. Growing native plants of the Rocky Mountain area. Morrisville, NC: Lulu.com. 260 p.
Douglas, G.W.; Bliss, L.C. 1977. Alpine and high subalpine plant communities of the North Cascades Range, Washington and British Columbia. Ecological Monographs. 47(2): 113-150.
Eldredge, E.; Novak-Echenique, P.; Heater, T.; Mulder, A.; Jasmine, J. 2013. Plants for pollinator habitat in Nevada. Tech. Note NV 57. Reno, NV: U.S. Department of Agriculture, Natural Resources Conservation Service. 65 p.
Ellison, L. 1954. Subalpine vegetation of the Wasatch Plateau, Utah. Ecological Monographs. 24(2): 89-184.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Fornwalt, P.J.; Kaufmann, M.R. 2014. Understorey plant community dynamics following a large, mixed severity wildfire in a Pinus ponderosa–Pseudotsuga menziesii forest, Colorado, USA. Journal of Vegetation Science. 25(3): 805-818.
Fraas, W.W.; Wambolt, C.L.; Frisina, M.R. 1992. Prescribed fire effects on a bitterbrush-mountain big sagebrush-bluebunch wheatgrass community. In: Clary, W.P.; McArthur, E.D.; Bedunah, D.; Wambolt, C.L., comps. Ecology and management of riparian shrub communities symposium proceedings; 1991 May 29-31; Sun Valley, ID. Gen. Tech. Rep. INT-289. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Research Station: 212-216.
Harvey, S.J. 1990. Responses of steppe plants to gradients of water, soil texture and disturbance in Montana, USA. Bozeman, MT: Montana State University. Thesis. 103 p.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
ITIS Database. 2023. Integrated Taxonomic Information System. Available: http://www.itis.gov/index.html
James, D.G.; Nunnallee, D. 2011. Life histories of Cascadia butterflies. Corvallis, OR: Oregon State University Press. 447 p.
Kaye, T.N. 1997. Seed dormancy in high elevation plants: Implications for ecology and restoration. In: Kaye, T.N.; Liston, A.; Luoma, D.L.; Meinke, R.J.; Wilson, M.V., eds. Conservation and management of native plants and fungi conference proceedings; 1997 November 15-17; Corvallis, OR. Corvallis, OR: Native Plant Society of Oregon: 115-120.
Keith, R.P. 2007. Characterizing fire regimes from understory vegetation composition in ponderosa pine dominated forests of the northern Coronado Front Range. Boulder, CO: University of Colorado. Thesis. 68 p.
Keith, R.P.; Veblen, T.T.; Schoennagel, T.L.; Sherriff, R.L. 2010. Understory vegetation indicates historic fire regimes in ponderosa pine-dominated ecosystems in the Colorado Front Range. Journal of Vegetation Science. 21(3): 488-499.
Kimball, S.; Wilson, P.; Crowther, J. 2004. Local ecology and geographic ranges of plants in the Bishop Creek Watershed of the eastern Sierra Nevada, California, USA. Journal of Biogeography. 31(10): 1637-1657.
Kruckeberg, A.R. 1967. Ecotypic response to ultramafic soils by some plant species of northwestern United States. Brittonia. 19(2): 133-151.
Kufeld, R.C.; Wallmo, O.C.; Feddema, C. 1973. Foods of the Rocky Mountain mule deer. Res. Pap. RM-111. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Forest and Range Experiment Station. 31 p.
Lady Bird Johnson Wildflower Center [LBJWC]. 2022. Erigeron compositus Pursh. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants/result.php?id_plant=ERCO4 [Accessed 2023 August 7].
Lambert, S. 2005. Guidebook to the seeds of native and non-native grasses, forbs and shrubs of the Great Basin. Boise, ID: U.S. Department of the Interior, Bureau of Land Management, Idaho State Office. 136 p.
Lavin, M.; Wojciechowski, M.F. 2002. Vascular flora of the Sweetwater Mountains of California and Nevada. 24 p. http://bristleconecnps.org/native_plants/checklists/SweetwatersChecklistJun2002.pdf [Accessed 2023 October 10].
Lesica, P. 2012. Manual of Montana vascular plants. Fort Worth, TX: Botanical Research Institute of Texas (BRIT) Press. 779 p.
Lesica, P.; Kittelson, P.M. 2010. Precipitation and temperature are associated with advanced flowering phenology in a semi-arid grassland. Journal of Arid Environments. 74(9): 1013-1017.
Lloyd, A.H.; Armbruster, W.S.; Edwards, M.E. 1994. Ecology of a steppe-tundra gradient in interior Alaska. Journal of Vegetation Science. 5(6): 897-912.
Looman, J.; Campbell, J.B. 1960. Adaptation of Sorensen’s K (1948) for estimating unit affinities in prairie vegetation. Ecology. 41(3): 409-416.
Love, A.; Love, D.; Kapoor, B.M. 1971. Cytotaxonomy of a century of Rocky Mountain orophytes. Arctic and Alpine Research. 3(2): 139-165.
Love, S.L.; Akins, C.J. 2019. Second summary of the native seed germination studies of Norman C Deno: Species with names beginning with letters C through E. Native Plants Journal. 20(1): 65-97.
Love, S.L.; McCammon, T.A. 2017. Compatible host/parasite pairs enhance propagation of paintbrush (Castilleja spp.). Native Plants Journal. 18(3): 252-266.
Luna, T; Evans, J.; Wick, D. 2008. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Erigeron compositus Pursh plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2023 August 7].
Marie-Victorin, F. 1938. Phytogeographical problems of eastern Canada. The American Midland Naturalist. 19(3): 489-558.
Maser, C.; Strickler, G.S. 1978. The sage vole, Lagurus curtatus, as an inhabitant of subalpine sheep fescue, Festuca ovina, communities on Steens Mountain- An observation and interpretation. Northwest Science. 52(3): 276-284.
McLean, A.; Tisdale, E.W. 1972. Recovery rate of depleted range sites under protection from grazing. Journal of Range Management. 25(3): 178-184.
McManamen, C.; Nelson, C.R.; Wagner, V. 2018. Timing of seeding after herbicide application influences rate of germination and seedling biomass of native plants used for grassland restoration. Restoration Ecology. 26(6): 1137-1148.
McNeal, D.W. 1976. Annotated check list of the alpine vascular plants of Specimen Mountain, Rocky Mountain National Park, Colorado. The Southwestern Naturalist. 20(4): 423-435.
Mee, W.; Barnes, J.; Kjelgren, R.; Sutton, R.; Cerny, T.; Johnson, C. 2003. Water wise: Native plants for Intermountain landscapes. Logan, UT: Utah State University Press. 220 p.
Mueggler, W.F.; Stewart, W.L. 1980. Grassland and shrubland habitat types of western Montana. Gen. Tech. Rep. INT-66. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station. 154 p.
Mueggler, W.F.; Stewart, W.L. 1981. Forage production on important rangeland habitat types in western Montana. Journal of Range Management. 34(5): 347-353.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Nesom, G.L. 2006. 186. Erigeron. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 20 Magnoliophyta: Asteridae, part 7: Asteraceae, part 2 Asterales, part 2 (Aster order). New York, NY: Oxford University Press: 256-348.
Noyes, R.D.; Soltis, D.E. 1996. Genotypic variation in agamospermous Erigeron compositus (Asteraceae). American Journal of Botany. 83(10): 1292-1303.
Noyes, R.D.; Soltis, D.E.; Soltis, P.S. 1995. Genetic and cytological investigations in sexual Erigeron compositus (Asteraceae). Systematic Botany. 20(2): 132-146.
Ogle, D.; St. John, L.; Stannard, M.; Holzworth, L. 2012. Conservation plant species for the Intermountain West. Plant Materials Technical Note 24. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 57 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Orloci, L.; Stanek, W. 1980. Vegetation survey of the Alaska Highway, Yukon Territory: Types and gradients. Vegetatio. 41(1): 1-56.
Ostler, W.K.; Harper, K.T. 1978. Floral ecology in relation to plant species diversity in the Wasatch Mountains of Utah and Idaho. Ecology. 59(4): 848-861.
Parkinson, H. 2003. Landscaping with native plants of the Intermountain Region. Tech. Ref. 1730-3. Boise, ID: U.S. Department of the Interior, Bureau of Land Management. 46 p.
Pennington, V.E.; Schlaepfer, D.R.; Beck, J.L.; Bradford, J.B.; Palmquist, K.A.; Lauenroth, W.K. 2016. Sagebrush, greater sage-grouse, and the occurrence and importance of forbs. Western North American Naturalist. 76(3): 298-312.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Pollinator Partnership. 1996-2023. Pollinator Partnership: Protect their lives. Preserve ours. Available from https://www.pollinator.org/
Ramaley, R. 1919. Xerophytic grasslands at different altitudes in Colorado. Bulletin of the Torrey Botanical Club. 46(2): 37-52.
Reveal, J.L.; Moulton, G.E.; Schuyler, A.E. 1999. The Lewis and Clark collections of vascular plants: Names, types, and comments. Proceedings of the Academy of Natural Sciences of Philadelphia. 149: 1-64.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Riegel, G.M.; Miller, R.F.; Skinner, C.N.; Smith, S.E. 2006. Northeastern Plateaus bioregion. In: Sugihara, N.G.; van Wagtendonk, J.W.; Shaffer, K.E.; Fites-Kaufman, J.; Thode, A., eds. Fire in California’s ecosystems. Berkeley, CA: University of California Press: 225-263.
Rydberg, P.A. 1914. Phytogeographical notes on the Rocky Mountain region. III. Formations in the alpine zone. Bulletin of the Torrey Botanical Club. 41(9): 459-474.
Saunders, J.K. 1955. Food habits and range use of the Rocky Mountain goat in the Crazy Mountains, Montana. The Journal of Wildlife Management. 19(4): 429-437.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2020. SEINet Regional Networks of North American Herbaria. https://Symbiota.org/docs/seinet
Shiflet, T.N. 1994. Rangeland cover types of the United States. Denver, CO: Society for Range Management. 152 p.
Smyth, C.R. 1997. Early succession patterns with a native species seed mix on amended and unamended coal mine spoil in the Rocky Mountains of southeastern British Columbia, Canada. Arctic and Alpine Research. 29(2): 184-195.
Society for Ecological Restoration; International Network for Seed Based Restoration; Royal Botanic Gardens [SER, INSR, RBGK]. 2023. Seed Information Database (SID). Available from: https://ser-sid.org/
Spence, J.R.; Shaw, R.J. 1983. Observations on alpine vegetation near Schoolroom Glacier, Teton Range, Wyoming. The Great Basin Naturalist. 43(3): 483-491.
Sperry, O.E. 1936. A study of the growth, transpiration, and distribution of the conifers of the Rocky Mountain National Park. Bulletin of the Torrey Botanical Club. 63(2): 75-103.
St. John, H.; Warren, F.A. 1937. The plants of Mount Rainier National Park, Washington. The American Midland Naturalist. 18(6): 952-985.
Taye, A.C. 1995. Alpine vascular flora of the Tushar Mountains, Utah. The Great Basin Naturalist. 55(3): 225-236.
Taylor, R.J. 1992. Sagebrush country: A wildflower sanctuary. Missoula, MT: Mountain Press Publishing Company. 211 p.
Taylor, R.L.; Brockman, R.P. 1966. Chromosome numbers of some western Canadian plants. Canadian Journal of Botany. 44(8): 1093-1109.
Tisdale, E.W. 1947. The grasslands of the southern Interior of British Columbia. Ecology. 28(4): 346-382.
Turner, N.J.; Thompson, L.C.; Thompson, M.T.; York, A.Z. 1990. Thompson ethnobotany: Knowledge and usage of plants by the Thompson Indians of British Columbia. Royal British Columbia Museum Memoir No. 3. Victoria, British Columbia, Canada: Royal British Columbia Museum. 335 p.
Twining, H. 1940. Foraging behavior and survival in the Sierra Nevada rosy finch. The Condor. 42(1): 64-72.
University of Wyoming Extension [UW Ext.] 2018. Native plants for the Intermountain West. Laramie, WY: University of Wyoming. http://www.wyoextension.org/westernnativeplants
USDA Forest Service [USDA FS] 1937. Range plant handbook. Washington, DC: U.S. Department of Agriculture, Forest Service. 816 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2023. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2021. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 44 p.
USDI Environmental Protection Agency [USDI EPA]. 2018. Ecoregions. Washington, DC: U.S. Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2020. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Vestal, A.G. 1917. Foothills vegetation in the Colorado Front Range. Botanical Gazette. 64(5): 353-385.
Vetter, M.A. 2000. Grasslands of the Aishihik-Sekulmun Lakes Area, Yukon Territory, Canada. Arctic. 53(2): 165-173.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Wesser, S.D.; Armbruster, W.S. 1991. Species distribution controls across a forest-steppe transition: A causal model and experimental test. Ecological Monographs. 61(3): 323-342.
Wiese, J.L.; Meadow, J.F.; Lapp, J.A. 2012. Seed weights for northern Rocky Mountain native plants with an emphasis on Glacier National Park. Native Plants Journal. 13(1): 39-49.
Wright, R.D. 1963. Some ecological studies on bristlecone pines in the White Mountains of California. Los Angeles, CA: University of California. Dissertation. 118 p.
Young, J.A.; Young, C.G. 1986. Collecting, processing and germinating seeds of wildland plants. Portland, OR: Timber Press. 236 p.
Young, S.A.; Schrumpf, B.; Bouck, M.; Moore, M. 2020. How the Association of Official Seed Certifying Agencies (AOSCA) tracks wildland sourced seed and other plant propagating materials. Logan, UT: Utah State University, Utah Agricultural Experiment Station. 36 p.
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2024. Cutleaf daisy (Erigeron compositus). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org/species/cutleaf-daisy-erigeron-compositus/