Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
February 2022
Nomenclature
Lemon scurfpea (Psoralidium lanceolatum [Pursh] Rydb.) belongs to the Fabaceae (formerly Leguminosae) family.
Family
Fabaceae – Pea family
Genus
Psoralidium
Species
lanceolatum
NRCS Plant Code
PSLA3 (USDA NRCS 2021).
Subtaxa
Currently, there are no subspecies or varieties recognized (ITIS 2021; USDA NRCS 2021).
Synonyms
Psoralea lanceolata Pursh, P. l. var. purshii (Vail) Piper, P. l. var. stenophylla (Rydb.) Toft & S.L. Welsh, P. l. var. stenostachys (Rydb.) S.L. Welsh, P. l. subsp. scabra (Nutt.) Piper, P. micrantha A. Gray, Psoralidium lanceolatum var. stenophyllum (Rydb.) S.L. Welsh, P. l. var. stenostachys (Rydb.) S.L. Welsh, P. stenophyllum (Rydb.) Rydb., P. scabra Nutt., P. stenostachys Rydb. (USDA NRCS 2021)
Common Names
Lemon scurfpea, dune scurfpea, lance-leafed scurfpea, lemon-weed, plains scurfpea, sand psoralea, slenderleaf scurfpea, and wild lemonweed (Tolstead 1942; Toft and Welsh 1972; Barneby 1989; Welsh et al. 2016; ITIS 2021).
Chromosome Number
2n = 22 (Barneby 1989; Welsh et al. 2016).
Hybridization
There were no hybrids described in the reviewed literature.
Distribution
Lemon scurfpea is widely distributed, occurring in the western half of the US and in Alberta and Saskatchewan, Canada (Hermann 1966; Toft and Welsh 1972; Hickman 1993; USDA NRCS 2021). It is most widespread in the western Great Plains and the Intermountain West (Barneby 1989; Grimm 2001; Egan and Reveal 2009; Welsh et al. 2016).
Habitat And Plant Associations
Lemon scurfpea grows in a variety of grassland, shrubland, woodland, and forest vegetation types and is common in sandy habitats throughout its range (Hermann 1966; Pavlik 1985; Hickman 1993; Abella 2008; Reif et al. 2009; Tilley and St. John 2010; Welsh et al. 2016).
Intermountain West. Lemon scurfpea occurs in many grassland and shrubland communities in the Intermountain West (Fig. 1). It grows on active windward slopes of sand dunes near the Columbia River in Oregon (Franklin and Dyrness 1973), in sagebrush (Artemisia spp.) shrublands in the Great Basin (Lambert 2005), and in sagebrush scrub in California (Munz and Keck 1973). In big sagebrush (A. tridentata) communities in southeastern Idaho, where annual precipitation averages 8.7 in (220 mm), lemon scurfpea cover averaged 0.13% (Anderson and Inouye 2001). In south-central Nevada, lemon scurfpea occurred in shadscale saltbush (Atriplex confertifolia) communities (Beatley 1976). At Little Sahara Dunes in Utah’s Juab County, lemon scurfpea cover averaged 37.1% on sand dunes with prickly Russian thistle (Salsola tragus) and Indian ricegrass (Achnatherum hymenoides) and 2.3% in big sagebrush, cheatgrass (Bromus tectorum), wheatgrasses (Agropyron spp.) vegetation adjacent to the dunes. In this area where most of the precipitation is delivered as rain and snow in winter and spring, annual precipitation averages 12.3 in (312 mm). Average annual precipitation when the vegetation was studied was much below average (4.3 in [110 mm] in 2002). Most of the precipitation is delivered as rain and snow in winter and spring (Rosenthal et al. 2005). In the Rocky Mountain Region of the US Forest Service (Region 2), lemon scurfpea occurred in big sagebrush/bluebunch wheatgrass (Pseudoroegneria spicata) vegetation on gravelly slopes with clay subsoils and in prairie sandreed/needle and thread (Calamovilfa longifolia/Hesperostipa comata) vegetation on sandy ridges with deep (≤ 75 in [191 cm]) well-drained to very well-drained fine sand or sandy loam (Johnston 1987). In Park County, Wyoming, lemon scurfpea grew with rabbitbrush, big sagebrush, Indian ricegrass, and needle and thread at a 5,000-ft (1,500 m) elevation site with gravelly sandy loam soil and average annual precipitation of 9 in (230 mm) (Tilley 2011). In south-central Colorado, lemon scurfpea occurred on 60 of 78 plots of active and stabilized dunes at Great Sand Dunes National Monument (McArthur and Sanderson 1991) and grew with rubber rabbitbrush (Ericameria nauseosa) and Indian ricegrass on sands in the Baca National Wildlife Refuge (7,500–8,200 ft [2,300–2,500 m) in elevation (Anderson 2006).

Figure 1. Big sagebrush habitat in Wyoming where lemon scurfpea was collected. Photo: USDI Bureau of Land Management (BLM) WY-020 Seeds of Success (SOS) .
Southwest. Lemon scurfpea grows in forests, woodlands, desert shrublands, and disturbed habitats in the Southwest (Abella 2008; Reif et al. 2009). Relative cover of lemon scurfpea was 8% in old-growth ponderosa pine (Pinus ponderosa) forests in northern Arizona. Total understory cover was low (12%) in this study area where the tree density averaged 80 trees/ac (190 trees/ha) (Abella 2008). In a vegetation survey of the Tusas and Jemez Mountains in north-central New Mexico, lemon scurfpea grew in twoneedle pinyon-oneseed juniper (P. edulis–Juniperus monosperma) woodlands, desert shrublands, montane riparian vegetation, and on floodplain arroyos and roadsides. Desert shrublands were dominated by some combination of basin big sagebrush (Artemisia tridentata subsp. tridentata), black sagebrush (A. nova), fourwing saltbush (Atriplex canescens), rubber rabbitbrush, and broom snakeweed (Gutierrezia sarothrae). Montane riparian vegetation occupied the margins of intermittent and perennial streams. Floodplain arroyos occurred along major rivers and were home to species adapted to dry, disturbed conditions with periodic flooding (Reif et al. 2009).
Great Plains. Lemon scurfpea is common in prairie sandhill vegetation in the Great Plains. In descriptions of rangeland cover types, it was a common forb in the prairie sandreed-needle-and-thread type found in the northern Great Plains of Saskatchewan, central and western North Dakota, eastern Montana, southeastern Wyoming, and northern Colorado. It also occurred in the wheatgrass (Pascopyrum smithii, Elymus lanceolatus, E. caninus)-little bluestem (Schizachyrium scoparium)-needlegrass (Hesperostipa spartea, H. curtiseta, H. comata, Nassella viridula) type occurring on glacial-derived soils from southeastern Saskatchewan through southwestern Manitoba, northwestern and central North Dakota through east-central South Dakota, and Nebraska where annual precipitation averages 15 to 18 in (381–457 mm). Lemon scurfpea was occasional in the sand sagebrush (A. filifolia) type in the central and southern Great Plains on deep sandy soils, where annual precipitation averaged 12 to 24 in (305–610 mm), high winds were common, and summers were hot (Shiflet 1994).
Lemon scurfpea grows with prairie sandreed in the prairie provinces of Canada (Looman 1963, 1980). It is common in prairie sandhills in Alberta with sand dropseed (Sporobolus cryptandrus), Indian ricegrass, and prairie sandreed (Moss 1955). Frequency of lemon scurfpea was 10% in blue grama (Bouteloua gracilis)-needle and thread mixed prairie with coarse-textured soils in southeastern Alberta and southern Saskatchewan (Coupland 1950).
Plant associates in the US Great Plains are similar to those of the prairie provinces of Canada. Along the upper Missouri River in west-central North Dakota, lemon scurfpea cover averaged 5.2% in mixed-grass prairie (big bluestem [Andropogon gerardii], needle and thread) (Clambey 1992). In eastern Colorado, it occurred in shortgrass prairie and in sandhill blowouts (McGinnies et al. 1991). Lemon scurfpea is considered an important forb in dry and distrubed habitats in Nebraska (Tolstead 1942; Kaul 1989 cited in Whitcomb 1989). In the Nebraska Sandhills, lemon scurfpea was a principal species in grasslands with sand bluestem (Andropogon hallii), little bluestem (Schizachyrium scoparium var. scoparium), and sandhill muhly (Muhlenbergia pungens). It was also a principal species in blowouts with prairie sandreed and blowout grass (Redfieldia flexuosa) and a secondary species in wiregrass communities with threeawn (Aristida spp.) and plains muhly (M. cuspidata) (Pool 1914).
Elevation
State-level elevation ranges occupied by lemon scurfpea were only reported for Utah (4,030–6,000 ft [1,230–1,830 m]) (Welsh et al. 2016) and California (< 8,200 ft [2,500 m]) (Hickman 1993). Most of the elevation ranges reported in other studies reviewed for this species, fell within these reported ranges, except for a lower elevation for dunes in Harding County, northwestern South Dakota, where lemon scurfpea grew at about 2,950 ft (900 m) (Bergmann et al. 2009).
Soils
Lemon scurfpea is common in sites with poor, coarse-textured soils, especially sands (Fig. 2) (Hermann 1966; Lambert 2005). In sagebrush communities, it is common on alluvial plains in sandy soils, less common in semi-stabilized soils, and rare in heavy soils (Barneby 1989; Egan and Reveal 2009).

Figure 2. Lemon scurfpea growing in sandy soil. Photo: Matt Lavin, Montana State University (MSU).
Soils in Intermountain West habitats. Soils were described for the Little Sahara Dunes in Juab County, Utah, where lemon scurfpea was common. On the actively moving sand dunes, the soil pH averaged 8.5 and had little organic matter (0.7%) and few nutrients (total nitrogen = 0.09%, total phosphorus = 0.03%) (Ludwig et al. 2006). In the Rocky Mountain Region of the USDA Forest Service, lemon scurfpea occurred in big sagebrush/grasslands on gravelly slopes with clay subsoils and in grasslands on sandy ridges with deep (≤ 75 in [191 cm]) well-drained to very well-drained fine sands or sandy loam soils (Johnston 1987). In another study of this area, the soils of sand dunes dominated by lemon scurfpea, prickly Russian thistle, and Indian ricegrass were compared to those of neighboring off-dune habitats dominated by big sagebrush, cheatgrass, and wheatgrasses (Rosenthal et al. 2005). Although, lemon scurfpea occurred in both habitats, its cover was much greater on the dunes than in big sagebrush. Dune soils had more sand, less clay, and a silt fraction that increased with increasing depth when compared to the big sagebrush soils, which had a sand fraction that decreased and clay content that increased with soil depth. Both soils had similar water content on June 6 and July 1, but sand soils had significantly less water content than big sagebrush soils on July 30 and August 30 (P < 0.01). Soil nitrogen was extremely low in both soils. Nitrogen was consistently low at all depths for dunes and decreased with depth in big sagebrush. Soil phosphorus of the two soils was similar. Phosphorus levels were lowest at the surface and highest at 30 in (75 cm) on dunes, and the reverse was true for big sagebrush soils. The carbon to nitrogen ratio was significantly greater on dunes than in big sagebrush (P < 0.05) with the differences being driven by nitrogen values. Vegetation on the dunes had consistently higher water potentials than those in big sagebrush, suggesting dune plants were deeply rooted and conservative water users (Rosenthal et al. 2005).
Soils in Southwestern habitats. In Arizona, lemon scurfpea occupied volcanic cinders and sandy loam soils. In the semi-arid lower San Francisco volcanic fields of Coconino County, lemon scurfpea grew in twoneedle pinyon-oneseed juniper vegetation on barren black cinders, sands, and drainages where the bimodal precipitation averaged 15 in (380 mm) and ranged from 10 to 26 in (250–650 mm) (Christie 2008). In northen Arizona, lemon scurfpea occurred in the understory of old-growth ponderosa pine forests on volcanic red cinder soils. The gravelly sandy loam soils were about 55% sand, 15% clay, and 35% silt, had low nitrogen content (337 lb/ac [378 kg/ha] at 0–6 in [15 cm] deep), low soil moisture (0.7%) and a pH of 6.6 (Abella 2008).
Soils in Great Plains habitats. The soils occupied by lemon scurfpea in Great Plains habitats are similar to those described above. In Harding County, northwestern South Dakota, lemon scurfpea grew on sand dunes with loamy fine sand soils. These dunes occurred at about 2,950 ft (900 m) in elevation where the climate was continental with temperature extremes of 108 °F (42 °C) in July and -38 °F (-39 °C) in January, average annual precipitation of 15 in (376 mm), and annual average wind speeds of 12 mph (19 km/hr) (Bergmann et al. 2009). In Mellette County, west-central South Dakota, lemon scurfpea was restricted to very weakly developed sands (White 1971). In northwestern Nebraska, it occurred on the south slopes of ponderosa pine forests on sandy loam soils (Nixon 1967). In the Sandhills of Nebraska, lemon scurfpea was a common early-seral species in blowouts, which were 13 to 33 ft (4–10 m) deep crater-shaped depressions averaging 0.2 to 0.7 ac (0.1–0.3 ha) in size. Sandhills had sandy soils with low fertility, organic matter, and water-holding capacity (Stubbendieck et al. 1989).
Description
Lemon scurfpea is a spreading to mat-forming perennial legume from deep, branched, woody roots and rhizomes (Toft and Welsh 1972; Barneby 1989; Hiickman 1993; Tilley and St. John 2010; Welsh et al. 2016). Plants are mostly erect and much branched, at least basally (Fig. 3) (Hickman 1993; Egan and Reveal 2009). Stems are often less than 16 in (40 cm) tall but range from 6 to 27 in (15–70 cm) for this extremely variable species (Toft and Welsh 1972; Barneby 1989; Hickman 1993; Welsh et al. 2016). Stems typically have five or more elongated internodes (Welsh et al. 2016). They are often smooth to slightly striate below and become ribbed above (Barneby 1989). The foliage is green to yellow-green, aromatic, and glabrous to densely pubescent (Toft and Welsh 1972; Barneby 1989; Egan and Reveal 2009; Welsh et al. 2016).

Figure 3. Erect, much branched (basally) lemon scurfpea plant. Photo: Matt Lavin, MSU.
Lemon scurfpea produces alternate, palmately compound leaves mostly with three leaflets, although some have five (Egan and Reveal 2009; Welsh et al. 2016). Petioles measure up to 1.2 in (3 cm) long. Leaflets are linear to obovate, conspicuously gland dotted (Fig. 4), and 0.6 to 2 in (1.4–5 cm) long and 0.5 to 16 mm wide (Munz and Keck 1973; Barneby 1989; Welsh et al. 2016). Leaflets are more narrow and linear upward and can be covered with straight, stiff, flattened hairs (Fig. 4) (Tilley and St. John 2010; Welsh et al. 2016). Lance-subulate stipules are quickly to tardily deciduous (Egan and Reveal 2009).

Figure 4. Three-petiolate leaves of lemon scurfpea with appressed hairs and glands on both sides. Photo: Matt Lavin, MSU.
Lemon scurfpea produces flowers in a dense to interrupted pseudoraceme with one to four but mostly two flowers per node and a total of 5 to 41 flowers per inflorescence (Fig. 5) (Barneby 1989; Egan and Reveal 2009; Welsh et al. 2016). Individual flowers are white to purple or bicolored, papillionaceous, and 4 to 7 mm long (Barneby 1989; Hickman 1993). Flower pedicels (6–7 mm) have persistent ovate to lanceolate bracts (1.2–2.8 mm long). Pedicels are usually less than half as long as the calyx tubes. Calyx is campanulate, five-lobed, with a short tube. Corollas include a reflexed banner (30–90°) (Fig. 6). Stamens are diadelphous in two series (Egan and Reveal 2009; Welsh et al. 2016). Flowers are persistent and do not enlarge as the fruit is develops (Toft and Welsh 1972; Welsh et al. 2016)

Figure 5. Lemon scurfpea inflorescences. Photo: Matt Lavin, MSU.
Lemon scurfpea fruits are one-seeded pods. Pods are round to broadly elliptic and densely villous with glandular warts (Hermann 1966; Munz and Keck 1973; Tilley and St. John 2010; Welsh et al. 2016). Pods are indehiscent, deciduous above the receptacle, and 4 to 7 mm wide and long (Barneby 1989; Egan and Reveal 2009; Welsh et al. 2016). Seeds are brown, smooth, and 4 to 5 mm wide and long (Barneby 1989; Hickman 1993).

Figure 6. Lemon scurfpea papillionaceous flowers. Photo: Matt Lavin, MSU.
Lemon scurfpea produces an expansive network of roots and underground stems. Individual aboveground stems can be separated by more than 2 ft (0.6 m) by this horizontal underground system. Based on the excavation of plants from Kansas, Nebraska, and Colorado prairies, roots can reach depths of 8 to 10 ft (2.4–3 m) (Weaver 1958). On sand dunes in Cherry County, Nebraska, lemon scurfpea plants had an aboveground spread of 4.5 ft (1.4 m). Roots were long, mostly unbranched to 4 ft (1.2 m) deep with branching and lateral roots appearing at 4.5 to 6.5 ft (1.4–2 m) deep (Tolstead 1942). Six lemon scurfpea plants were excavated from the dark brown soil zone in mixed-prairie vegetation in Saskatchewan. Rhizomes were about 3 mm in diameter and spread a distance of 24 in (60 cm) at about 3 to 4 in (7–10 cm) below the soil surface. Roots of about 1.5 to 2 mm in diameter were produced at intervals of several centimeters along the rhizomes. Roots had no or little branching in the uppermost 24 in (60 cm) of soil. At soil depths of 24 to 47 in (60–120 cm), roots produced multi-branched laterals as long as 12 in (30 cm). Nitrogen-fixing nodules occurred at soil depths below 12 in (30 cm). The greatest root depth for plants in this study was 4.3 ft (1.3 m) (Coupland and Johnson 1965).
Reproduction
Lemon scurfpea reproduces from seed (Pool 1914). Regeneration from rhizome fragments is also likely but was not reported in the reviewed literature.
Pollination
Dependence of seed production on pollination has not been examined. Flowers do, however, attract an abundance of native bees [LBJWC 2007]. Flowering is indeterminate, and blooms are possible from April to August (Nixon 1967; Munz and Keck 1973; Beatley 1976; Barneby 1989).
Ecology
Lemon scurpea is a disturbance-tolerant, early-seral forb. It survives top-kill and aboveground damage or burial even as a seedling. Roots also survive heat, wind, and drifting sands (Looman 1980).
Successional status. There are many examples of lemon scurfpea functioning as a pioneer species in early seral conditions (Tolstead 1942; Coupland 1950; Castle 1954; Kelso et al. 2004; Hugenholtz 2010). In the prairie provinces of Canada, lemon scurfpea is common in the early succession of sand dunes. Lemon scurfpea and prairie sagewort (Artemisia frigida) often make up 60% of the total forb cover of early seral sand dune communities. On bare sand, veiny dock (Rumex venosus) is first to colonize and is followed closely by lemon scurfpea, described as a sand-binding species (Coupland 1950).
In a successional study of sand dunes in Kane County, Utah, lemon scurfpea was a pioneer in the valleys between elevated sand dunes and sand dune slopes where soil moisture was greatest and wind exposure lowest. Plant stems elongated rapidly to keep above accumulating sand. At many sites, plant crowns were buried deeply but new growth was found above the sand. In locations where sand had blown away, researchers found tangles of long lemon scurfpea stems (Castle 1954).
Lemon scurfpea in the Bigstick Sand Hills of southwestern Saskatchewan abundance increased with dry and windy conditions. Sand dune succession and vegetation changes were evaluated for a partially filled parabolic dune (Hugenholtz 2010). The dune (140 mi² [360 km²]), is a vegetated and stabilized field surrounded by mixed-grassland communities. It has east-trending dunes and blowouts traveling at an average rate of 11.5 ft (3.5 m)/year. In 2003, lemon scurfpea occupied 8% of the study dune’s surface. In 2009, it occupied 56% of the surface. Increases in lemon scurfpea abundance coincided with drier than normal conditions and steady growing-season winds (Hugenholtz 2010).
Lemon scurfpea and yellow wildrye (Leymus flavescens) are common in the pioneer stage of succession on active sand dunes in Freemont County, southeastern Idaho. The pioneer stage lasts up to 30 years (Chadwick and Dalke 1965). On active dunes, sand moved northeast at an average rate of 10 ft (3 m)/year. In pioneer communities, lemon scurfpea cover averaged 25%. The next seral stage was represented by a rubber rabbitbrush-yellow wildrye-lemon scurfpea community that persists for 10 to 70 years. Lemon scurfpea cover decreased as succession progressed and was often restricted to places with low plant cover, open sand exposed, and evidence of disturbance. In a nearby stabilized sand site dominated by antelope bitterbrush (Purshia tridentata) and chokecherry (Prunus virginiana), lemon scurfpea cover increased to up to 24% following a severe fire evaluated 8 to 18 years later. Lemon scurfpea cover was lower in this burned community than in pioneer sand dune communities (Chadwick and Dalke 1965).
Seed And Seedling Ecology
Lemon scurfpea seedlings were commonly observed in crater formations in the sandhills of Nebraska (Pool 1914). In northern Cherry County, Nebraska, lemon scurfpea grew best as a pioneer species in blowouts and other disturbed sites on sand dunes and in dry meadows. Plants in stable late-seral vegetation did not produce seed (Tolstead 1942).
In the prairie provinces of Canada, seed production was low, averaging 135 seeds/plant (Looman 1980). Insect damage to seeds was often 60 to 100%. The likely seed predator was bean weevils (Acanthoscelides spp. lemon scurfpea seedling root growth was rapid, averaging 1 in (2.5 cm)/day. Belowground stems appeared 10 to 15 days after emergence (Looman 1980).
Disturbance Ecology
Lemon scurfpea commonly occurs on disturbed sites and increases in abundance following disturbance (Pool 1914; Toft and Welsh 1972; McGinnies et al. 1991). As indicated above, lemon scurfpea thrives on active sand dunes, which result from heavy grazing and drought in dry grasslands in the prairie provinces of Canada (Looman 1980). In the sandhills of Nebraska, occurrence of lemon scurfpea increased with heavy grazing in sand bluestem and little bluestem grasslands (Pool 1914). Following the great drought of 1934 in Nebraska and Kansas, lemon scurfpea and other forbs replaced grasses on heavily grazed prairies (Weaver and Albertson 1936). In the Sangre de Cristo Mountains of north-central New Mexico, lemon scurfpea grew along roadsides and in floodplain-arroyo riparian vegetation types. The riparian sites occupied low-elevation floodplains along the Rio Grande River that were exposed to disturbances and dry conditions (Larson et al. 2014).
Wildlife And Livestock Use
Ungulates, birds, insects, and pollinators utilize lemon scurfpea. It is grazed by pronghorn (Antilocapra americana) (Hepworth 1965) and is considered a preferred food of mule deer (Odocoileus hemionus) in New Mexico (Bender 2006 cited in Kramer et al. 2015). Descriptions of its palatability to livestock, however, range from palatable (Milchunas and Noy-Meir 2004) to not readily grazed (Tolstead 1942) to poisonous when ingested in large quantities (Looman 1980).
Lemon scurfpea provides food and cover to game birds. It was found in 29% of the crops but made up just 0.1% of crop volume of seven gray partridge (Perdix perdix) birds harvested in the fall from the Colville Confederated Tribal Reservation in Washington (Knight et al. 1979). Sharp-tailed grouse (Tympanuchus phasianellus) broods used it for cover in South Dakota (Janson 1956, cited in Grosz 1988).
Lemon scurfpea flowers are visited by many native bees [LBJWC 2007]. Lemon scurfpea habitats are also important to endemic tiger beetles (Cicindela albissima) in Coral Pink Sand Dunes in Utah (Kinsley and Hill 2001) and to many insect species in Great Sand Dunes National Monument, Colorado (Weissmann 1997).
Ethnobotany
Western tribes used lemon scurfpea as a tool, medicine, food, and in ceremonies. The Paiute called it ‘pooy sonib’, and they and other Great Basin tribes used the roots, which can be split into fine water-proof pieces, as strings in nets and other tools (Van Allen Murphey 1990; Moerman 2003). Arapahos chewed fresh leaves to treat throat and voice issues (Van Allen Murphey 1990). They also applied an infusion of leaves to treat dry skin, itching, and sores. Arapahos inhaled leaves for headaches and chewed roots for hoarseness (Moerman 2003). The Navajo used a decoction of lemon scurfpea and other plants to treat venereal disease. A cold infusion of lemon scurfpea was applied as a lotion during Lifeway and Gameway chants and used for protection from witches. This cold infusion was also drunk to treat stomach aches and menstrual pains (Vestal 1952). Zuni ate fresh flowers for stomach aches (Moerman 2003). Cheyenne Indians ate lemon scurfpea roots and used the plant ceremonially (Yanovsky 1936; Moerman 2003).
Horticulture
A 2021 internet search suggests that lemon scurfpea is not readily available as seed or plants from native nurseries or online sources. Yet it may have potential as a low-maintenance horticultural species in the future for use along roadways, at rest areas, or in campgrounds. It would provide pollinator food and wildlife habitat for dry sites with coarse-textured or sandy soils.
Revegetation Use
Lemon scurfpea has several characteristics that make it a good species to use in the restoration of dry, course-textured, or sandy soil types (Hermann 1966; Lambert 2005). It tolerates early-seral conditions, poor soil development, and drought situations, and as a nitrogen-fixer, it may help ecosystems recover from disturbance faster than a species that does not fix nitrogen (Tolstead 1942; Coupland 1950; Castle 1954; Kelso et al. 2004; Hugenholtz 2010). It also tolerates disturbances as a young plant (Looman 1980). Its vegetative growth makes it an excellent soil binder and important in moving succession forward (Kearney and Peebles 1960 cited in Hermann 1966). It is also used by wildlife and pollinators (Janson 1956, cited in Grosz 1988; Hepworth 1965; Knight et al. 1979; Bender 2006 cited in Kramer et al. 2015; LBJWC 2007). Low rates of flower production and indeterminate ripening of seed, however, make seed field production of lemon scurfpea a costly, if improbable endeavor (D. Tilley, USDA NRCS, personal communication, January 2022).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for lemon scurfpea, generalized provisional seed zones developed by Bower et al. (2014), may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat to moisture index). In Figure 7, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Restoration Tool (Richardson et al. 2020) can also guide revegetation planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
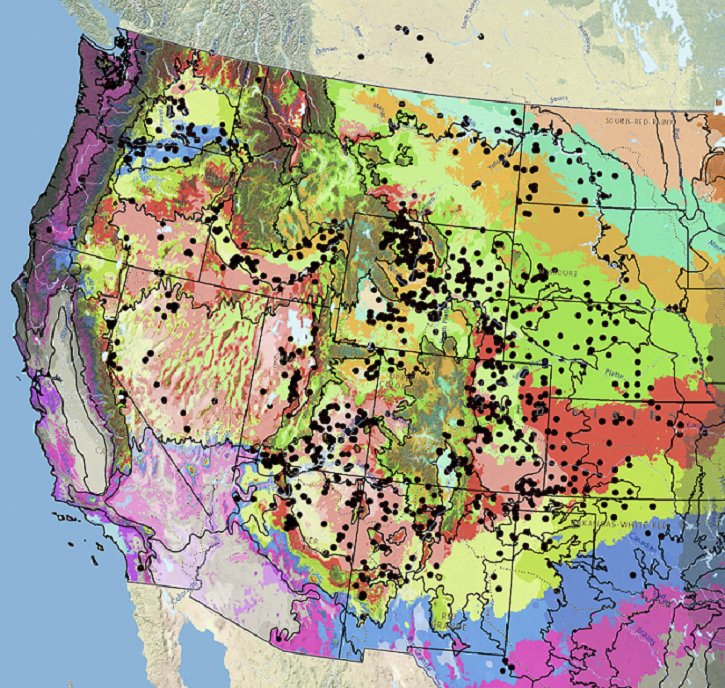
Figure 7. Distribution of lemon scurfpea (black circles) based on geo-referenced herbarium specimens and observational data from 1881–2016 (CPNWH 2020; SEINet 2020; USDI USGS 2020). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USDI USGS.
Releases
As of 2021, there were no lemon scurfpea germplasm releases
Wildland Seed Collection
Lemon scurfpea seeds ripen indeterminately, and although pods do not shatter, they may be dispersed when seeds are mature. Monitoring and revisiting populations is important to collect any quantity of high-quality seed (Tilley and St. John 2010). In the prairie provinces of Canada, lemon scurfpea seed production was low, averaging 135 seeds/plant, and between 60 and 100% of the seeds were insect damaged (Looman 1980). This suggests that wildland seed harvests may be time consuming.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (UCIA 2015; Young et al. 2020). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Seed is often mature in July or August (Tilley 2011; USDI BLM SOS 2020). The Bureau of Land Management’s Seeds of Success collection crews made eight lemon scurfpea seed collections from 2011 to 2016 (USDI BLM SOS 2020). Harvests were made in Utah, Colorado, and Wyoming. The earliest collection occurred on June 29, 2014, at 4,252 ft (1,296 m) in elevation in Baca County, Colorado. The latest collection was made on September 1, 2016, at a 7,057-ft (2,151-m) site in Carbon County, Wyoming. At many sites, subsequent collections were made from the same sites 3 days to almost 2 weeks following initial collections (USDI BLM SOS 2020). Tilley (2011) collected lemon scurfpea seed from a 5,000-ft (1,524-m) elevation sagebrush grassland in Park County, Wyoming. Seed was collected from July 25 to August 8 at this site, where soils were gravelly sandy loams, and annual precipitation averaged 9 in (230 mm).
Collection Methods
Wildland seed can be collected by hand stripping or clipping inflorescences into a container (Tilley and St. John 2010). Because seed ripens indeterminately, harvests made by hand-stripping would be the preferred collection method early in the seed maturation process.
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of lemon scurfpea.
Post-Collection Management
Seed collections should be stored in breathable containers and protected from rodents until they can be thoroughly dried and cleaned. Insects present in seed harvests can be controlled by freezing or adding mothballs to the collection (D. Tilley, USDA NRCS, personal communication, January 2022).
Seed Cleaning
The Aberdeen Plant Materials Center (US Department of Agriculture, Natural Resources Conservation Service) reported the following procedure to clean a small hand-collected seed lot (no seed weight provided). Seed was separated from the pods using a Westrup Model LA-H laboratory brush machine with a #7 mantel, a speed of 2, and the gate fully closed. The brush machine ran for about 2 minutes. Inert material was removed using a multi-deck air screen cleaner with a 4.75-mm top screen, a 2.10-mm bottom screen, and an air speed high enough to blow off chaff and empty, shriveled, and insect-damaged seed (Tilley 2011).

Figure 8. Lemon scurfpea pod (left) and seed (right). Photo: BLM SOS WY-020.
Seed Storage
No description of seed storage or seed testing requirements or procedures specific to lemon scurfpea were available in the reviewed literature.
Germination Biology
Lemon scurfpea seed has a hard seed coat. Just 14% of lemon scurfpea seed collected in eastern Washington germinated following a hot-water scarification treatment (Link 2017). This process involved boiling water, removing it from the heat source, then adding seed and allowing the water and the seed to cool overnight. Scarifying the seed with sandpaper caused the seed to split. Link (2017) suggested that acid scarification used to improve germination of other hard-seeded peas (Fabaceae) could be useful for lemon scurfpea. Effects of acid scarification on lemon scurfpea germination should be tested on a sample before conducting any large-scale treatments and to determine time required for effective treatment.
Wildland Seed Yield And Quality
Post-cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 1 (USFS BSE 2017). The results indicate that lemon scurfpea seed can generally be cleaned to high levels of purity and seed fill but that the viability of fresh seed is variable.
Table 1. Seed yield and quality of lemon scurfpea seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Seed lot characteristic |
Mean |
Range |
Samples (no.) |
|
Bulk weight (lbs) |
1.42 |
0.34–4.19 |
6 |
|
Clean weight (lbs) |
0.43 |
0.15–0.80 |
6 |
|
Clean-out ratio |
0.43 |
0.08–0.72 |
6 |
|
Purity (%) |
97 |
91–99 |
6 |
|
Fill (%)¹ |
90 |
86–96 |
6 |
|
Viability (%)² |
89 |
79–97 |
5 |
|
Seeds/lb |
23,514 |
20,040–25,676 |
6 |
|
Pure live seeds/lb |
20,149 |
16,960–24,392 |
5 |
¹100 seed X-ray test
²Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well.
Agricultural Seed Production
Agricultural production of lemon scurfpea seed was not reported in the available literature. The following fungal pests were found on wildland populations of lemon scurfpea: stem spots (Septoria psammophila and S. umatillensis), and Uromyces psoraleae (Farr and Rossman 2017). It is unknown if any of these pests would impact agricultural or nursery production.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (UCIA 2015; Young et al. 2020).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Nursery Practice
Link (2017) grew lemon scurfpea seedlings from seed collected in eastern Washington. Seed was hot-water scarified (see Germination Biology section). In November or December, treated seeds and any germinated seeds were sown in cone-tainers (10 in³ [164 ml]) filled with 46% Miracle Gro™ potting soil, 46% sand, 4% perlite, and 4% vermiculite. Cone-tainers were placed in the greenhouse, watered for 15 minutes three times each day, and fertilized (NPK 20-20-20) once each week starting the second week of January (Link 2017). Number of seedlings produced was not reported.
Wildland Seeding And Planting
Based on the available literature, use of lemon scurfpea in wildland restoration has been limited. Nursery plants grown by Link (2017) were successfully outplanted at a winery in the driest part of eastern Washington (Benton County, 682 ft [208 m] elevation, and 5 to 7 in [127–178 mm] of precipitation/yr). Seed to grow these seedlings was collected within 6.8 miles (11 km) of the winery. Seedlings were planted in two vineyards, one on loamy fine sand soils and the other on silt loam soils. Seedlings were planted on both sides of the grapevines 3 to 3.9 ft (0.9–1.2 m) from the vines and 0.5 ft (0.2m) apart within rows. Individual seedlings were irrigated with about 9.5 gal (36 L) of water just once after planting (March–April 2009). Seedlings were protected from herbivory by mesh plastic tubes. Stem count changes were evaluated for 3 years. After 3 months, lemon scurfpea stem counts were 30 to 40% of original stem counts, but after 39 months, stem counts were 55% on loamy fine sands and 5% on silt loams. At the loamy fine sands location, stem counts in 2011, were 22% of the original, and in 2012, they increased to 55%. Increases in 2012 were highly variable, ranging from 0 to 136% across all six study plots. Some surviving plants produced enough belowground growth to support numerous stems. In 2012, lemon scurfpea plants averaged 12 in (31 cm) tall, and there were 43 total fruits produced for the species. About 10% of surviving plants flowered 39 months after planting, and the percentage of flowering plants was increasing, but no establishment from self-seeding was observed. There was no evidence of herbivory in any year (Link 2017).
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Matt Lavin, Botanist, University of Montana; Derek Tilley, Aberdeen Plant Materials Center Manager, USDA NRCS; E. Durant McArthur, Emeritus Scientist, USDA Forest Service Rocky Mountain Research Station.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Abella, S.R. 2008. A unique old-growth ponderosa pine forest in northern Arizona. Journal of the Arizona-Nevada Academy of Science. 40(1): 1-11.
Anderson, J.E.; Inouye, R.S. 2001. Landscape-scale changes in plant species abundance and biodiversity of a sagebrush steppe over 45 years. Ecological Monographs. 71(4): 531-556.
Anderson, L.C. 2006. Ericameria nauseosa subsp. ammophila (Asteraceae), a new rabbitbrush from the San Luis Valley of Colorado. SIDA, Contributions to Botany. 22(2): 867-872.
Association of Official Seed Analysts [AOSA]. 2016. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Barneby, R.C. 1989. Intermountain Flora Volume 3, Part B: Fabales. In: Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K., eds. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Bronx, NY: The New York Botanical Garden. 279 p.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Beatley, J.C. 1976. Vascular plants of the Nevada Test Site and central-southern Nevada: Ecologic and geographic distributions. TID-26881. Washington, DC: Technical Information Center, Energy Research and Development Administration. 306 p.
Bergmann, D.; Zehfus, M.; Zierer, L.; Smith, B.; Gabel, M. 2009. Grass rhizosheaths: Associated bacterial communities and potential for nitrogen fixation. Western North American Naturalist. 69(1): 105-114.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Castle, E.S. 1954. The succession of vegetation on a southern Utah sand dune. Provo, UT: Brigham Young University. Thesis. 98 p.
Chadwick, H.W.; Dalke, P.D. 1965. Plant succession on dune sands in Fremont County, Idaho. Ecology. 46(6): 766-780.
Christie, K. 2008. Vascular flora of the Lower San Francisco Volcanic Field, Coconino County, Arizona. Madroño. 55(1): 1-14.
Clambey, G.K. 1992. Ecological aspects of the Knife River Indian Villages National Historic Site, west-central North Dakota. In: Smith, D.D.; Jacobs, C.A., eds. Recapturing a vanishing heritage: Proceedings, 12th North American Prairie Conference; 1990 August 5-9; Cedar Falls, IA. Cedar Falls, IA: University of Northern Iowa: 75-78.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2020. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Coupland, R.T. 1950. Ecology of mixed prairie in Canada. Ecological Monographs. 20(4): 271-315.
Coupland, R.T.; Johnson, R.E. 1965. Rooting characteristics of native grassland species in Saskatchewan. Journal of Ecology. 53(2): 475-507.
Egan, A.N.; Reveal, J.L. 2009. A new combination in Pediomelum and a new genus, Ladeania, from western North America (Fabaceae, Psoraleeae). Novon. 19(3): 310-314.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Farr, D.F.; Rossman, A.Y. 2017. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://nt.ars-grin.gov/fungaldatabases/.
Franklin, J.F.; Dyrness, C.T. 1973. Natural vegetation of Oregon and Washington. Gen. Tech. Rep. PNW-8. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station. 452 p.
Grimm, E.C. 2001. Trends and palaeoecological problems in the vegetation and climate history of the northern Great Plains, U.S.A. Biology and Environment: Proceedings of the Royal Irish Academy. 101B(1/2): 47-64.
Grosz, K.L. 1988. Sharp-tailed grouse nesting and brood rearing habitat in grazed and nongrazed treatments in southcentral North Dakota. Fargo, North Dakota: North Dakota State University. Thesis. 72 p.
Hepworth, B. 1965. Investigation of pronghorn antelope in Wyoming. In: Proceedings of the 1st Annual antelope States Workshop; 1965 April 14-15. Santa Fe, NM: New Mexico Department of Fish and Game. 1-12 p.
Hermann, F. 1966. Notes on western range forbs: Cruciferae through Compositae. Agric. Handb. 293. Washington, DC: U.S. Department of Agriculture, Forest Service. 365 p.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
Hugenholtz, C.H. 2010. Topographic changes of a supply-limited inland parabolic sand dune during the incipient phase of stabilization. Earth Surface Processes and Landforms. 35(14): 1674-1681.
ITIS Database. 2021. Integrated Taxonomic Information System. Available: http://www.itis.gov/index.html
Johnston, B.C. 1987. Plant associations of Region 2: Potential plant communities of Wyoming, South Dakota, Nebraska, Colorado, and Kansas. Edition 4. R2-ECOL-87-2. Lakewood, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. 429 p.
Kelso, T.; Bower, N.; Halteman, P.; Tenney, K.; Weaver, S. 2004. Dune communities of southeast Colorado: Patterns of rarity, disjunction and succession. In: Barlow, I.P.; Anderson, J.; McDonald, C., tech eds. Southwestern rare and endangered plants: Proceedings of the 4th Conference. 2001 March 22-26; Las Cruces, NM; Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 37-48.
Kinsley, C.B.; Hill, J.H. 2001. Biology and conservation of the Coral Pink Sand Dunes tiger beetle Cicindela limbata albissima Rumpp. Western North American Naturalist. 61(4): 381-394.
Knight, R.L.; Every, A.D.; Erickson, A.W. 1979. Seasonal food habits of four game bird species in Okanogan County, Washington. The Murrelet. 60(2): 58-66.
Kramer, D.W.; Sorensen, G.E.; Taylor, C.A.; Cox, R.D.; Gipson, P.S.; Cain, J.W. 2015. Ungulate exclusion, conifer thinning and mule deer forage in northeastern New Mexico. Journal of Arid Environments. 113: 29-34.
Lady Bird Johnson Wildflower Center [LBJWC]. 2007. Psoralidium lanceolatum (Pursh) Rydb. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants-main [Accessed 2021 October 10].
Lambert, S. 2005. Guidebook to the seeds of native and non-native grasses, forbs and shrubs of the Great Basin. Boise, ID: U.S. Department of the Interior, Bureau of Land Management, Idaho State Office. 136 p.
Larson, J.; Reif, B.; Nelson, B.E.; Hartman, R.L. 2014. Floristic studies in north central New Mexico, U.S.A. The Sangre de Cristo Mountains. Journal of the Botanical Research Institute of Texas. 8(1): 271-303.
Link, S.O. 2017. Success of establishing nitrogen-fixing native species as cover crops in silt loam and loamy fine sand in a vineyard in the Columbia Basin. Native Plants Journal. 18(1): 62-76.
Looman, J. 1963. Preliminary classification of grasslands in Saskatchewan. Ecology. 44(1): 15-29.
Looman, J. 1980. The vegetation of the Canadian Prairie Provinces II. The grasslands, part 1. Phytocoenologia. 8(2): 153-190.
Ludwig, F.; Jewitt, R.A.; Donovan, L.A. 2006. Nutrient and water addition effects on day- and night-time conductance and transpiration in a C3; desert annual. Oecologia. 148(2): 219-225.
McArthur, E.D.; Sanderson, S.C. 1991. Great Sand Dunes National Monument vegetation patterns. University of Wyoming National Park Service Research Center Annual Report. 15(37): 1-5.
McGinnies, W.J.; Shantz, H.L.; McGinnies, W.G. 1991. Changes in vegetation and land use in eastern Colorado: A photographic study, 1904-1986. ARS-85. Washington, DC: U.S. Department of Agriculture, Agricultural Research Service. 180 p.
Milchunas, D.G.; Noy-Meir, I. 2004. Geologic grazing refuges and grassland diversity: A shortgrass steppe study. Journal of Range Management. 57(2): 141-147.
Moerman, D. 2003. Native American ethnobotany: A database of foods, drugs, dyes, and fibers of Native American peoples, derived from plants. Dearborn, MI: University of Michigan. http://naeb.brit.org
Moss, E.H. 1955. The vegetation of Alberta. Botanical Review. 21(9): 493-567.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Nixon, E.S. 1967. A vegetational study of the Pine Ridge of northwestern Nebraska. The Southwestern Naturalist. 12(2): 134-145.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Pavlik, B.M. 1985. Sand dune flora of the Great Basin and Mojave Deserts of California, Nevada, and Oregon. Madroño. 32(4): 197-213.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Pool, R.J. 1914. A study of the vegetation of the sandhills of Nebraska. Minneapolis, MN: University of Minnesota. Thesis. 327 p.
Reif, B.; Larson, J.; Jacobs, B.F.; Nelson, B.E.; Hartman, R.L. 2009. Floristic studies in north central New Mexico, U.S.A. the Tusas Mountains and Jemez Mountains. Journal of the Botanical Research Institute of Texas. 3(2): 921-961.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Rosenthal, D.M.; Ludwig, F.; Donovan, L.A. 2005. Plant responses to an edaphic gradient across an active sand dune/desert boundary in the Great Basin Desert. International Journal of Plant Sciences. 166(2): 247-255.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2020. SEINet Regional Networks of North American Herbaria. https://Symbiota.org/docs/seinet
Shiflet, T.N. 1994. Rangeland cover types of the United States. Denver, CO: Society for Range Management. 152 p.
Stubbendieck, J.; Flessner, T.R.; Weedon, R.R. 1989. Blowouts in the Nebraska Sandhills: The habitat of Penstemon haydenii. In: Bragg, T.B.; Stubbendieck, J., eds. Prairie pioneers: Ecology, history and culture: Proceedings, 11th North American Prairie Conference. 1988 August 7-11; Lincoln, NE; Lincoln, NE: University of Nebraska: 223-225.
Tilley, D. 2011. Propagation protocol for production of propagules Psoralidium lanceolatum (Pursh) Rydb. seeds. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2021 October 5].
Tilley, D.; St. John, L. 2010. Request for seed collections: Dune scurfpea (Psoralidium lanceolatum [Pursh] Rydberg). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 2 p.
Toft, C.A.; Welsh, S.L. 1972. A revision of the Psoralea lanceolata complex (Leguminosae). The Great Basin Naturalist. 32(2): 76-87.
Tolstead, W.L. 1942. Vegetation of the northern part of Cherry County, Nebraska. Ecological Monographs. 12(3): 255-292.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2021. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2016. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 37 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2020. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2020. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Van Allen Murphey, E. 1990. Indian uses of plants. Glenwood, IL. Meyerbooks. 81 p.
Vestal, P.A. 1952. Ethnobotany of the Ramah Navaho. Reports of the Ramah Project: No. 4. Papers of the Peabody Museum of American Archeology and Ethnology. Cambridge, MA: Harvard University. 40(4): 1-94.
Weaver, J.E. 1958. Classification of root systems of forbs of grassland and a consideration of their significance. Ecology. 39(3): 394-401.
Weaver, J.E.; Albertson, F.W. 1936. Effects of the great drought on the prairies of Iowa, Nebraska, and Kansas. Ecology. 17(4): 567-639.
Weissmann, M.J. 1997. Natural history of the giant sand treader camel cricket, Daihinibaenetes giganteus Tinkham (Orthoptera: Rhaphidophoridae). Journal of Orthoptera Research. 6: 33-48.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Whitcomb, R.F. 1989. Nebraska Sand Hills: The last prairie. In: Bragg, T.B.; Stubbendieck, J., eds. Prairie pioneers: Ecology, history and culture: Proceedings, 11th North American Prairie Conference. 1988 August 7-11; Lincoln, NE; Lincoln, NE: University of Nebraska: 57-69.
White, E.M. 1971. Some soil age-range vegetation relationships. Journal of Range Management. 24(5): 360-365.
Yanovsky, E. 1936. Food plants of the North American Indians. Misc. Pub. 237. Washington, DC: U.S. Department of Agriculture. 84 p.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2022. Lemon scurfpea (Psoralidium lanceolatum). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: http://greatbasinfirescience.org/western-forbs-restoration