Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
November 2018
Update
October 2025
Nomenclature
Munro’s globemallow (Sphaeralcea munroana [Douglas ex Lindley] Spach.) belongs to the Malvaceae family and the Malveae tribe (Kearney 1935; La Duke 2016).
Family
Malvaceae – Mallow family
Genus
Sphaeralcea
Species
munroana
NRCS Plant Code
SPMU2 (USDA NRCS 2025).
Subtaxa
The Flora of North America does not recognize any subspecies or varieties (La Duke 2016).
Synonyms
Malva munroana Douglas ex Lindley, M. creeana Graham; Malvastrum munroanum (Douglas ex Lindley) A. Gray, Malveopsis munroana (Douglas ex Lindley) Kuntze, Nuttallia munroana (Douglas ex Lindley) Nuttall, Sphaeralcea munroana subsp. subrhomboidea (Rydberg) Kearney, S. munroana var. subrhomboidea (Rydberg) Kearney, S. subrhomboidea Rydberg (La Duke 2016).
Common Names
Desert mallow, Munroe’s desert-mallow, Munroe globemallow, orange globemallow, salmon globemallow, scarlet globemallow, whitestem globemallow (Andersen and Holmgren 1976; Gautier and Everett 1979; USDA NRCS 2000; Pavek et al. 2011; La Duke 2016; Welsh et al. 2015).
Chromosome Number
Chromosome numbers are: 2n = 10 or 20 (Rumbaugh and Pendery 1993; Holmgren et al. 2005).
Hybridization
Munro’s globemallow and small-leaf globemallow (S. parvifolia) may be conspecific (Holmgren et al. 2005; La Duke 2016). Intergradation of Munro’s globemallow with gooseberryleaf globemallow (S. grossulariifolia) and small-leaf globemallow is suspected where species distributions overlap (Kearney 1935; Rumbaugh and Pendery 1993; Holmgren et al. 2005). Hybrid swarms between seeded and natural populations of Munro’s globemallow, gooseberryleaf globemallow, and desert globemallow (S. ambigua) were suspected when allozymes and molecular genetic markers were evaluated (McArthur and Sanderson 2005).
Distribution
Munro’s globemallow is a western species occurring in southern British Columbia, east of the Cascades in Washington and Oregon, as far south as Nevada and Utah and as far east as southwestern Wyoming (Holmgren et al. 2005; La Duke 2016; Welsh et al. 2015; LBJWC 2023). Distribution is very limited in California, where it is known only in Placer County (LaDuke 2012). Munro’s globemallow is more common in northern Utah and the Wasatch Plateau, whereas gooseberryleaf globemallow is more common in the southern part of the state (Pendery and Rumbaugh 1986).
Habitat And Plant Associations
Munro’s globemallow is commonly found on xeric desert plains and lower mountain slopes receiving 6 to 14 in (152-356 mm) of annual precipitation (Ogle et al. 2012; La Duke 2016; Hitchcock and Cronquist 2018). It is often found growing in mixed desert shrub, sagebrush (Artemisia spp.), saltbush (Atriplex spp.), rabbitbrush (Chrysothamnus spp.), juniper (Juniperus spp.), and mountain brush communities (Figs. 1, 2) Rumbaugh and Pendery 1993; Holmgren et al. 2005; Welsh et al. 2015). Munro’s globemallow is common on roadsides (Fig. 2, Lesica 2012).

Figure 1. Munro’s globemallow growing in sagebrush habitat in Oregon. Photo: USDI Bureau of Land Management (BLM) OR030 Seeds of Success (SOS).
Elevation
Munro’s globemallow populations occupy sites ranging from 330 to 8,000 ft (100-2,440 m) in elevation across its range (Holmgren et al. 2005; Welsh et al. 2015; La Duke 2016). It occurs from 4,500 to 8,000 ft (1,370-2,440 m) in Utah (Welsh et al. 2015).
Soils
Munro’s globemallow grows in many soil types but is most common on sandy to clay-loam or gravelly to rocky soils with low to extremely low moisture (Taylor 1992; Pendery and Rumbaugh 1993; Luna et al. 2018; Oregon Flora 2025). It is often found on dry, open, disturbed sites with light soils of volcanic origin (Kearney 1935; Pendery and Rumbaugh 1993; La Duke 2012).
Description
Munro’s globemallow is a cool-season perennial with a woody, branching crown from a long, thick, strong woody taproot (Pendery and Rumbaugh 1986; Taylor 1992; Kildisheva and Davis 2012). It is sometimes considered a subshrub (La Duke 2012). The taproot is branched with several surface roots (USDA NRCS 2000). Several to many slender, erect stems (7-35 in [18-90 cm] tall) arise from the caudex (Fig. 3) (Munz and Keck 1973; Welsh et al. 2015; La Duke 2016). Stems are up to 6 mm in diameter at the base, sparsely to densely hairy, and unbranched or with short branches (Kearney 1935; Munz and Keck 1973; Holmgren et al. 2005; La Duke 2016).
Leaves range from yellowish to bright to dark green or gray-green with a dense covering of star-shaped hairs (Taylor 1992; Welsh et al. 2015). Plants do not produce basal leaves; stem leaves are arranged alternately (Holmgren et al. 2005; Pavek et al. 2011). Leaf blades are thin, egg-shaped to triangular, and distinctly shallowly to deeply three- to five-lobed with round-toothed margins and stellate pubescent surfaces (Kearney 1935; Munz and Keck 1973; Pavek et al. 2011; La Duke 2016). Lobes extend half way or more to the midvein in some cases causing the leaves to appear compound (Mansfield 2000; Parkinson 2003). Leaves are about 0.4 to 2 in (1-6 cm) long and wide with 0.8 to 1.5-in (2-4 cm) long petioles (Munz and Keck 1973; Welsh et al. 2015). Leaves are palmately five-veined from the base, but the veins are not prominent on the undersides of leaves (Kearney 1935; Holmgren et al. 2005).

Figure 2. Munro’s globemallow growing along a Utah roadside in sagebrush habitat. Photo: BLM UT931 SOS.

Figure 3. Munro’s globemallow growing in Idaho. Photo: BLM ID931 SOS.
Flowers (often >25) are clustered in the leaf axils of leafy, narrow, racemose inflorescences (Fig. 4) (Rumbaugh and Pendery 1993; Welsh et al. 2015; La Duke 2016; Hitchcock and Cronquist 2018; LBJWC 2023). Individual flowers are about 2 cm in diameter with five pale orange to brick red petals. Petals overlap at the base to form a bowl containing a column of numerous stamens and the pistil (Taylor 1992; Holmgren et al. 2005; Pavek et al. 2011; La Duke 2016). Sepals are densely stellate-pubescent and retained through anthesis (Munz and Keck 1973; Holmgren et al. 2005; Welsh et al. 2015). Munro’s globemallow produces rounded schizocarps, which are divided into 10 to 13 generally single-seeded segments (mericarps) at maturity (Fig. 5, Taylor 1992; Lesica 2012). The mericarps are partially dehiscent and contain the seeds, which are brown, kidney-shaped, slightly hairy, and about 1.7 mm long (Fig. 6, Holmgren et al. 2005; Welsh et al. 2015; La Duke 2016).

Figure 4. Munro’s globemallow flowers on a plant growing in Utah. Photo: BLM UT931 SOS.

Figure 5. Munro’s globemallow schizocarps and what are generally single-seeded mericarps collected from plants in Idaho. Photo: BLM ID931 SOS.

Figure 6. Munro’s globemallow seed. Photo: J. Cane, USDA ARS.
It can be difficult to distinguish Munro’s globemallow from gooseberryleaf globemallow and small-leaf globemallow (Holmgren et al. 2005; La Duke 2016). In general, Munro’s globemallow occupies more northern habitats and is larger than small-leaf globemallow (La Duke 2016). Munro’s globemallow is more common at lower elevations than gooseberryleaf globemallow (Parkinson 2003). Munro’s globemallow mericarps dehisce more readily than those of other globemallow species (Sphaeralcea spp.) (Pendery and Rumbaugh 1986).
Reproduction
Munro’s globemallow reproduces entirely from seed (Pendery and Rumbaugh 1986).
Phenology
Munro’s globemallow flowers are common from May to July (LBJWC 2023).
Breeding System
Flowers are strongly outcrossing and insect-pollinated (Pendery and Rumbaugh 1986; 1993). In the greenhouse, few schizocarps and little seed were produced when flowers were self-pollinated. When manually outcrossed, 8 to 10 times more schizocarps and 5 to 9 times as many seeds/fruit were produced , which was similar to seed production by openly visited plants growing outdoors in Logan, Utah (Cane 2009).
Pollination
Munro’s globemallow is a good pollinator species (Ogle et al. 2012), and its flowers are visited by a variety of specialist and generalist bee and wasp species (Fig. 5) (Pendery and Rumbaugh 1986). Important pollinators include sweat bees (Agapostemon and Halictus spp.), aphid wasps (Ammoplanus spp.), honey bees (Apis spp.), panurgine bees (Calliopsis spp.), sunflower bees (Diadasia spp.), and long-horned bees (Melissodes spp.) (Pendery and Rumbaugh 1986). At sites in Idaho, Nevada, Utah, and Wyoming, an average of 14.5 native bee visitors were observed for every 100 globemallow (gooseberryleaf and Munro’s) plants examined (Cane and Love 2016).
Ground nesting bees (Diadasia diminuta, D. lutzi, and Colletes sphaeralcea) are globemallow and related species specialists (Cane cited in Pavek et al. 2011). Native sunflower bees (Diadasia spp.) were present in every wild population of globemallows sampled across Nevada, eastern Oregon, southern Idaho, and northwestern Utah in 2006. These ground nesters were also found nesting in globemallow seed production plots planted in Ontario, Oregon (Cane cited in Pavek et al. 2011).
Munro’s globemallow flowers were visited most by ground nesting (Colletes spp.), sunflower, sweat (Lasioglossum spp.), and mining (Andrena spp.) bees in big sagebrush habitats near Boise, Idaho, where cover of nonnative species was high (Sun 2023). The field study was designed to test the effects of tall tumblemustard (Sisymbrium altissimum) on pollinator visitation of Munro’s globemallow but no consistent effect was found. Munro’s globemallow flowers were also visited by long-horned (Eucera spp.), cuckoo (Nomada spp.), sweat (Agapostemon and Halictus spp.) and Ashmaediella bees. Only mining and sweat (Lasioglossum spp.) bees were netted from both Munro’s globemallow and tall tumblemustard (Sun 2023).
Ecology
Munro’s globemallow is often found on disturbed or open sites (Andersen and Holmgren 1976; Pendery and Rumbaugh 1993; Lesica 2012). It is common to see globemallow populations fluctuate over time, establishing in large numbers during favorable conditions or on favorable sites, surviving and reproducing for a few years, and then disappearing or becoming less abundant but persisting at low densities or in the seed bank (Pendery and Rumbaugh 1993).
Seed And Seedling Ecology
Munro’s globemallow seed has a hard seed coat that requires scarification to become water permeable (Kildisheva et al. 2011), however, its longevity in the seedbank is unknown. In Wyoming big sagebrush vegetation in Tooele County, Utah, Munro’s globemallow was more common in the seed bank than in aboveground vegetation (Pekas and Schupp 2013).
Seed production can vary by site, climatic conditions, and pest pressure. Birds, rabbits, and weevils (Macrophoptus spp.) feed on globemallow seeds and are capable of limiting seed production (Pendery and Rumbaugh 1986). When plant growth was compared at two dryland nursery sites in northern Utah and southern Idaho, precipitation differences were the likely cause of differences in Munro’s globemallow seed production. Much more seed was produced at the Utah site, which received 2 to 16 in (59-405 mm) more annual precipitation over the 4-year study period (Pendery and Rumbaugh 1990).
Harvester ants (Pogonomyrmex occidentalis) may be important dispersers of Munro’s globemallow seed (Mull 2003). In big sagebrush-rabbitbrush vegetation near Kemmerer, Wyoming, Munro’s globemallow seed was recovered from nearly half harvester ant middens (13 of 30). Middens represent the collection of materials discarded by the ants over time. An average of 50.7 seeds/ft² (557.5/m²) were recovered from the middens, which were located near the entrances to the ant mounds. Seed was not recovered from soil samples from areas beyond the mounds or middens, which included the denuded areas surrounding the mounds and middens to a distance of 16 ft (5 m) from mound centers (Mull 2003).
An experiment evaluating the fate of broadcast seeds on disturbed sites suggests that seed fate depends on the terrain and substrate (Chambers 2000). When seeds of Munro’s globemallow seed were broadcast at a rate of 5.5 viable seeds/ft² (60/m²) at a windy, severely disturbed mining site near Kemmerer, Wyoming, the greatest number of seeds was recovered from artificially constructed large holes (20 in [50 cm] wide and 4 in [10 cm] deep), followed by 0.8-in (2 cm) thick gravel layers. The third highest seed total was recovered from small, artificially constructed, and uniformly spaced holes. Overall emergence of all seeded species was low (<2%) but greatest from large holes. Emergence from small hole and gravel treatments was negligible (Chambers 2000).
Seedling ecology. Studies suggest that Munro’s globemallow seedlings grow quickly under ideal conditions (Parkinson 2008; Parkinson et al. 2013). In a greenhouse, seedling relative growth rate (RGR) averaged 0.13 to 0.15 g/g/day from 14 to 42 days after cotelydons appeared. The RGR of Munro’s globemallow was comparable to that of the invasive species investigated (spotted knapweed [Centaurea stoebe], rush skeletonweed [Chondrilla juncea], Dalmatian toadflax [Linaria dalmatica], whitetop [Cardaria draba], Fuller’s teasel [Dipsacus fullonum], and Scotch cottonthistle [Onopordum acanthium]) with RGRs ranging from 0.09-0.17 g/g/day (James and Drenovsky 2007). In another greenhouse study, the RGR of Munro’s globemallow seedlings was 0.41 mg/mg/week for shoots, 0.53 mg/mg/week for roots, and 0.41 mg/mg/week overall. Seedling root systems were fan-shaped with similar lateral and vertical development and considerable branching at the soil surface and throughout the depth of the container (18 inches [45 cm]). Seedling biomass averaged 2.0 g at 6 weeks, 8.8 g at 9 weeks, and 24 g at 12 weeks. Seedling biomass at 12 weeks was 1 to 3 times that of other native species evaluated (hoary tansyaster [Dieteria canescens], royal penstemon [Penstemon speciosus], sulphur-flower buckwheat [Eriogonum umbellatum], and lomatium [Lomatium spp.]). When grown in the presence of grass seedlings, biomass of Munro’s globemallow was significantly reduced by cheatgrass (Bromus tectorum) (P <0.001) but not by the native grasses, Sandberg’s bluegrass (Poa sandbergii) or squirreltail (Elymus elymoides) (Parkinson 2008; Parkinson et al. 2013).
Warm temperatures (Kildisheva and Davis 2013) and disturbance (Whitcomb 2011) may encourage growth of Munro’s globemallow seedlings. In greenhouse experiments, seedlings grown from wildland seed collected from five Oregon and Idaho locations were less well developed and produced less biomass under cool (63/37 °F [17/3 °C]) than warm (73/48 °F [23/9 °C]) diurnal (8/16 hour) temperatures (Kildisheva and Davis 2013). Seedlings grown under moisture-limited conditions (irrigated every 9 or 12 days) produced greater belowground biomass than seedlings watered every 3 or 6 days (P = 0.03). Seedling mortality was rare under the temperature and moisture regimes tested, suggesting seedlings are relatively resistant to temperature and moisture fluctuations (Kildisheva and Davis 2013). In field experiments, Munro’s globemallow seedling transplants were not affected by increased air and soil temperatures created with warming open-top chambers (Whitcomb 2011). A 2 °F (1.1 °C) increase in air temperature to a daily high of 97 °F (36 °C) with a corresponding 2.5 °F (1.4 °C) increase in soil temperature to a daily high of 88 °F (31 °C) had no significant effect on plant size or biomass. Seedling densities were greater with soil disturbance (created by movement of the warming chamber) and warming. Warmed plants also senesced later than controls. Findings suggested that in restoration project planning, Munro’s globemallow is a good choice for withstanding future temperature increases associated with climate change (Whitcomb 2011).
Seedling growth is improved in nutrient-rich environments, as was found in controlled experiments comparing above- and below-ground biomass of plants grown with and without biological soil crust amendments (Pendleton et al. 2003). Two-week-old seedlings were transplanted into 6-in (15-cm) pots and grown for 70 days in a glasshouse in sand collected near Moab, Utah, sand with an added 1-cm top layer of excised soil crust (from the same location), or in a container filled entirely with crushed crust material. Plants growing with crusts (both treatments) were significantly larger (P < 0.05) than those grown in sand. The fibrous root system was extensive for all seedlings but particularly for those grown entirely in crushed crust (Table 1). Those plants were largest and flowered after 70 days. Available nitrogen, magnesium, and organic matter content were greater in crust topped sand and crushed crust material than in sand alone (Pendleton et al. 2003).
Table 1. Size of Munro’s globemallow plants grown in a glasshouse in the presence or absence of biological crust amendments for 70 days (Pendleton et al. 2003).
|
Growth media |
Height (in) |
Shoot biomass (g) |
Root biomass (g) |
Total biomass (g) |
Root diameter (mm) |
Total root length (ft) |
|
Crushed crust* |
13.2a |
3.91a |
0.86a |
4.77a |
0.23a |
20,105a |
|
Crust topped sand |
1.7b |
0.51b |
0.29b |
0.80b |
0.25a |
8,852b |
|
Sand |
1.1c |
0.30c |
0.25b |
0.55c |
0.21a |
10,686b |
*Only plants grown in crushed crust material flowered after 70 days.
Disturbance Ecology
Munro’s globemallow often occurs on disturbed sites (Pendery and Rumbaugh 1993; Whitcomb 2011). Cover of Munro’s globemallow was greater on burned (2%) than on adjacent unburned sites (0%) 4 years following a fall prescribed fire in mountain big sagebrush on the Uinta National Forest (Goodrich 2006). In defoliation experiments, clipping did not significantly affect Munro’s globemallow biomass or pools of total non-structural carbohydrates (TNC), nitrogen, phosphorus, potassium, calcium, or magnesium. Clipping experiments were conducted in a dryland nursery where Munro’s globemallow and crested wheatgrass seedlings were transplanted in May 1985. Munro’s globemallow seedlings were surrounded by 4 crested wheatgrass (Agropyron cristatum) seedlings and half of the field was clipped to 2 in (5 cm) on May 10, 1986. When values were averaged for the year and all plant parts (roots, crowns, shoots, or whole plants), the carbohydrates and nutrients of clipped and unclipped plants were not significantly different (P < 0.05) (Pendery et al. 1993).
Wildlife And Livestock Use
Reported value of Munro’s globemallow to big game, other wildlife, and livestock ranged from good to none (Hermann 1966). Its importance may depend on the availability of more palatable forage. Ogle et al. (2012) reported that livestock and big game utilize Munro’s globemallow when it is green in early spring and following summer and fall precipitation. At times, deer (Odocoileus spp.) consume all foliage, leaving only the woody base (Pavek et al. 2011). When pronghorn (Antilocapra americana) diets were evaluated over a period of about 1.5 years in salt desert and Wyoming big sagebrush vegetation in southeastern Oregon, Munro’s globemallow made up a high of 6% of spring and 10% of fall diets (McInnis and Vavra 1987).
As forage for domestic cattle and sheep, Munro’s globemallow is considered acceptable but not preferred (Pendery and Rumbaugh 1993). At the US Sheep Experiment Station (USSES) near Dubois, Idaho, Munro’s globemallow production was greater on spring and fall than on fall only sheep use paddocks. At the time of paddock construction, herbage density of Munro’s globemallow was similar across the study area. After 25 years of grazing, production was 0.49 lb/ac (0.56 kg/ha) in fall-grazed and 0.64 lb/ac (0.72 kg/ha) in spring and fall-grazed paddocks (Mueggler 1950).
Munro’s globemallow flowers and seeds are eaten by various birds, small mammals, and lizards (Mayhew 1963; Johnson and Hansen 1979; Pendery and Rumbaugh 1986). Flowers and young leaves are eaten by greater sage-grouse (Centrocercus urophasianus) (Luna et al. 2018) and plants were found at brood-rearing plots in south-central Owyhee County, Idaho (Wik 2002). Over a one-year period, the relative density of Munro’s globemallow recovered from the feces of Nuttall cottontails (Sylvilagus nuttallii) was 9.2% and from bushy-tailed woodrat (Neotoma cinerea) feces was 17.3%. Feces were collected from basin big sagebrush vegetation in south-central Idaho (Johnson and Hansen 1979). Munro’s globemallow flowers were found in the digestive tracts of chuckwalla lizards (Sauromalus obesus), which are considered strict vegetarians (Others cited in Mayhew 1963).
Munro’s globemallow is a nutrient source for many bees, butterflies, and moths including the common checkered-skipper (Burnsius communis, formerly Pyrgus communis) and northern white-skippers (Heliopetes ericetorum) (LBJWC 2023; Oregon Flora 2025). It is also a known larval host for common checkered-skippers, northern white-skippers, Exaeretia thoracefasciella moths, and Zenodoxus palmii chewing moths (James and Nunnallee 2011; Robinson et al. 2024).
In a common garden experimental array, grasshopper use and damage was severe for native grasses but minimal for Munro’s globemallow. Seedlings were planted in a common garden in March 1997 and subjected to natural densities (1.4-1.8/ft² [15-20/m²]) of red-shanked grasshoppers (Xanthippus corallipes) and little pasture spur-throated grasshoppers (Melanoplus confuses). When the cumulative effect of grasshopper herbivory was evaluated in June 1998 (near the end of the grasshopper’s life cycles), about 1% of individual Munro’s globemallow plants were consumed. Grasshoppers consumed more than 70% of individual squirreltail, western wheatgrass (Pascopyrum smithii), and Indian ricegrass (Achnatherum hymenoides) plants and 30% or less of individual Sandberg bluegrass and needle and thread (Hesperostipa comata) plants (Beckstead et al. 2008).
Nutritional Value
Forage characteristics of Munro’s globemallow in new pastures near Kimberly, Idaho (Table 2), met the nutrient requirements for sheep and medium-sized yearling heifers in the fall and spring (Rumbaugh et al. 1993).
Table 2. Nutritional content (evaluated and averaged for fall and spring) of Munro’s globemallow plants growing near Kimberley, ID (Rumbaugh et al. 1993).
|
Measurement |
Microminerals (µg/g) |
Macrominerals mg/g |
||||||||
|
Element |
Cu |
Fe |
Mn |
Na |
Zn |
Ca |
Mg |
N |
P |
K |
|
Leaves |
16 |
1200 |
58 |
150 |
41 |
27 |
6.0 |
37 |
3.6 |
18 |
|
Stems |
14 |
360 |
20 |
280 |
28 |
13 |
5.6 |
21 |
2.3 |
22 |
Clipping 1-year-old transplants to 2 in (5 cm) in a Munro’s globemallow and crested wheatgrass pasture once in spring did not significantly (P < 0.05) affect nutritional content of Munro’s globemallow (Table 3; Pendery et al. 1993).
Table 3. Concentrations of nutrients and elements in Munro’s globemallow shoots (evaluated and averaged for clipped and unclipped plants) for seven sampling dates in 1986 in Cache Valley, UT (Pendery et al. 1993).
|
Date |
May 1 |
May 20 |
Jun 1 |
Jul 11 |
Sept 22 |
Nov 5 |
|
—————mg————— |
||||||
|
TNC* |
52.5 |
62.5 |
72.8 |
74.4 |
84.2 |
58.5 |
|
N |
19.1 |
15.2 |
16.5 |
11.6 |
7.8 |
6.4 |
|
P |
2.6 |
2.3 |
2.6 |
2.2 |
1.1 |
1.1 |
|
K |
11.4 |
15.5 |
21.2 |
20.1 |
13.0 |
10.3 |
|
Ca |
14.1 |
13.9 |
13.5 |
12.8 |
17.0 |
14.8 |
|
Mg |
4.1 |
3.9 |
3.4 |
2.7 |
4.1 |
4.3 |
*Total non-structural carbohydrates.
Ethnobotany
Munro’s globemallow was noted as a plant traditionally used in fertility regulation (Brondegaard 1973 cited in Kumar et al. 2012). The Gosiute of Utah pounded plants into a gummy paste that was used to smooth the rough inner surfaces of dishes (Moerman 2003).
Horticulture
Showy flowers, attractiveness to pollinators, and tolerance of harsh conditions make Munro’s globemallow a desirable species for use in horticulture. It is available commercially for residential native gardens (Meyer et al. 2009) and is hardy to USDA zone 4 (Pavek et al. 2011). At a native plant garden in Boise, Idaho, plants were easily grown from seed. They were watered four times in the first year and once a month in subsequent years (Parkinson 2003).
Revegetation Use
Munro’s globemallow has many traits and characteristics that make it a desirable species for revegetation (Fig. 7). It has a rapid growth rate, tolerates disturbances and harsh environments, occurs on exposed or eroded sites, and is important to a variety of pollinators. It is utilized by wildlife and livestock, providing green forage early in the spring and after summer and fall precipitation (Gautier and Everett 1979; Ogle et al. 2012, 2019; Eldredge et al. 2013). It may also be a good candidate for soil stabilization (Kildisheva et al. 2011).

Figure 7. Second year Munro’s globemallow seeded on a burned site in northern Utah. Photo: USFS.
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for Munro’s globemallow, generalized provisional seed zones developed by Bower et al. (2014) may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (Bower et al. 2014). In Figure 8, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2024) Threat and Resource Mapping (WWETAC) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Selection Tool (Richardson et al. 2020) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
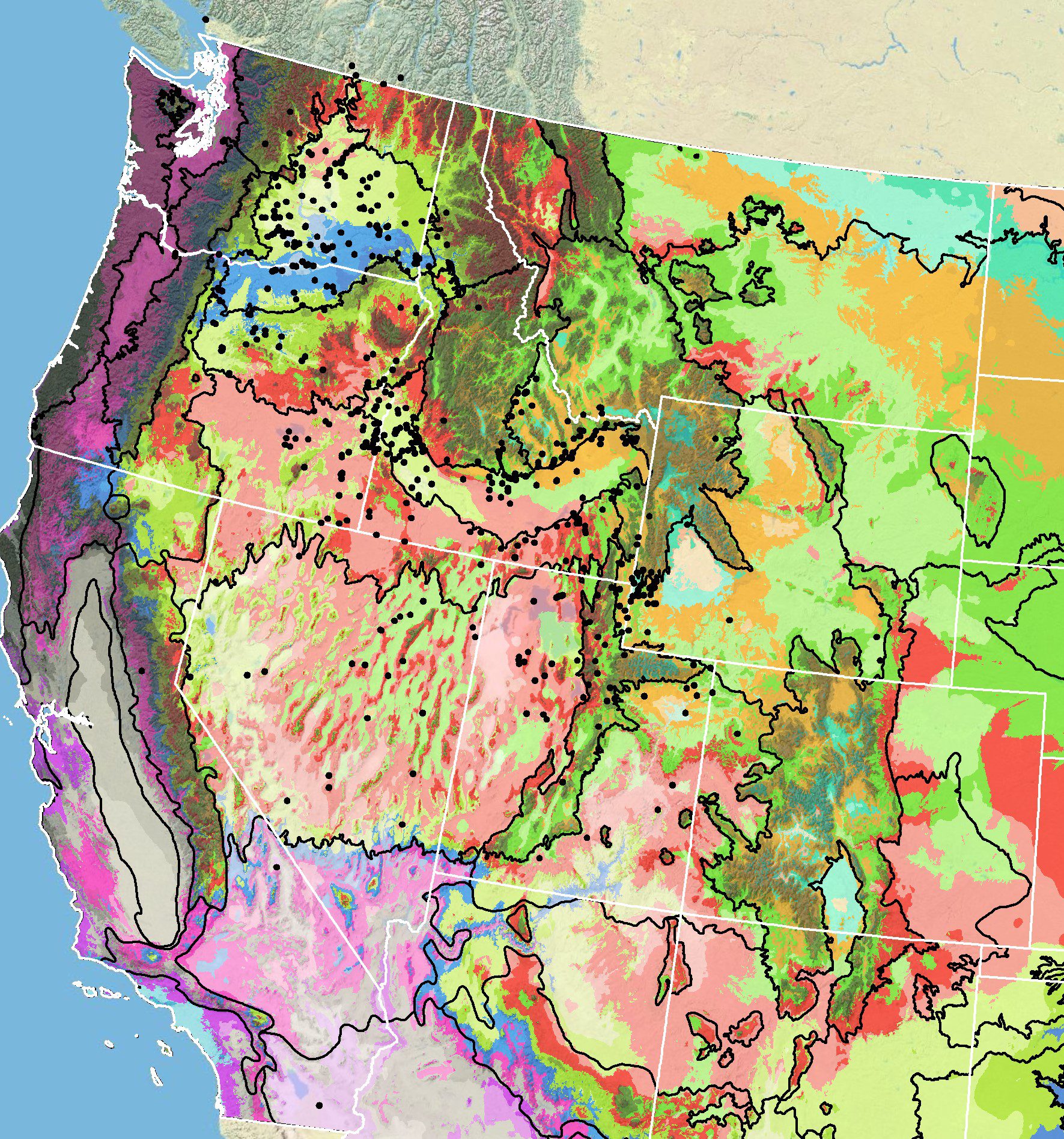
Figure 8. Distribution of Munro’s globemallow (black circles) based on geo-referenced herbarium specimens and observational data from 1886-2016 (CPNWH 2017; SEINet 2017; USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (white outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2024). Map prepared by M. Fisk, USGS.
Releases
Munro’s globemallow, ARS 2892, a Selected Germplasm was released for commercial increase use in restoration (Rumbaugh and Pendery 1993; Stevens et al. 1996). The release was selected because of its large shoot size, succulence, leafy growth, and excellent seed yield potential. It was one of 13 accessions grown and evaluated in non-competitive plant nurseries in northern Utah and southern Idaho from 1987 to 1992 (Table 4). Germplasm ARS 2892 came from wildland seed collected at 4,350 ft (1,325 m) elevation near Hyrum Lake Dam in Cache County, Utah. At this site, annual precipitation averages 16 in (406 mm) and soils are disturbed, sandy, and rocky. The release is tetraploid (2n = 20). It is drought tolerant, heat tolerant, winter hardy, and survives well in semiarid environments (Rumbaugh and Pendery 1993).
Table 4. Growth characteristics of Munro’s globemallow accessions (including Selected Germplasm ARS 2892) grown in non-competitive conditions in northern Utah and southern Idaho (Rumbaugh and Pendery 1993).
|
Attribute |
ARS-2892 |
All other accessions |
|
Survival (%) |
97 |
95 |
|
Plant weight (g) |
188 |
102 |
|
Stems (no/plant) |
22 |
18 |
|
Stem length (in) |
15 |
14 |
|
First flower (Julian day) |
174 |
169 |
|
Schizocarps ripe at harvest (%) |
39 |
33 |
|
Seed weight (g/plant) |
1.1 |
0.7 |
Wildland Seed Collection
Seed can generally be harvested from Munro’s globemallow starting in June and continuing through mid- to late summer (Andersen and Holmgren 1976; Shaw 1995). Seed collection windows, however, vary with site and weather conditions (Holmgren et al. 2005).
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (UCIA 2015; Young et al. 2020). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Munro’s globemallow flowers indeterminately, which suggests a broad collection window. However, length of flowering and seed ripening vary by location. Flowering in southeastern Idaho and southwestern Wyoming was ephemeral with all buds within an inflorescence opening over a short period of time. Duration of flowering was longer for plants growing in locations further south (Holmgren et al. 2005).
Based on 33 recorded collections spanning 10 years in Oregon, Idaho, Nevada, Utah, and Colorado, BLM SOS collection crews reported an earliest harvest date of June 7 and a latest date of September 3 (USDI BLM SOS 2017, 2025). Seed was often harvested in June (11 collections) or July (19 collections). The earliest June harvest was made in 2009 from a 6,098-ft (1,859 m) elevation site in Toole County, Utah. The September harvest was made in 2010, from a 7,650-ft (2,330 m) elevation site in Wasatch County, Utah. Harvests were most commonly made in June (11 collections) or July (20 collections) (USDI BLM SOS 2017, 2025).
Seed collectors for the Great Basin Native Plant Project (GBNPP) suggest that hand harvests can be maximized by collecting when at least 40% of the schizocarps are open or have begun to split and schizocarps near the top of the inflorescence are still greenish and have not split. Plants will be gray-green and have few to no flowers remaining at this time (A. Malcomb, Upper Snake River Tribes Foundation [USRT] and M. Fisk, USGS, personal communications, August 2018). Pendery and Rumbaugh (1993) suggest that seed yield of individual plants is at a maximum when about 25% of the schizocarps are ripe, light green to brown, and just about to open. At this time, the lowest schizocarps (commonly referred to as globes) have started to split. These differences in collection timing may relate to collection methods (hand vs. mechanical). Additional seed ripening that may occur following harvest was not reported.
Collection Methods
Wildland seed was harvested by GBNPP collectors by wrapping a gloved hand gently around the inflorescence and pulling upward. Munro’s globemallow is often covered with fine irritating hairs, making skin and eye protection important. Ripe schizocarps will strip off easily and can be deposited into a paper bag. Bug-repellent tabs in the collection bags can prevent continued seed damage, as schizocarps can harbor seed-feeding insects. Because flowering and seed maturation are indeterminate, collections can be made from the same plants and populations multiple times to maximize ripe seed harvests and genetic diversity. Munro’s globemallow plants that have senesced almost fully and retain an abundance of unopened schizocarps should be avoided. This is a sign of seed damage. To maximize collection efforts, check that full seeds are being harvested (A. Malcomb, USRT; M. Fisk, USGS, personal communications, August 2018).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2023).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2023). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of Munro’s globemallow.
Post-Collection Management
Seed should be kept in a dry, shaded place until collections can be moved to a controlled short-term storage environment. Short-term storage should be dry, cool, and inaccessible to rodents or other seed predators. If insects are suspected in any collection, seed should be frozen for 48 hours or treated with an appropriate insecticide. The more plant material in the collection, the more ventilation and drying a seed lot will likely need (Gold n.d.; Parkinson and DeBolt 2005; Hay and Probert 2011).
Seed Cleaning
Dry seed can be cleaned using a debearder to remove seeds from the schizocarps. If necessary, a clipper or fanning mill can be used to further clean the seed (Pendery and Rumbaugh 1993). For agricultural seed fields in Utah, seed was collected by windrowing and combining plants. Once dried, the lot was processed through a debearder and then an air-screen separator and gravity table (Stevens et al. 1996). Protective clothing, masks, and eyewear are recommended when handling Munro’s globemallow seed collections (A. Malcomb, USRT; M. Fisk, USGS, personal communications, August 2018).
The USFS Bend Seed Extractory developed detailed cleaning procedures for a small seed lot (1.6 lbs [0.7 kg]) of wildland collected seed. Dry seed was first processed using a Westrup Model LA-H, (Seedburo Equipment Co., Des Plaines, IL), laboratory brush machine (Seedburo Equipment Co., Des Plaines, IL), using a #28 mantel and medium speed. Seed was then air-screened using an office Clipper with a no. 6 round top screen, no. 1/20 bottom screen, and medium speed and air (Barner 2009).
Seed Storage
Munro’s globemallow seed is orthodox and reports suggest it can be stored dry for 6 to 15 years without substantial loss of viability (Pendery and Rumbaugh 1986; Stevens et al. 1996). General storage guidelines for native seed suggest storage at or below 15% relative humidity at 59 °F (15 °C) for less than 5 years of storage and at -0.4 °F (-18 °C) for more than 5 years of storage (Erickson and Merritt 2016). Seed was stored cold at the Bend Seed Extractory (33-38 °F [0.6-3.3 °C]) (Barner 2009), but longevity in storage was not reported. Because seed is attractive to insects and small mammals, it should be stored in a protected area and frozen or insecticide treated as necessary (Pendery and Rumbaugh 1986).
Seed Testing
There is an Association of Official Seed Analysts (AOSA) tetrazolium (TZ) viability testing guideline for globemallow species (AOSA 2010) but no germination testing rule (AOSA 2023).
Because some seeds may be hard, they are prepared for treatment by clipping a small piece of the seed coat near the distal end of the cotyledons (AOSA 2010). Clipped seeds are imbibed on moist media overnight at 68 to 77 °F (20-25 °C). Imbibed seeds are prepared for staining by either cutting them deeply, leaving the two halves together and soaking them in 0.1% TZ overnight at 95 °F (35 °C) or by extracting the embryos and soaking them in 1.0% TZ overnight at 95 °F (35 °C). Seeds are counted as viable if the entire embryo and endosperm are evenly stained.
Germination Biology
Munro’s globemallow seed exhibits physical dormancy. Dormant seeds are unable to take up water, a critical step in the germination process, due to the presence of an impermeable seed coat and a cap-like structure, which also blocks water uptake through the hilum (a pore). Dislodging this structure or abrasion of the seed coat through natural or artificial scarification is necessary to enable germination of fresh or cold-stored seeds (Kildisheva et al. 2011).
In wildland settings, scarification likely occurs naturally over time and may represent an evolutionary survival tactic for the species. For restoration projects, rapid and uniform germination is often desired. Many have researched the mechanisms and methods for improving germination of Munro’s globemallow. Mechanical scarification that detached the water restrictive plug in the hilum with a scalpel was best for seed germination, but it is not a feasible large-scale seed treatment (Kildisheva et al. 2011). Boiling water scarification (Kildisheva et al. 2013) and pneumatic scarification using a Mater seed scarifier (Mater Seed Equipment, Corvallis, OR) (Kildisheva et al. 2018) worked well as potential large-scale seed treatments. The variation in the extent of germination across studies may suggest a high degree of variation in the depth of dormancy across seed lots, which may be affected by population genetics, growing season, collecting, cleaning, or storage conditions (Kildisheva et al. 2011).
Water uptake was improved by abrading the seed coat with a scalpel more than by submerging seed in boiling water (10 s in 212 °F [100 °C]) (Kildisheva et al. 2011). For fresh seed collected near Payette, Idaho, seed mass increased 86% following water uptake after seeds were cut and increased 22% following boiling water treatments (Kildisheva et al. 2011). For cold-stored seed (6 mos at 35 °F [1.5 °C]) collected from the Wasatch Mountains of northern Utah, germination of mechanically scarified seeds on blotter paper was 87%, and increased to 93.4% when scarified seeds were hydro-primed (soaked in aerated deionized water) for 24 hours (Kildisheva et al. 2011). For seeds collected in west-central Idaho, submergence in sulfuric acid did not significantly increase germination (P > 0.05) (Roth et al. 1987). Soaking seeds in dioxane produced the highest germination levels (up to 56%) when compared to sulfuric acid and sulfuric acid plus dioxane treatments. However, dioxane is a highly flammable, cancer causing agent that is not readily available and is not recommended for use.
In additional studies, boiling water, tumbling, burning, heating, and burning with heating were evaluated as potential methods to increase germination of Munro’s globemallow seed on a large scale (Table 5; Kildisheva et al. 2013). Germination was best (49%) after boiling water treatments. The other treatments produced germination of 20% or less. Treatments were conducted on cold-stored seeds (6 mos at 35 °F [1.5 °C]) collected from the Wasatch Mountains of Utah, and germination was monitored for 21 days. For boiled seeds, the germination curve failed to reach its asymptote after 21 days, suggesting that with continued monitoring germination may have continued at a low rate (Kildisheva et al. 2013).
Table 5. Germination of cold-stored Munro’s globemallow seed at 21 days following various scarification treatments. Post-treatment germination conditions: incubation at 75 °F (24 °C) day/63 °F (17 °C) night (Kildisheva et al. 2013).
|
Treatment |
Germination (%) |
|
Control |
10.7 |
|
Boiling (10 s in 212 °F water) |
49.0 |
|
Tumbling (72 hrs in rotary rock tumbler with dry Al2O3 grit) |
20.3 |
|
Burning (1 min alcohol soak then 10 s burning with butane torch) |
17.0 |
|
Heating (176 °F oven for 60 min) |
10.7 |
|
Burning + Heating (combination of above) |
4.2 |
In an attempt to find and mimic natural seed scarification through abrasion, temperature fluctuations, and burning, Kildisheva et al. (2018) evaluated the following seed treatments: wet heat (5-300 s in 194 °F [90 °C] water), freezing + wet heat (2 hrs at -112 or -4 °F [-80 or -20 °C] then 5 or 30 s in 194 °F [90 °C] water), freeze-thaw cycle (2 hrs at -112 or -4 °F [-80 or -20 °C] then 2 hrs at 73 °F [23 °C] repeated 1-6 times), pneumatic scarification (10-160 s in a Mater seed scarifier), and manual scarification (120-grit sand paper). Seed permeability was increased by wet heat (90-94%), pneumatic scarification for 80 or more seconds (82-88%), and manual scarification (88%). Germination percentages were viability adjusted and included a cut-test to assess embryo condition and final seed viability. Germination was tested only for mechanically scarified seeds and was highest (>75%) with manual scarification and reached about 50% with 80 or 160 seconds of pneumatic scarification. Researchers advised testing the effects of a longer pneumatic scarification period on germination, because this type of treatment is feasible for use in treating large seed lots. Because the loss of physical dormancy is irreversible, seeds could be treated and stored in a “restoration-ready” state (Kildisheva et al. 2018).
Kildisheva et al. (2019) found that exposure to gibberellic acid (GA3) and karrikinolide (KAR1) resulted in slower germination than seed exposure to water regardless of temperature. The Munro’s globemallow seed tested was wild collected at an elevation of 2,598 ft (792 m) from Succor Creek in eastern Oregon. It was dried at 41 °F (15 °C) and 15% relative humidity for 3 to 4 weeks and kept at these conditions for up to 3 months prior to testing. Seed fill averaged 94%, and for untreated seed germination was quicker at 50 to 77 °F (10-25 °C) than at 41 °F (5 °C) (Kildisheva et al. 2019).
Wildland Seed Yield And Quality
Post-cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 6 (USFS BSE 2017, 2023). The results indicate that Munro’s globemallow seed can generally be cleaned to high levels of purity but that seed fill and viability are variable. The seed weights reported by others (USFS GBNPP 2014; SER, INSR, RBGK, SID 2023) fall within the range reported in Table 6.
Table 6. Seed yield and quality of Munro’s globemallow seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Seed lot characteristic |
Mean |
Range |
Samples (no.) |
|
Bulk weight (lb) |
2.09 |
0.05-25 |
32 |
|
Clean weight (lb) |
0.33 |
0.0-3.36 |
32 |
|
Clean-out ratio |
0.24 |
0.0-0.74 |
32 |
|
Purity (%) |
97 |
80-99 |
31 |
|
Fill (%)¹ |
91 |
65-99 |
31 |
|
Viability (%)² |
89 |
65-98 |
19 |
|
Seeds/lb |
347,430 |
249,088-623,700 |
31 |
|
Pure live seeds/lb |
268,825 |
187,597-393,981 |
19 |
¹100 seed X-ray test
²Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well.
For Munro’s globemallow seed, acceptable viability is reported as 90% and purity 80% or higher if vendors use a gravity table in the cleaning process (Stevens et al. 1996). Germination can be expected to be erratic due to dormant seed (Stevens et al. 1996). In 2010, Munro’s globemallow seed averaged $57/lb. This cost was a standardized market price based on end-user market price and suggested Plant Materials Center (PMC) stock seed prices (Young and Bouck 2011).
Agricultural Seed Production
Researchers growing Munro’s globemallow in Cache Valley, Utah, reported it was more easily grown in an agronomic environment than many other wildland forbs (Fig. 9) (Pendery and Rumbaugh 1986).

Figure 9. Munro’s globemallow seed production field growing near Willow Creek, OR. Photo: USFS.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (UCIA 2015; Young et al. 2020).
Seed growers should apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Site Preparation
Weed-free planting sites are recommended for Munro’s globemallow seed production plots (Stevens et al. 1996).
Seed Pretreatments
Mechanical seed treatments can be used to improve the amount and uniformity of germination (see Germination Biology section). Stratification (to remove physiological dormancy) is not required as seed dormancy is entirely physical. Submergence in boiling water can be an effective pre-sowing treatment for nursery production (Jensen 2011 cited in Pavek et al. 2011). In establishment of seed production plots at the USFS Provo Shrub Sciences Laboratory (PSSL), seeds were submerged in boiling water for 1 minute, rinsed with cool water, then dried prior to seeding. This pretreatment at times resulted in high seedling emergence following fall or spring planting but at other times resulted in almost no emergence. Rapid and thorough drying of the boiled seed was critical because seed that remained moist often germinated within days (S. Jensen, USFS, personal communication, July 2018). Cool moist stratification for 90 days failed to improve germination of Munro’s globemallow at the Pullman PMC. Seed from three accessions was planted in containers in mid-October and stratified outdoors for 90 days. Germination began within 14 days of moving containers into the greenhouse, but germination was low (6-25%) (Pavek et al. 2011).
Weed Management
Weed control by hand weeding is recommended, although cultivation can also be utilized (Stevens et al. 1996). Herbicides are not registered for this species, and results or findings presented here do not constitute an endorsement of specific products or recommendations for use. In research plots in Provo, Utah, Munro’s globemallow emergence was nearly equal with pre-emergent herbicide treatments of bromoxynill, pendimethalin, and imazethapyr at all rates tested and to the ammonium salt of imazapic at medium and low application rates (Roundy et al. 2005). Comparisons with no herbicide treatment were not provided.
Seeding
Fall planting of non-scarified Munro’s globemallow seed is recommended. Spring or late summer seeding is possible if seed has been scarified (see Germination Biology section) but may delay seed production beyond the second year. A seeding rate of 20 to 40 PLS/linear ft (67-133/m) of row with 28 to 36-in (71-91 cm) row spacing and seeding depths of 0.13 to 0.25 in (0.3-0.6 cm) in sandy loam to clay-textured soils is recommended for seed production stands (Stevens et al. 1996).
Establishment And Growth
Munro’s globemallow seed is eaten by birds and rabbits. Fields may need protection from predation in the establishment and early growth phases (Pendery and Rumbaugh 1986). Seed is not produced until the second year, reaching 50 to 75% of full yield. By the third growing season, seed production reaches full yield (Stevens et al. 1996). According to a private seed producer, Munro’s globemallow establishment is easy, but due to the physical dormancy of seed (hard seed coats), seedling emergence may span several years if the seeds are not scarified (Dunne cited in Pavek et al. 2011).
Irrigation
Stands do well with 11 to 14 in (280-360 mm) of annual precipitation. Irrigation is recommended as needed for seedling establishment and again in the spring and during the flowering and seed-set period (Stevens et al. 1996).
Pollinator Management
Outcrossing increases Munro’s globemallow seed production. In one study, seed production was increased four times when flowers were outcrossed. When flower visits were monitored, 37 of 57 bee visitors to flowers of Munro’s globemallow (and relatives) were ground-nesting specialists including globemallow bees (Diadasia spp.) and plasterer bees (Colletidae). The other 20 bee visitors were generalist ground-nesters. To protect pollinators, cultivation should be avoided in seed production plots when plants are blooming (Cane cited in Pavek et al. 2011).
Pest Management
Munro’s globemallow is a host species for the following fungi: Puccinia malvacearum, P. schedonnardi, and P. sherardiana (Farr and Rossman 2025). Planthoppers (Oecleus glochin) were collected from plants in Nevada and Utah (Kramer 1977). The degree to which these pests may impact seed production plots was not reported.
Seed Harvesting
Indeterminate flowering and seed ripening make mechanical seed harvest challenging for Munro’s globemallow. With summer precipitation, plants remain green, flowering continues throughout the growing season, and seeds shatter soon after they have ripened. In areas lacking summer precipitation, it is possible to manipulate flowering and seed ripening by withholding water early in the growing season (Dunne cited in Pavek et al. 2011). In Utah, fields were harvested between early July and early August by windrowing plants when lower schizocarps were dry and had started to open. Windrows were harvested by combining (Stevens et al. 1996).
Seed Yields And Stand Life
Munro’s globemallow will produce seed in the second growing season but it generally takes 3 years to reach maximum yields (Stevens et al. 1996), which can range annually from 300 to 1,400 lbs/ac (340-1,580 kg/ha) (Stevens et al. 1996; Anderson et al. 2007). Longevity of seed production fields can be 3 to 5 years (Stevens et al. 1996). Seed production may be increased with fertilization and by clipping plants after seed set (Gautier and Everett 1979).
In research plots growing in Provo, Utah, Munro’s globemallow seed production on a per plant basis was greatest from plants spaced 18 in (46 cm) apart (22.3-31 g of seed/plant), but production on a per acre basis was greatest for plants spaced 6 in (15 cm) apart (1,000-1,400 lb of seed/ac [1,125-1,575 kg/ha]) (Roundy et al. 2005; Anderson et al. 2007). Fields with plastic mulch to control weed competition produced the greatest amount of seed. Production was less with fabric mulch, even less without mulch, and least with paper mulch (Roundy et al. 2005). When fields were at least 3 years old, seed yield was a little less than 900 lbs/ac (1,010 kg/ha) at 6-in (15 cm) between row spacing. When fields were at least 4 years old, seed yield was less than 200 lbs/ac (225 kg/ha) and stand mortality was substantial. Seed collection methods also differed between the high and low yield years. Seed was harvested by hand over an extended period in the high yield year and once mechanically in the low yield year (Anderson et al. 2007).
In experiments on container-grown Munro’s globemallow, fertilizing and clipping stems and leaves to a 2-in (5-cm) height increased seed production, but timing of the treatments was important (Table 7, Gautier and Everett 1979). Schizocarp production was greatest when clipping occurred after seed ripening and fertilization occurred at the early flowering stage and again after clipping. Flowering period was extended when clipping occurred after seed ripening, and clipped plants continued growing until the first hard frost. Clipping at the early flowering stage resulted in significantly lower seed production (P < 0.05). Unclipped plants produced mature seed in late July and then stopped growing. For this study, treatments and harvests occurred in the same year. Seed came from a single site, and plants were grown in low nitrogen, low phosphorus, loamy sandy soils in a greenhouse for 3 months before being transferred to a lathhouse (Gautier and Everett 1979).
Table 7. Schizocarp and biomass production of container-grown Munro’s globemallow. Fertilizer treatments were 89 lbs/ac of nitrogen and 45 lbs/ac phosphorus. Values within a column followed by different letters are significantly different (P < 0.05). (Gautier and Everett 1979).
|
Treatment |
Schizocarps (no./16 plants) |
Biomass of foliage and stems (g) |
|
Unclipped, unfertilized |
37.5bc |
3.0c |
|
Unclipped, fertilized at early flowering stage |
53.5ab |
8.3bc |
|
Clipped at early flowering, unfertilized |
0.8d |
4.6c |
|
Clipped and fertilized at early flowering |
2.3d |
25.0a |
|
Clipped at early flowering, fertilized after clipping |
15.6cd |
16.4b |
|
Clipped and fertilized at early flowering, fertilized after clipping |
9.5cd |
36.6a |
|
Clipped after seed set, fertilized after clipping |
24.3bcd |
14.0bc |
|
Clipped after seed ripening, fertilized at early flowering and after clipping |
76.5a |
28.8a |
Nursery Practice
Munro’s globemallow was grown successfully in greenhouse experiments designed to monitor growth, resource use, and biomass allocation (see Seed and Seedling Ecology section). Seed was germinated on moist blotter paper in a growth chamber at 75 °F (24 °C) with 14 hr/10 hr light/dark cycle. Seedlings were transplanted into containers and grown in the greenhouse at 64 to 75 °F (18-24 °C) with 14 hours of light. Seedling roots were cone-shaped, distributed as much laterally as vertically with considerable branching at the soil surface and throughout the depth of the container (18 in [45 cm]) (Parkinson 2008; Parkinson et al. 2013).
In a study evaluating growth in high and low nitrogen and water conditions, biomass and RGR of Munro’s globemallow were greatest in high nitrogen and high water treatments and root mass ratio was greatest in low nitrogen and low water treatments (Drenovsky et al. 2012). Plants were grown in a 1:2 fritted clay: sandy loam mix. Two weeks following development of true leaves, plants were provided 2 g of slow release NPK 10-10-10 fertilizer or no fertilizer, and 6 weeks after true leaves appeared, plants were subjected to drought or well-watered treatments. Munro’s globemallow growth differences and biomass allocation were not significantly different between the treatments (Drenovsky et al. 2012). When a companion study evaluated the distribution of fertilizers in the soil profile, researchers concluded that Munro’s globemallow had biomass and root foraging traits similar to the invasive species evaluated (whitetop, spotted knapweed, rush skeletonweed, and Dalmatian toadflax) (Drenovsky et al. 2008).
Wildland Seeding And Planting
Although Munro’s globemallow has been seeded and planted successfully in semi-arid western environments, its use has been limited in restoration of public lands in the Great Basin and Colorado Plateau. Based on data from Land Treatment Digital Library (accessed in May 2015), Munro’s globemallow was not seeded in the Great Basin before 2000, but it was seeded 17 times between 2000 and 2009 and two times from 2010 to 2013. These instances of use come from a dataset representing 1,881 aerial seeding and 2,478 drill seeding treatments (Pilliod et al. 2017). In a similar study, data from the Utah Watershed Restoration Initiative, the Land Treatment Digital Library, as well as project data from the Bureau of Land Management and the National Park Service were used to develop the Colorado Plateau Restoration outcomes database. In the compiled data, Munro’s globemallow appeared in only two seeding mixes used at a total of 194 sites between 1999 and 2014 (Balazs et al. 2020).
In sites where globemallow species were seeded and establishment was monitored, recruitment occurred but seedlings failed to produce as much biomass as other forbs on fertile sites or sites receiving more than 12 in (305 mm) annual precipitation. Globemallows have been established on harsher, disturbed, or lower precipitation sites making this species useful for sites with a high incidence of heat and drought stress (Pendery and Rumbaugh 1993; Ogle et al. 2012).
Seeding. Dormant fall seeding is recommended (Ogle et al. 2012). Seeds can be scarified to encourage high initial germination rates (Pavek et al. 2011) or for success with spring seeding (S. Jensen, USFS, personal communication, July 2018). In wildland settings, Munro’s globemallow occurs at low to moderate densities and relies heavily on the seedbank for persistence. Because of this, seeding methods that add a dormant portion of seed to the seedbank, may benefit long-term persistence (Kildisheva et al. 2013).
Seeds should not be planted deeper than 0.25 in (0.6 cm) (Ogle et al. 2012). Recommended pure stand seeding rates for globemallows range from 0.25 to 2 lbs/ac (0.28-2.25 kg/ha) considering the desired stand composition. This species is not recommended for pure stands (Pendery and Rumbaugh 1993; Ogle et al. 2012).
A mid-April planting of Munro’s globemallow seed failed at a Curlew Valley site in southern Idaho but not at a cooler, wetter Cache Valley site in northern Utah, supporting the overall recommendation for fall seeding (Pendery and Rumbaugh 1990). Researchers suggested insufficient moisture, annual weed competition, and soil crusting as potential reasons for failed seedling establishment in southern Idaho. Seed was scarified with sandpaper and seeded in a 3:1 crested wheatgrass: Munro’s globemallow mix at a rate of 5 PLS lbs/ac (5.6 kg/ha). At the Cache Valley site, Munro’s globemallow established before alfalfa or crested wheatgrass (alfalfa was also seeded 3:1 with crested wheatgrass). Density of Munro’s globemallow dropped dramatically after the first year and average yield for 4 years after seeding was 76 g/m², compared to 389 g/m² for alfalfa. Gooseberryleaf globemallow was also seeded at the 3:1 ratio with crested wheatgrass, its 4-year yield average was 48 g/m². Plots seeded with gooseberryleaf globemallow had fewer relatively large plants while those seeded with Munro’s globemallow had more relatively small plants (Pendery and Rumbaugh 1990).
Wildland restoration examples. Researchers evaluating the effects of seeding treatments and environmental factors for 20 native forb species for two years (2013 and 2014) at three sites (Wells and Orovada, Nevada, and Fountain Green, Utah) suggest Munro’s globemallow is a good candidate for direct seeding (Jensen et al. 2022). Treatments combinations were seeding depth (1.4, 2.6, 3.6 or 4 cm) and row cover (present or absent). Munro’s globemallow emergence was low overall, just 0.4% for all treatments and study areas. Row cover improved emergence at Fountain Green in 2013, where emergence was 99% lower in control than in row cover plots (P<0.0001). Emergence was improved with increasing seeding depth by 8.7% in 2013 and 86.9% in 2014 at Fountain Green and by 123.6% in 2014 at Orovada (Jensen et al. 2022).
In a study evaluating strategies for increasing diversity in crested wheatgrass stands, Munro’s globemallow was the most abundant forb 3 to 5 years after seeding glyphosate-treated crested wheatgrass stands. Munro’s globemallow was seeded as part of a mixture at a rate of 5 PLS lbs/ac (0.56 kg/ha) with a minimum till drill on sandy loam soils southeast of Elko, Nevada (McAdoo et al. 2017).
In another study attempting to establish native species in crested wheatgrass stands, Munro’s globemallow established at two sites, but survival to post-seeding year 2 or 3 was limited to one site (Hulet et al. 2010). Munro’s globemallow was drill seeded at a rate of 5 PLS lbs/ac (0.56 kg/ha) in a mixture in October 2005 and 2006. Seeding occurred in disked, double disked, or herbicide-treated crested wheatgrass stands in Toole County, Utah. Loss of Munro’s globemallow from one site and decreases at the other were likely due to precipitation that was 51% below average in 2008 (Hulet et al. 2010).
In a post-fire restoration study, Ott et al. (Ott et al. 2015, 2016) found that Munro’s globemallow density increased slightly as time since seeding increased from year 1 to year 2 and that drill seeding method (Kemmerer Rangeland or Roughrider Minimum Till) had little effect on establishment and survival, which varied by site. Seed collected from Uintah County, Utah, at 5,085 ft (1,550 m) was seeded in fall or winter following a June 2010 fire in Wyoming big sagebrush in Elmore County, Idaho, at 3,940 ft (1,200 m). Munro’s globemallow was at a rate of 3.6 PLS/ft² (40/m²) within a seed mixture. Density of Munro’s globemallow in the seeded area was 282 plants/ac (705/ha) in 2011 and 624 plants/ac (1,560/ha) in 2013. Because a portion of the restoration area burned again in July 2012, burned and unburned areas were monitored separately in 2013. There were 1,305 Munro’s globemallow plants in burned and 279 plants in unburned areas (Fig. 10) (Ott et al. 2015, 2016).

Figure 10. Munro’s globemallow growing in a post-fire restoration seeding in Elmore County, Idaho. Photo: USFS.
Greenhouse studies evaluating the growth rate of Munro’s globemallow suggest it may be competitive with invasive species (Parkinson 2008; Parkinson et al. 2013). See Seed and Seedling Ecology section for details.
Planting. Use of transplants in restoration generally results in more rapid maturity and greater survival than seeding. Munro’s globemallow seedlings can be produced by planting scarified seeds in the greenhouse in January. Plants need 2 to 4 weeks of hardening before transplanting in the field. Recommended spacing between transplants is 10 to 30 in (25-75 cm) (Pavek et al. 2011). In field experiments at Utah State University’s Green Canyon Research Area, 16% of Munro’s globemallow transplants flowered in their transplant year. Seedlings originating from direct fall seeding failed to flower in their first growing season (Whitcomb 2011).
Average survival was high for Munro’s globemallow (95%) a year after transplanting in dryland nurseries in Cache Valley, northern Utah and Curlew Valley, southern Idaho. Transplants were grown from sand paper-scarified seed in 9 in³ (150 cm³) cones filled with four parts sand, two parts peat moss, and one part vermiculite. Transplanting occurred in clean-tilled sites in mid-April 1987. In summer 1988, transplants weighed an average of 102 g (range: 83-183 g), produced an average of 18 stems (range: 16-19) with maximum length averaging 14 in (35 cm) (range: 10-17 in [25-44 cm]). Plant biomass was greater, plants flowered earlier, and seed production was greater at Cache Valley, where annual precipitation from 1985 to 1988 was 2 to 16 in (59-405 mm) greater than at Curlew Valley (average ppt: 12.6 in [321 mm]) (Pendery and Rumbaugh 1990).
Koutzoukis et al. (2023) monitored survival of transplanted seedlings for up to 4 years as planting distance varied (50-250 cm) from Wyoming big sagebrush canopies (Table 9). Survival was highest and significantly greater at 150 cm from the middle of the shrub canopy than at greater and nearer distances (P<0.05). Seedlings were planted at many Great Basin and Columbia Plateau sites with Wyoming big sagebrush. They were planted in fall of 2018 and 2019. Seedlings in the 2019 cohort experienced at much drier first year than the 2018 cohort. Survival of Munro’s globemallow seedlings planted in 2018, showed positive associations with higher biological soil crust and rock cover and negative associations with litter cover (Koutzoukis et al. 2023).
Table 9. Percent survival of two cohorts of Munro’s globemallow seedling transplants at several locations in the Great Basin and Colorado Plateau (Koutzoukis et al. 2023).
| Cohort | Year 1 | Year 2 | Year 3 | Year 4 |
| 2018 | 29.15 | 23.35 | 14.13 | 6.16 |
| 2019 | 23.36 | 10.55 | 2.25 | no data |
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Olga Kildisheva, University of Western Australia and Matt Fisk, USGS.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Andersen, B.A.; Holmgren, A.H. 1976. Mountain plants of northeastern Utah. Circular No. 319. Logan, UT: Utah State University Extension Services. 148 p.
Anderson, V.; Johnson, R.; Roundy, B. 2007. Agronomic and cultural care of wildland plants and seed yield losses in forbs (Asteraceae) due to fruit flies (Tephritidae). In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2006 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 53-59.
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Association of Official Seed Analysts [AOSA]. 2023. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Balazs, K.R.; Kramer, A.T.; Munson, S.M.; Talkington, N.; Still, S.; Butterfield, B.J. 2020. The right trait in the right place at the right time: Matching traits to environment improves restoration outcomes. Ecological Applications. 30(4): 1-7, e2110.
Barner, J. 2009. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Sphaeralcea munroana (Douglas) Spach. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2018 June 13].
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Beckstead, J.; Meyer, S.E.; Augsperger, C.K. 2008. The indirect effects of cheatgrass invasion: Grasshopper herbivory on native grasses determined by neighboring cheatgrass. In: Kitchen, S.G.; Pendleton, R.L.; Monaco, T.A.; Vernon, J., comps. Proceedings-Shrublands under fire: Disturbance and recovery in a changing world; 2006 June 6-8; Cedar City, UT. Proc. RMRS-P-52. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 41-48.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Cane, J.H. 2009. Pollination and seed predator studies. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2008 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 62-65.
Cane, J.H.; Love, B. 2016. Floral guilds of bees in sagebrush steppe: Comparing bee usage of wildflowers available for postfire restoration. Natural Areas Journal. 36(4): 377-391.
Chambers, J.C. 2000. Seed movements and seedling fates in disturbed sagebrush steppe ecosystems: Implications for restoration. Ecological Applications. 10(5): 1400-1413.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Drenovsky, R.E.; Khasanova, A.; James, J.J. 2012. Trait convergence and plasticity among native and invasive species in resource-poor environments. American Journal of Botany. 99(4): 629-639.
Drenovsky, R.E.; Martin, C.E.; Falasco, M.R.; James, J.J. 2008. Variation in resource acquisition and utilization traits between native and invasive perennial forbs. American Journal of Botany. 95(6): 681-687.
Eldredge, E.; Novak-Echenique, P.; Heater, T.; Mulder, A.; Jasmine, J. 2013. Plants for pollinator habitat in Nevada. Tech. Note NV 57. Reno, NV: U.S. Department of Agriculture, Natural Resources Conservation Service. 65 p.
Erickson, T.E.; Merritt, D.J. 2016. Seed collection, cleaning, and storage procedures. In: Erickson, T.E.; Todd, E.; Barrett, R.L.; Merritt, D.J.; Dixon, K.W., eds. Pilbara seed atlas and field guide: Plant restoration in Australia’s arid northwest. Dickson, Australian Capital Territory: CSIRO Publishing: 7-16.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Farr, D.F.; Rossman, A.Y. 2025. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://fungi.ars.usda.gov/
Gautier, C.; Everett, R. 1979. Fertilizing and clipping effects on seed capsule and forage production of orange globemallow. Ogden, UT: Res. Note INT-251. U.S. Department of Agriculture, Forest Service. 6 p.
Gold, K. n.d. Post-harvest handling of seed collections. Technical Information Sheet 04. UK: Royal Botanic Gardens Kew and Millennium Seed Bank Partnership. 4 p.
Goodrich, S. 2008. Trout Creek 1999 burn. In: Kitchen, S.G.; Pendleton, R.L.; Monaco, T.A.; Vernon, J., comps. Proceedings – Shrublands under fire: Disturbance and recovery in a changing world. 2006 June 6-8; Cedar City, UT. Proc. RMRS-P-52. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 147-150.
Hay F.R.; Probert, R.J. 2011. Chapter 20: Collecting and handling seeds in the field. In: Guarino, L.; Ramanatha, V.; Goldberg, E. Collecting plant genetic diversity: Technical Guidelines-2011 update. Rome, Italy: Bioversity International. 33 p.
Hermann, F. 1966. Notes on western range forbs: Cruciferae through Compositae. Agric. Handb. 293. Washington, DC: U.S. Department of Agriculture, Forest Service. 365 p.
Hitchcock, C.L.; Cronquist, A. 2018. Flora of the Pacific Northwest: An illustrated manual. Second Ed. Giblin, D.E.; Legler, B.S.; Zika, P.F.; Olmstead, R.G., eds. Seattle, WA: University of Washington Press. 882 p.
Holmgren, N.H.; Holmgren, P.K.; Cronquist, A. 2005. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Volume Two, Part B: Subclass Dilleniidae. Bronx, NY: New York Botanical Garden Press. 488 p.
Hulet, A.; Roundy, B.A.; Jessop, B. 2010. Crested wheatgrass control and native plant establishment in Utah. Rangeland Ecology and Management. 63(4): 450-460.
James, D.G.; Nunnallee, D. 2011. Life histories of Cascadia butterflies. Corvallis, OR: Oregon State University Press. 447 p.
James, J.J.; Drenovsky, R.E. 2007. A basis for relative growth rate differences between native and invasive forb seedlings. Rangeland Ecology and Management. 60(4): 395-400.
Johnson, M.K.; Hansen, R.M. 1979. Foods of cottontails and woodrats in south-central Idaho. Journal of Mammalogy. 60(1): 213-215.
Kearney, T.H. 1935. The North American species of Sphaeralcea subgenus Eusphaeralcea. Berkeley, CA: University of California Press. 127 p.
Kildisheva, O.A.; Davis, A.S. 2012. Restoration potentials and challenges for Sphaeralcea munroana. In: Haase, D.L.; Pinto, J.R.; Riley, L.E., eds. National proceedings: Forest and Conservation Nursery Associations-2011; 2011 July 25-28, Huntington, WV. RMRS-P-68. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 65-71.
Kildisheva, O.A.; Davis, A.S. 2013. Role of temperature and moisture in the survival and seedling physiology of a Great Basin perennial. Ecological Restoration. 31(4): 388-394.
Kildisheva, O.A.; Dumroese, R.K.; Davis, A.S. 2011. Overcoming dormancy and enhancing germination of Sphaeralcea munroana seeds. Hortscience. 46(12): 1672-1676.
Kildisheva, O.A.; Dumroese, R.K.; Davis, A.S. 2013. Boiled, tumbled, burned, and heated: Seed scarification techniques for Munro’s globemallow appropriate for large-scale application. Native Plants Journal. 14(1): 43-47.
Kildisheva, O.A.; Erickson, T.E.; Madsen, M.D.; Dixon, K.W.; Merritt, D.J. 2019. Seed germination and dormancy traits of forbs and shrubs important for restoration of North American dryland ecosystems. Plant Biology. 21: 458-469.
Kildisheva, O.A.; Erickson, T.E.; Merritt, D.J.; Madsen, M.D.; Dixon, K.W.; Vargas, J.; Amarteifio, R.; Kramer, A.T. 2018. Do abrasion- or temperature-based techniques more effectively relieve physical dormancy in seeds of cold desert perennials? Rangeland Ecology and Management. 71(3): 318-322.
Koutzoukis, S.; Pyke, D.A.; Brunson, M.W.; Baggio, J.; Calzado-Martinez, C.; Veblen, K.E. 2023. Goldilocks forbs: Survival is highest outside-but not too far outside-of Wyoming big sagebrush canopies. Restoration Ecology. 31(6): e13919.
Kramer, J.P. 1977. Taxonomic study of the planthopper genus Oecleus in the United States (Homoptera: Fulgoroidea: Cixiidae). Transactions of the American Entomological Society. 103(2): 379-449.
Kumar, D.; Kumar, A.; Prakash, O. 2012. Potential antifertility agents from plants: A comprehensive review. Journal of Ethnopharmacology. 140(1): 1-32.
La Duke, J. 2016. 48. Sphaeralcea. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 6 Malvaceae. http://www.efloras.org
La Duke, John C. 2012. Sphaeralcea munroana in Jepson Flora Project (eds.). Jepson eFlora, https://ucjeps.berkeley.edu/eflora/eflora_display.php?tid=45147 [Accessed 2025 September 9].
Lady Bird Johnson Wildflower Center [LBJWC]. 2023. Sphaeralcea munroana (Douglas) Spach. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants/result.php?id_plant=SPMU2 [Accessed 2025 September 9].
Lesica, P. 2012. Manual of Montana vascular plants. Fort Worth, TX: Botanical Research Institute of Texas (BRIT) Press. 779 p.
Luna, T.; Mousseaux, M.R.; Dumroese, R.K. 2018. Common native forbs of the northern Great Basin important for greater sage-grouse. Gen. Tech. Rep. RMRS-GTR-387. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station; Portland, OR: U.S. Department of the Interior, Bureau of Land Management, Oregon-Washington Region. 76 p.
Mansfield, D.H. 2000. Flora of Steens Mountain. Corvallis, OR: Oregon State University Press. 410 p.
Mayhew, W.W. 1963. Some food preferences of captive Sauromalus obesus. Herpetologica. 19(1): 10-16.
McAdoo, J.K.; Swanson, J.C.; Murphy, P.J.; Shaw, N.L. 2017. Evaluating strategies for facilitating native plant establishment in northern Nevada crested wheatgrass seedings. Restoration Ecology. 25(1): 1-10.
McArthur, D.; Sanderson, S. 2005. Forb and shrub genetics research. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2004 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 5-8.
McInnis, M.L.; Vavra, M. 1987. Dietary relationships among feral horses, cattle, and pronghorn in southeastern Oregon. Journal of Range Management. 40(1): 60-66.
Meyer, S.E.; Kjelgren, R.K.; Morrison, D.G.; Varga, W.A. 2009. Chapter 6: Native landscape pioneers tell their stories. In: Landscaping on the new frontier: Waterwise design for the Intermountain West. Logan, UT: Utah State University Press: 131-158.
Moerman, D. 2003. Native American ethnobotany: A database of foods, drugs, dyes, and fibers of Native American peoples, derived from plants. Dearborn, MI: University of Michigan. http://naeb.brit.org
Mueggler, W.F. 1950. Effects of spring and fall grazing by sheep on vegetation of the Upper Snake River Plains. Journal of Range Management. 3(4): 308-315.
Mull, J.F. 2003. Dispersal of sagebrush-steppe seeds by the western harvester ant (Pogonomyrmex occidentalis). Western North American Naturalist. 63(3): 358-362.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Ogle, D.; St. John, L.; Stannard, M.; Holzworth, L. 2012. Conservation plant species for the Intermountain West. Plant Materials Technical Note 24. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 57 p.
Ogle, D.; Tilley, D.; Cane, J.; St. John, L.; Fullen, K.; Stannard, M.; Pavek, P. 2019. Plants for pollinators in the Intermountain West. Plant Materials Tech. Note 2A. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 40 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Oregon Flora. 2025. Corvallis, OR: Oregon State University. https://oregonflora.org/ [Accessed 2025 September 9].
Ott, J.; Shaw, N. 2015. Seeding native species following wildfire in Wyoming big sagebrush (Artemisia tridentata ssp. wyomingensis): Effects of a new fire incident. In: Kilkenny, F.; Halford, A.; Malcomb, A., eds. Great Basin Native Plant Project: 2014 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 181-190.
Ott, J.E.; Cox, R.D.; Shaw, N.L.; Newingham, B.A.; Ganguli, A.C. ; Pellant, M.; Roundy, B.A.; Eggett, D.L. 2016. Post-fire drill-seeding of Great Basin plants: Effects of contrasting drills on seeded and nonseeded species. Rangeland Ecology and Management. 69(5): 373-385.
Parkinson, H. 2003. Landscaping with native plants of the Intermountain Region. Tech. Ref. 1730-3. Boise, ID: U.S. Department of the Interior, Bureau of Land Management. 46 p.
Parkinson, H.; Zabinski, C.; Shaw, N. 2013. Impact of native grasses and cheatgrass (Bromus tectorum) on Great Basin forb seedling growth. Rangeland Ecology and Management. 66(2): 174-180.
Parkinson, H.A. 2008. Impacts of native grasses and cheatgrass on Great Basin forb development. Bozeman, MT: Montana State University. Thesis. 73 p.
Pavek, P.L.; Cane, J.H.; Kildisheva, O.A.; Davis, A.S. 2011. Plant guide: Munro’s globemallow (Sphaeralcea munroana) (Douglas) Spach. Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 5 p.
Pekas, K.M.; Schupp, E.W. 2013. Influence of aboveground vegetation on seed bank composition and distribution in a Great Basin Desert sagebrush community. Journal of Arid Environments. 88: 113-120.
Pendery, B.M.; Rumbaugh, M.D. 1986. Globemallows: Forbs for Utah rangelands. Utah Science: 41-45.
Pendery, B.M.; Rumbaugh, M.D. 1990. Survival and growth of globemallow (Sphaeralcea) species in dry land spaced-plant nurseries. Journal of Range Management. 43(5): 428-432.
Pendery, B.M.; Rumbaugh, M.D. 1993. Globemallows. Rangelands. 15(3): 127-130.
Pendery, B.M.; Rumbaugh, M.D.; Mayland, H.F.; Harrison, P.A. 1993. Nonstructural carbohydrate and element pools in globemallow (Sphaeralcea): Defoliation effects and seasonal trends. Great Basin Naturalist. 53(4): 332-340.
Pendleton, R.L.; Pendleton, B.K.; Howard, G.L.; Warren, S.D. 2003. Growth and nutrient content of herbaceous seedlings associated with biological soil crusts. Arid Land Research and Management. 17(3): 271-281.
Pilliod, D.S.; Welty, J.L.; Toevs, G. 2017. Seventy-five years of vegetation treatments on public rangelands in the Great Basin of North America. Rangelands. 39(1): 1-9.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Robinson, G.S.; Ackery, P.R.; Kitching, I.; Beccaloni, G.W.; Hernández, L.M. 2025. HOSTS: A Database of the World’s Lepidopteran Hostplants [Data set]. Natural History Museum. https://doi.org/10.5519/havt50xw
Roth, T.E.; Holechek, J.L.; Hussain, M.Y. 1987. Germination response of three globemallow species to chemical treatment. Journal of Range Management. 40(2): 173-175.
Roundy, B.; Anderson, V.J.; Johnson, R. 2005. Forb production cultural practices. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2004 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 38-42.
Rumbaugh, M.D.; Mayland, H.F.; Pendery, B.M.; Shewmaker, G.E. 1993a. Element concentrations in globemallow herbage. Journal of Range Management. 46(2): 114-117.
Rumbaugh, M.D.; Pendery, B.M.; Mayland, H.F.; Shewmaker, G.E. 1993c. Registration of ARS-2936 scarlet globemallow germplasm. Crop Science. 33(5): 1106-1108.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shaw, R.J. 1995. Utah wildflowers: A field guide to northern and central mountains and valleys. Logan, UT: Utah State University Press. 218 p.
Society for Ecological Restoration; International Network for Seed Based Restoration; Royal Botanic Gardens [SER, INSR, RBGK]. 2023. Seed Information Database (SID). Available from: https://ser-sid.org/
Stevens, R.; Jorgensen, K.R.; Young, S.A.; Monsen, S.B. 1996. Forb and shrub seed production guide for Utah. 1996. AG 501. Logan, UT: Utah State University Extension. 52 p.
Sun, E.R. 2023. Assessing the effects of non-native flowering plants and anti-herbivory plastic (Vexar®) tubes on insect visitation to native plants. Boise, ID: Boise State University. Thesis. 79 p.
Taylor, R.J. 1992. Sagebrush country: A wildflower sanctuary. Missoula, MT: Mountain Press Publishing Company. 211 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Great Basin Native Plant Project [USFS GBNPP]. 2014. Seed weight table calculations made in-house. Report on file. Boise, ID: U.S. Department of Agriculture, Forest Service, Boise Aquatic Sciences Laboratory. Available: https://www.fs.fed.us/rm/boise/research/shrub/Links/Seedweights.pdf
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2024. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2000. Washington and Oregon guide for conservation seedings and plantings. Portland, OR: U.S. Department of Agriculture, Natural Resources Conservation Service. 126 p.
USDA Natural Resources Conservation Service [USDA NRCS]. 2025. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2016. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 37 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2023. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 45 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2025. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 1987. A Utah flora. The Great Basin Naturalist Memoir 9. Provo, UT: Brigham Young University. 894 p.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Whitcomb, H.L. 2011. Temperature increase effects on sagebrush ecosystem forbs: Experimental evidence and range manager perspectives. Logan, UT: Utah State University. Thesis. 125 p.
Wik, P.A. 2002. Ecology of greater sage-grouse in south-central Owyhee County, Idaho. Moscow, ID: University of Idaho. Thesis. 141 p.
Young, S.; Bouck, M. 2011. Stock seed production of native plants for the Great Basin. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2010 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 131-135.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2025. Munro’s globemallow (Sphaeralcea munroana). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org/species/munros-globemallow-sphaeralcea-munroana/