Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
March 2020
Nomenclature
Nakedstem sunray (Enceliopsis nudicaulis) (A. Gray) A. Nelson belongs to the Ecliptinae subtribe and Heliantheae tribe within the Asteraceae family. Nomenclature follows Welsh et al. (2016).
Family
Asteraceae – Aster family
Genus
Enceliopsis
Species
nudicaulis
NRCS Plant Code
ENNU (USDA NRCS 2019).
Subtaxa
Some systematists (Cronquist 1972; Welsh et al. 2016) recognize nakedstem sunray varieties: corrugata and bairdii, although others do not support varietal distinctions (Sanders and Clark 1987; Curtis 2006).
Synonyms
Encelia nudicaulis A. Gray
Common Names
Nakedstem sunray, Ash Meadows sunray, nakedstem, naked-stemmed daisy, sunray (USFWS 1983; Curtis 2006; Welsh et al. 2016; USDA NRCS 2019)
Chromosome Number
Reported chromosome numbers include: 2n = 32, 34, and 36; but most consistently reported are 2n = 34 and 36 (Reveal and Styer 1974; Hickman 1993; Curtis 2006; Welsh et al. 2016).
Hybridization
The possibility of hybrids within the Enceliopsis genus have been suggested based on DNA evidence. Sequencing of two nuclear and two chloroplast regions suggests that a single plant collected from co-occurences of nakedstem sunray and Panamint daisy (E. covillei) in California may represent a hybrid or backcrossed individual (Fehlberg and Ranker 2007). Hybrids of nakedstem sunray, Panamint daisy, and silverleaf sunray (E. argophylla) were created artificially under cultivation but did not survive long enough to evaluate fertility (Clark 1998).
Distribution
Nakedstem sunray occurs in Idaho, Utah, Nevada, Arizona, and California (Curtis 2006). It is widespread throughout the Great Basin (Clark 1998) and occurs as restricted populations in southwestern and central Idaho, northern Arizona’s Mohave and Coconino counties, and southeastern California’s Inyo and San Bernardino counties (Cronquist et al. 1994).
Variety corrugata occurs only in the Ash Meadows area of Nye County, Nevada, where soils are strongly alkaline and poorly drained (Cronquist 1972). Variety bairdii occurs in Washington County, Utah, and Mojave County, Arizona. It is especially common on limestone and dolomite substrates in the southern Beaver Dam Mountains in Utah and the Moenkopi sandstone formation and southeast of St. George, Utah, and adjacent Mohave County, Arizona (Welsh et al. 2016). Variety corrugata is federally listed as a Threatened species (USFWS 2020).
Habitat And Plant Associations
Nakedstem sunray occurs in dry, open (Fig. 1) desert shrubland communities. In the Intermountain West, it occupies well-drained slopes, often with sagebrush (Artemisia spp.), juniper (Juniperus spp.), and sometimes with saltbush (Atriplex spp.) (Cronquist et al. 1994). At its northern most distribution in central Idaho, nakedstem sunray occurs on dry rocky bluffs in sagebrush communities above the Salmon River (Blake 1913). In the arid rain shadow of the White Cloud and Salmon River mountains it is found in association with shadscale saltbush (A. confertifolia), Indian ricegrass (Achnatherum hymenoides), Douglas’ dustymaiden (Chaenactis douglasii), and, Challis milkvetch (Astragalus amblytropis, a rare endemic), on south- and west-facing, xerophytic slopes along the Salmon River (Rittenhouse and Rosentreter 1994).

Figure 1. Nakedstem sunray growing in a mixed desert shrubland in Utah. Photo: USDI Bureau of Land Management BLM UT 020–5 Seeds of Success (SOS).
In Utah, nakedstem sunray occurs with blackbrush (Coleogyne ramosissima), rabbitbrush (Chrysothamnus spp.), mormon tea (Ephedra viridis), shadscale saltbush, spiny hopsage (Grayia spinosa), and pinyon (Pinus spp.)-juniper communities (Fig. 2) (Welsh et al. 2016). On the San Rafael Swell in south-central Utah, nakedstem sunray is common in salt desert shrub communities dominated by shadscale saltbush and mixed desert shrub communities dominated by one or several of the following species: big sagebrush (Artemisia tridentata), Bigelow sage (A. bigelovii), bud sagebrush (Picrothamnus desertorum), broom snakeweed (Gutierrezia sarothrae), or yellow rabbitbrush (Chrysothamnus viscidiflorus) (Harris 1983).

Figure 2. Nakedstem sunray growing at the edge of a pinyon-juniper woodland in Utah. Photo: Provo Shrub Sciences Laboratory, USFS, Rocky Mountain Research Station.
In more southern and western areas in Nevada, nakedstem sunray occurs with creosote bush (Larrea tridentata) (Munz and Keck 1973; Cronquist et al. 1994). Additional local populations of nakedstem sunray are associated with limestone outcrops at the Nevada Test Site and in central-southern Nevada where is occurs with saltbush and black sagebrush (Artemisia nova) (Beatley 1976). In the Clark Mountains in trans-montane southern California, nakedstem sunray occurs in gypsum-rich soils with shadscale saltbush, cottontop cactus (Echinocactus polycephalus), Schott’s pygmycedar (Peucephyllum schottii), and Fremont’s dalea (Psorothamnus fremontii) (Thorne 1982). Northwest of Las Vegas, in Ash Meadows valley, nakedstem sunray (variety corrugata) occurs in dry washes with saline soils in association with shadscale saltbush, alkali goldenbush (Isocoma acradenia var. acradenia), desert bearpoppy (Arctomecon merriamii), and basin yellow cryptantha (Cryptantha confertiflora) (Mozingo and Williams 1980). In the southern and eastern parts of Ash Meadows where soils are arid and alkaline it occurs with burrobush (Ambrosia dumosa) (Sada 1990).
In southern California’s Clark Mountains, nakedstem sunray populations are found in gypsum-rich soils associated with shadscale saltbush, cottontop cactus (Echinocactus polycephalus), Schott’s pygmycedar (Peucephyllum schottii), and Fremont’s dalea (Psorothamnus fremontii) (Thorne 1982).
Elevation
Nakedstem sunray occupies habitats from 2,300 to 7,600 ft (700–2,320 m) (Cronquist et al. 1994; Curtis 2006; Welsh et al. 2016). Its elevation range is slightly narrower in Utah, 3,400 to 7,600 ft (1,040–2,320 m) (Welsh et al. 2016), and in California from 3,200 to 6,600 ft (950–2,000 m) (Hickman 1993).
Soils
Throughout its range, nakedstem sunray populations are found on deep, well-drained, dry soils (Harris 1983; Cronquist et al. 1994; Rittenhouse and Rosentreter 1994), rocky or hard compacted clays (Blake 1913; Munz and Keck 1973) with high levels of carbonates (Welsh et al. 2016), salinity (USFWS 1983), or gypsum (Thorne 1982). In east-central Idaho, nakedstem sunray occurs on xerophytic, south- and west-facing slopes where soils exceed 3 ft (> 1 m) (Rittenhouse and Rosentreter 1994). In Utah, it is common on gypsiferous or calciferous semi-barren knolls and in limestone or dolomite soils (Welsh et al. 2016). On the San Rafael Swell formation in south-central Utah, nakedstem sunray occurs in salt desert shrublands with alkaline clay soils and mixed-desert shrublands on deep, well-drained soils (Harris 1983). On the Grand Canyon rim west of Cathedral Monument in Arizona, nakedstem sunray is associated with red sandstone formations (Brian et al. 1999). In the Ash Meadows valley in Nevada, it grows in dry washes on saline clays (USFWS 1983). In other parts of California, it grows in sandy to rocky clays, compacted arid soils, and in the xerophytic Death Valley region (Munz and Keck 1973).
Description
Nakedstem sunray is a tufted perennial with erect stems developing from a stout multibranched caudex and thick woody taproot (Fig. 3) (Munz and Keck 1973; Cronquist et al. 1994; Curtis 2006; Welsh et al. 2016). Scapose stems typically grow 4 to 16 in (10–40 cm) tall, though some plants reach 24 in (60 cm) (Welsh et al. 2016). Stems are woody, densely leafy at the base, and leafless above. Both leaves and stems are covered in short white hairs which give the herbage a dull gray appearance (Hickman 1993; Cronquist et al. 1994; Curtis 2006; Welsh et al. 2016). Leaf axils often have tufts of woolly hairs (Munz and Keck 1973). Basal leaves are alternate and petiolate (Curtis 2006). Petioles can be one to three times as long as the leaf blades. Leaf blades are three-nerved, 0.8 to 3.5 in (2–9 cm) long, and vary greatly in width (0.4 to 4 in [2–10 cm]) (Munz and Keck 1973; Cronquist et al. 1994; Curtis 2006; Welsh et al. 2016). Blades are ovate to elliptic or orbicular to spatulate, cuneate to subcordate basally, and obtuse to rounded apically (Welsh et al. 2016).

Figure 3. Nakedstem sunray plant with woody taproot nearly as long as the aboveground stems are tall. Photo: USDI, Bureau of Land Management, ID 330, Seeds of Success.
Nakedstem sunray flower heads are 1.6 to 3.5 in (4–9 cm) in diameter and occur singly at the top of stems (Fig. 4). Flower heads are comprised of 200 to 500 or more yellow disk florets and 11 to 28 yellow ray florets, most commonly 21 (Hickman 1993; Cronquist et al. 1994; Curtis 2006). Ray florets are pistillate but sterile, and disk florets are bisexual and fertile (Curtis 2006; Welsh et al. 2016). Involucres are 0.5 to 0.9 in (1.3–2.2 cm) tall, 1.2 to 2.2 in (3–5.6 cm) wide, densely gray tomentose with short hairs, and have 30 to 65 or more persistent phyllaries in 3 to 5 series (Cronquist et al. 1994; Curtis 2006). Fruits are one–seeded achenes that are wedge shaped, dark brown to black, 0.4 to 0.5 in (9–12 mm) long, 0.1 in (3.5 mm) wide, and silky hairy (Blake 1913; Cronquist et al. 1994; Welsh et al. 2016). Achenes have two stout awl-shaped appendages that may or may not surpass the length of the long hairs covering the achene (Figs. 6 and 7) (Cronquist et al. 1994; Curtis 2006).

Figure 4. Small nakedstem sunray plant growing in rocky soils in Idaho. Photo: BLM ID 330 SOS.
Varietal descriptions. Nakedstem sunray varieties: corrugata and bairdii are distinguished morphologically by limited leaf and flowerhead characteristics but occupy restricted and unique locations and habitats (See Distribution section). Variety corrugata has small leaves, 0.4 to 0.8 in (1–2 cm long), that are strongly ruffled-corrugate, especially toward the margins (Cronquist 1972). Variety bairdii plants are large, 12 to 24 in (30–60 cm) tall, and those growing in Washington County, Utah, have larger than average flower heads (Welsh et al. 2016). In a comparative morphological study of the capitulum produced by sunrays (Enceliopsis spp.), researchers found no morphological differences supporting varietal descriptions (Sanders and Clark 1987).
Reproduction
Nakedstem sunray produces flower heads with pistillate but sterile ray florets and bisexual disk florets (Curtis 2006; Welsh et al. 2016). Clark (1998) suggests nakedstem sunray is self-incompatible (Clark 1998). Flowering is common in the spring but can occur as late as August (Beatley 1976; Brian et al. 1999; Curtis 2006). Abundance or variability in seed production and seedling recruitment were not reported in the literature. Germination was variable in controlled conditions, see Germination Biology section.
Ecology
Ecological and disturbance studies were lacking in the literature. Nakedstem sunray tolerates very dry conditions (Munz and Keck 1973), has a woody taproot, and is considered long-lived (Welsh et al. 2016). Given these traits, long-term persistence could be expected once plants are established.
Wildlife And Livestock Use
Adult and young (<85 days old) pronghorn (Antilocapra americana) utilized nakedstem sunray most in the summer on the Desert Experimental Range (DER) in western Millard County, Utah. Diet composition was evaluated from rumen and feces samples collected in spring and summer and maximum use was 13% (Smith and Beale 1980). When pronghorn were tracked for 2 years, nakedstem sunray forage was removed in the spring and early summer evaluation periods. Nakedstem sunray made up a high of 13% of pronghorn diets in 1 of the 2 years (Smith and Malechek 1974). In earlier studies (Beale and Smith 1970) reported that preference for nakedstem sunray was low on the DER. Herbage production of nakedstem sunray on the DER averaged 0.2 lb/ac (0.2 kg/ha) and was a low volume of spring and moderate volume of summer pronghorn diets.
Ethnobotany
Shoshone Indians in Nevada used a tea from boiled nakedstem sunray roots to treat bloody diarrhea, veneral diseases and a tea from boiled leaves to treat coughs. Tribal members would travel long distances to collect the plant (Train et al. 1941).
Horticulture
Although not currently grown for horticulture, large yellow flower heads extending above tufts of gray green leaves, suggest this species may be considered useful for xeriscaping (LBJWC 2020).
Revegetation Use
Although no studies reported use of nakedstem sunray in restoration, the species has been grown in test plots and farms for seed production (Shock et al. 2018; Jensen, USFS RMRS, personal communication, Jan 2020). Its tolerance of dry conditions and compacted clay soils as well as its long-lived nature suggests it may be useful in revegetation of degraded or disturbed sites receiving little precipitation (Munz and Keck 1973; Welsh et al. 2016).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for nakedstem sunray, generalized provisional seed zones developed by Bower et al. (2014), may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat:moisture index). Omernik Level III Ecoregions (Fig. 5; Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Seedlot Selection Tool (Howe et al. 2017) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
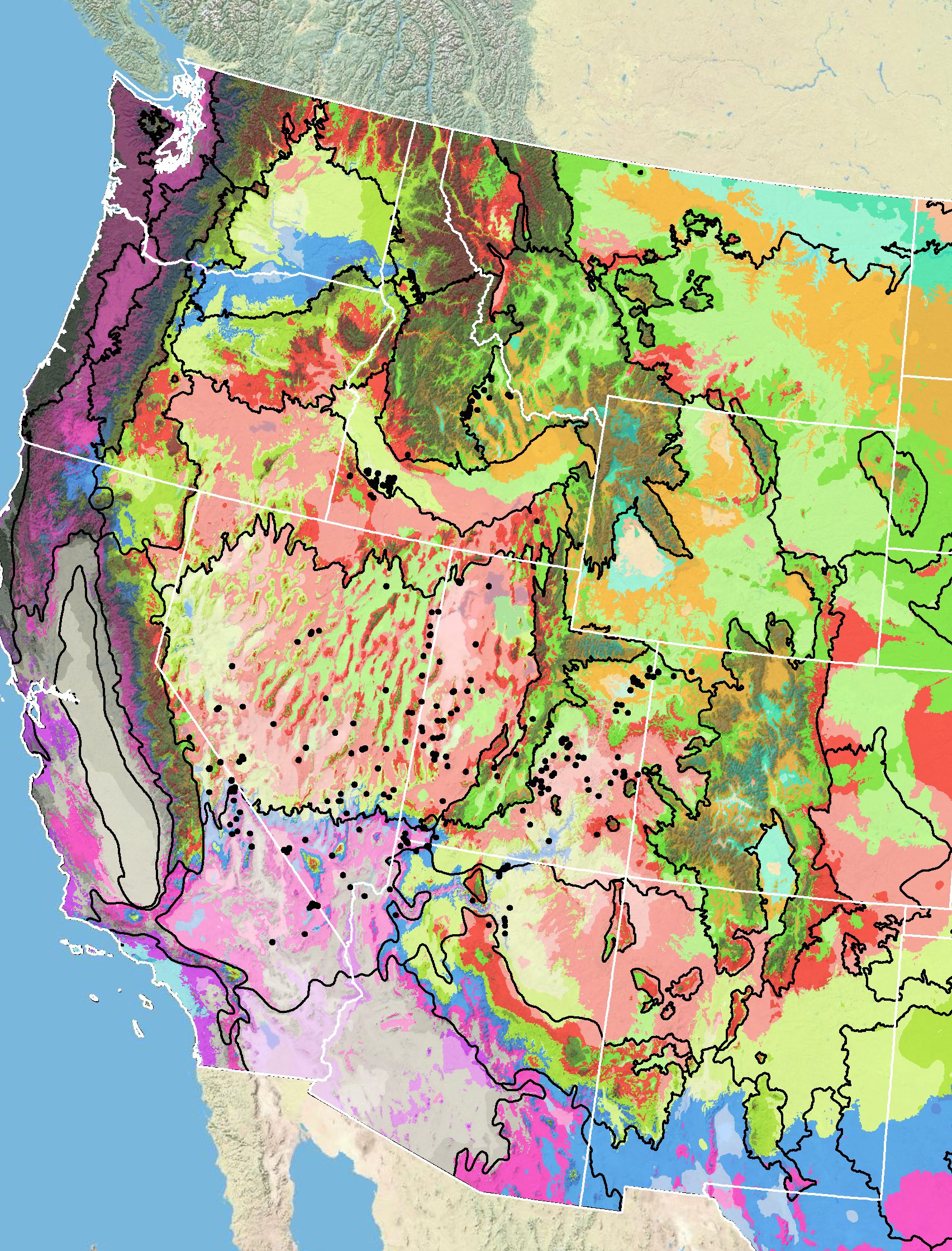
Figure 5. Distribution of nakedstem sunray (black circles) based on geo-referenced herbarium specimens and observational data from 1881-2016 (CPNWH 2017; SEINet 2017; USDI USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USDI USGS.
Releases
As of 2020, there were no nakedstem sunray germplasm releases.
Wildland Seed Collection
Nakedstem sunray occurs in small stands and produces seed indeterminately, which suggests successful harvesting of wildland seed requires planning, scouting, and flexibility in timing of seed collection efforts. In Utah, native stands of nakedstem sunray were typically small but ranged to about 5 ac (2 ha) in size. Insect damage was severe at only one of many wildland collection sites (Jensen, USFS RMRS, personal communication, Jan 2020).
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (Young et al. 2020; UCIA 2015). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Reported wildland seed collection (Fig. 6) dates for nakedstem sunray range from late May to early July in Idaho, Utah, and Nevada, but most were made in June (Table 1). The Bureau of Land Management’s Seeds of Success collection crews collected nakedstem sunray seed in late June and early July in Idaho, Utah, and Nevada. The latest of four collections was made from the highest elevation site (USDI BLM SOS 2017). Wildland seed collection dates in Utah made by Provo Shrub Lab personnel for establishment of common garden studies were made from May 25 to July 7 for sites ranging from 5,128 to 7,203 ft (1,563–2,195 m) (Jensen, USFS RMRS, personal communication, Jan 2020).
Table 1. Dates and locations of seed collections made by the USDI, BLM Seeds of Success collection crews.
|
Date |
Location |
Elevation (ft) |
Site |
|
June 24, 2010 |
Millard Co, UT |
5697 |
South slope, desert scrub |
|
June 29, 2010 |
Millard Co, UT |
6102 |
East slope, juniper, sand desert |
|
July 7, 2010 |
White Pine Co, NV |
7203 |
East slope, pinyon–juniper |
|
June 27, 2016 |
Custer Co, ID |
6250 |
East to south slopes, dry rolling hills in the Salmon River uplands |

Figure 6. Collection of less than 100 individual nakedstem sunray seeds. Photo: Provo Shrub Sciences Laboratory, USFS, Rocky Mountain Research Station.
Collection Methods
Seed is collected by hand. Seed disperses quickly when ripe, the presence of seed on the ground indicates seed maturity. Full seed heads can be clipped, but to collect only ripe seed, collectors can swat the seed heads over a collection hopper using a racquet. Ripe seed is dislodged, leaving remaining immature seed to continue to mature. Swatting results in a harvest that is easier and faster to clean than a collection of clipped seed heads. (Jensen, USFS RMRS, personal communication, Jan 2020).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of nakedstem sunray.
Post-Collection Management
Any vegetative material (e.g. stems, large leaves) should be removed from collected seed to reduce the moisture content and thus the potential for mold growth. Keep collections in dry, shaded locations, and do not allow seed to overheat. Seed should be loosely packed in well aerated collection bags to provide good air circulation. Seed can be dried by laying it out in thin layers on screens in a well-ventilated, rodent–free area. Potential seed insects should be controlled using appropriate chemicals or by freezing the seed collection for 48 hours once thoroughly dried. Temporary storage prior to delivery at the seed extractory should be in a cool (< 50 °F [10 °C]), dry (< 35% relative humidity) location.
Seed Cleaning
The USFS Bend Seed Extractory used the following procedure to clean wild–collected nakedstem sunray seed (Fig. 7): 1) Sieve off loose seed from stems and flower heads. 2) Process one time through a Missoula De-winger to reduce the amount of hairs on the seed using a black liner with a feed of 20, speed of 4, and tilt of 12 to 15. 3) Sieve seed again to remove more chaff from the seed heads. Large seed lots were processed using an office clipper with a 17 round screen to remove stems and air of 1 to remove empty seed. Smaller lots required less processing and were put directly through the Continuous Seed Blower (Mater CSB, Corvallis, OR) with a feed of 6 to 9 and an air setting of 250 to 300 (K. Herriman, USFS Bend Seed Extractory, personal communication, Jan 2020).

Figure 7. Four individual nakedstem sunray seeds measuring about 1 cm long and 5 mm wide. Photo: BLM UT 020 SOS.
Seed Storage
Dry seed should be stored where conditions are cool and dry.
Seed Testing
Seed purity is evaluated using standard procedures (AOSA 2016). There are no AOSA procedures for testing germination of nakedstem sunray seed. Tetrazolium chloride testing (TZ) should follow that suggested for other Asteraceae species, which includes cutting seeds longitudinally leaving the distal end intact, soaking for 6 to 12 hours in 0.1% TZ concentration at (86–95 °F [30–35 °C]). The embryos of viable seed will be entirely stained (AOSA 2010).
Germination Biology
Studies report low (10-30%; Rawlins et al. 2012) to high (75-100%; Kildisheva et al. 2018; Love and Akins 2019) germinability of nakedstem sunray seed. Love and Akins (2019) reported that nakedstem sunray seed germinated well at 40 °F (4 °C) and 70 °F (21 °C). After 2 to 4 weeks at 70 °F (21 °C), 75% of seed germinated and after 3 to 6 weeks at 40 °F (4 °C), 100% of seed germinated. No seed pretreatments were noted and no other study details were provided (Love and Akins 2019). In preliminary germination studies, Karrfalt and Vankus (2012) reported germination of nakedstem sunray was best after 4 weeks of moist pre-chilling and incubation at 59 °F (15 °C) or 68 °F (20 °C). Seed was not scarified.
Kildisheva et al. (2018) found that a cultivated population of nakedstem sunray grown in Utah was non-dormant with germination ranging between 55 and 100% after up to 4 weeks at temperatures from 41 to 77 °F (5–25 °C). Germination was nearly 100% and occurred most readily at temperatures of 59 °F (15 °C) and 68 °F (20 °C) suggesting temperature was most influential on germination timing. Seed germination was also improved with karrikinolide at 77 °F (25 °C) and 1mM gibberellic acid at all temperatures. Generally, nakedstem sunray was quick to germinate relative to the other Great Basin species tested, requiring only 11 days for 50% of the seeds in the tested population to germinate. Seeds were dried for 4 weeks and kept in sealed bags at (-18 °C) for 3 months prior to conducting experiments. Seeds were incubated at constant temperatures with alternating 12 hr light-12 hr dark cycles (Kildisheva et al. 2018).
In laboratory experiments, germination of nakedstem sunray seed collected in Blind Valley, Utah, was less than 30% at constant and simulated diurnal spring temperatures, and germination was much more rapid at warm than cool or cold diurnal temperatures. Seed was first dusted with fungicide then germinated (in groups of 30 seeds) on moist blotter paper in petri dishes in an incubation chamber. Incubation occurred at constant temperatures ranging from 41 to 95 °F (5–35 °C) and diurnal simulated soil temperatures on a cold March, cool April, and warm May day in a big sagebrush community in the Shoshone Mountains of Nevada (Rawlins et al. 2012). Germination was 10% or less at constant cold and warm to hot temeratures of 41, 68, 77, 86, and 95 °F (5, 20, 25, 30, and 35 °C). Germination was 24.2% at 50 °F (10 °C) and 29.2% at 59 °F (15 °C). Germination was 19.2% at cold, 25.8% at cool, and 24.2% at warm diurnal Great Basin temperatures. Days to 10% germination was 13.5 at cold, 17.7 at cool, and 0.92 at warm diurnal temperature incubation (Rawlins et al. 2012).
Wildland Seed Yield And Quality
Post–cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 1 (USFS BSE 2017). The results indicate that nakedstem sunray seed can generally be cleaned to high levels of purity and seed fill and that the viability of fresh seed is generally high. Seed weight and viability reported by others (57,607–77,098 seeds/lb [127,000–170,000 seeds/kg], >90% TZ viability (USFS GBNPP 2014; Kildisheva et al. 2018; Jensen, USFS RMRS, personal communication, Jan 2020) are similar to those reported in Table 2.
Table 2. Seed yield and quality of nakedstem sunray seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Seed lot characteristic |
Mean |
Range |
Samples (no.) |
|
Bulk weight (lbs) |
0.42 |
0.18–0.99 |
4 |
|
Clean weight (lbs) |
0.19 |
0.02–0.43 |
4 |
|
Clean–out ratio |
0.42 |
0.09–0.62 |
4 |
|
Purity (%) |
98 |
98–98 |
4 |
|
Fill (%)¹ |
96 |
92–98 |
4 |
|
Viability (%)² |
92 |
84–97 |
4 |
|
Seeds/lb |
66,162 |
43,901–84,013 |
4 |
|
Pure live seeds/lb |
62,188 |
39,581–80,686 |
4 |
¹ 100 seed X–ray test
² Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well.
Agricultural Seed Production
Nakedstem sunray seed production plots were grown and evaluated by the RMRS Provo Shrub Sciences Laboratory (PSSL) at Fountain Green, Utah (Fig.8) (Jensen, USFS RMRS, personal communication, Jan 2020) and at the Oregon State University’s Malheur Experiment Station (OSU MES) in Ontario, Oregon (Shock et al. 2018). At both sites, stand establishment was spotty and seed production often less than 50 lbs/ac (56 kg/ha). Seed is produced in the first growing season and at OSU MES was harvested for up to 3 years (Shock et al. 2018).

Figure 8. Large nakedstem sunray plant growing in a seed production field in Utah. Photo: Provo Shrub Sciences Laboratory, USFS, Rocky Mountain Research Station.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Site Preparation
At both the Utah and Oregon sites, nakedstem sunray was planted into weed-free plots. Row cover improved final emergence of nakedstem sunray in Oregon (Shock et al. 2014) but not in Utah (Jensen, USFS RMRS, personal communication, Jan 2020). At Fountain Green, Utah (5,700 ft [1,700 m]), soils are gravelly loams and the dominant shrub in surrounding vegetation is mountain big sagebrush (Artemisia tridentata subsp. vaseyana). At Fountain Green, fields were disked, fallowed, tilled and compacted prior to fall seeding nakedstem sunray (Jensen, USFS RMRS, personal communication, Jan 2020).
At OSU–MES, emergence of nakedstem sunray was improved when seeds were fungicide treated and covered with row cover (Shock et al. 2014). Seeded areas were weed-free, silt loam soils with pH of 8.3 and 1.1% organic matter (Shock et al. 2018). Soil crusting and bird damage were causes of poor stand emergence in earlier fall seeding of other forbs at OSU MES but observations revealed natural emergence of seed shed by established perennials occurred when seeds were covered by organic debris (Shock et al. 2014). So began tests of various seed protection measures, including row cover (N-sulate, DeWitt Co, Sikeston, MO) to protect against soil desiccation and bird damage; sawdust (0.26 oz/ft [24 g/m] of row) to mimic protection by organic debris; sand (0.65 oz/ft [60 g/m]) to hold seed in place, distributed over the sawdust when both were used; hydroseeding mulch (Hydrostraw, Manteno, IL) at 0.26 oz/ft (24 g/m); and fungicide seed pre–treatment (0.35 oz [10 g] each of Metalaxyl [Ridomil] and Captan in 1.1 pints (0.5 L) of water) to protect from damping off. Row cover and fungicide treatments together resulted in the greatest emergence of nakedstem sunray, which was still low (11.5%) Seeding occurred on November 15, 2012. Plots were drip irrigated for 24 hours on November 20, 2009, and emergence results evaluated on March 10, 2010 (Table 3; Shock et al. 2014).
Table 3. Emergence of nakedstem sunray on April 18, 2013 (5 mo after fall seeding) with various combinations of seed protection treatments at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2014).
|
Row cover |
Sawdust |
Sand |
Mulch |
Fungicide seed treatment |
% of seed planted* |
|
Yes |
Yes |
No |
No |
Yes |
10.9b |
|
Yes |
No |
No |
No |
Yes |
11.5b |
|
Yes |
Yes |
No |
No |
No |
6.4ab |
|
No |
Yes |
No |
No |
Yes |
0.1a |
|
Yes |
Yes |
Yes |
No |
Yes |
7.8ab |
|
No |
No |
No |
Yes |
Yes |
0a |
|
No |
No |
No |
No |
No |
3.7a |
*Emergence percentages followed by different letters are significantly different (P < 0.05).
Weed Management
In seed production plots in Oregon and Utah, weeds were largely controlled by hand or mechanically with periodic herbicide treatments. At OSU MES, stands were hand weeded, except in the fall of the second post-seeding year when pendimethalin, a pre-emergent, was broadcast applied at 2 pints/ac (2.3 L/ha) to control weed emergence in established stands. Success of the weed treatments was not reported (Shock et al. 2018). In Utah, imazapic applied at 5 oz/ac (350 g/ha) in the fall when nakedstem sunray plants were entirely dormant, caused no stand mortality. Although the treatment effect on seed production was not quantified, casual observation suggested it did not impact seed production in the following year (Jensen, USFS RMRS, personal communication, Jan 2020).
Herbicides are not registered for this species, and the results do not constitute an endorsement of specific companies or products or recommendations for use. The research, however, could contribute to future registration efforts.
Seeding
Fall seeding at shallow depths was used to establish nakedstem sunray (Shock et al. 2018; Jensen, USFS RMRS, personal communication, Jan 2020). At Fountain Green, Utah, nakedstem sunray was seeded 0.25 in (0.6 cm) deep, at a rate of 25 PLS/linear ft (82 PLS/linear m). In a seeding depth study, field emergence was reduced as seeding depth increased from 0.5 to 1.6 in (1.4–4 cm) and averaged 0.73% over all seeding depths at three field sites (Jensen, USFS RMRS, personal communication, Jan 2020).
At OSU MES, nakedstem sunray was seeded first on October 3, 2012 using a small-plot grain drill. Seed was deposited on the soil surface at 20 to 30 PLS/ft (66–98 PLS/m) of row, and rows were spaced 30 in (76 cm) apart. A narrow band of sawdust was applied over the seed at 0.26 oz/ft (18.7 g/m) of row, and plots were covered with N-sulate row cover. Because of poor stand establishment and growth, plots were seeded again on November 2, 2015, using the same method (Shock et al. 2018).
Establishment And Growth
In the seed production test plots in Oregon and Utah, nakedstem sunray failed to establish dense stands and this may have been a result of excess moisture (Shock et al. 2018; Jensen, USFS RMRS, personal communication, Jan 2020). Initial stand establishment after fall seeding in Utah was fair, but up to 50% mortality occurred within the first 2 post-seeding years. Researchers suspected root rot caused mortality from inadequate soil drainage, but noted that longevity of surviving plants was good (Jensen, USFS RMRS, personal communication, Jan 2020).
At OSU MES, several measures were used to prevent bird predation of emerging seedlings. Row cover protected seedlings from birds until March, when it was replaced with bird netting. As seedlings emerged, bird seed was provided away from the plots to distract California quail (Callipepla californica) (Shock et al. 2018).
Irrigation
Although supplemental irrigation increased nakedstem sunray seed production in some years at OSU MES, it was also associated with a decline in stand density (Shock et al. 2018). Irrigation trials began in March 2013 in plots seeded in late October 2012 and reseeded in early November 2015. Irrigation treatments of 0, 4, or 8 in (101 or 203 mm) of water were delivered at about 2-week intervals beginning at the time of bud formation and flowering (Table 4) through drip tape buried 12 in (30 cm) deep. Winter and spring precipitation was lower than the 5-year average in 2013 and in 2017, and fall, winter, and spring precipitation was lower in 2018 than average. Growing degree days were greater than average in 2013 through 2016 and in 2018 and near average in 2017 (Table 5). Extensive die off of nakedstem sunray stands occurred over the winter of 2014–15 and was most severe in plots receiving the highest levels of irrigation. Reseeding that occurred in fall 2015 was successful but stands continued to decline, especially irrigated plots (Table 7; Shock et al. 2018).
Table 4. Timing of flowering and seed harvest dates for seed production fields growing over a period of 6 years at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2018). Seed yields are provided in Table 7.
|
Year |
Flower start |
Flower peak |
Flower end |
Weekly harvest |
|
–––––––––––––––––––––––––––Date–––––––––––––––––––––––––– |
||||
|
2013 |
30 Jun |
–––– |
15 Sept |
8–30 Aug |
|
2014 |
5 May |
1 July |
30 July |
14 July–30 Aug |
|
2015 |
28 Apr |
13 May |
5 Aug |
2 Jun–15 Aug |
|
2016 |
20 Apr |
–––– |
30 July |
27 Apr–29 July |
|
2017 |
11 May |
7 Jun |
20 Aug |
4 Jun–15 Aug |
|
2018 |
30 Apr |
26 Jun |
30 July |
27 Apr–27 July |
Table 5. Precipitation (in) and growing degree hours (°F) at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2018).
|
Year |
Spring (in) |
Winter + Spring (in) |
Fall + Winter + Spring (in) |
Growing degree hrs (50–86 °F) Jan–June |
|
2013 |
0.9 |
2.4 |
5.3 |
1319 |
|
2014 |
1.7 |
5.1 |
8.1 |
1333 |
|
2015 |
3.2 |
5.9 |
10.4 |
1610 |
|
2016 |
2.2 |
5.0 |
10.1 |
1458 |
|
2017 |
4.0 |
9.7 |
12.7 |
1196 |
|
2018 |
1.9 |
4.9 |
5.8 |
1342 |
|
5–yr ave |
2.4 |
5.6 |
9.3 |
1207 (25–yr ave) |
Pollinator Management
At Fountain Green, Utah, pollination by native pollinators was excellent throughout the season and no insect damage issues with seed production were noted (Jensen, USFS RMRS, personal communication, Jan 2020).
Pest Management
Powdery mildew (Leveillula picridis) was collected from nakedstem sunray plants growing at OSU MES (Braun and Mohan 2013). Infected plants were not treated and infection was not reported as impacting growth or seed production.
Seed Harvesting
Flowering and seed production of nakedstem sunray is indeterminate, and to maximize seed yields multiple harvests are required. In Utah, seed was harvested by hand up to eight times a season, but the last harvest was done with a flail vac (Jensen, USFS RMRS, personal communication, Jan 2020). In Oregon, seed was harvested weekly using a blower in vacuum mode (Shock et al. 2018).
Seed Yields And Stand Life
Seed yields were typically less than 50 lbs/ac (56 kg/ha) in the Utah and Oregon test plots due to low density stands. However, longevity of surviving plants was good (Shock et al. 2018; Jensen, USFS RMRS, personal communication, Jan 2020).
Seed yield varied by accession at plots in Utah. Seed was harvested from 2011 through 2013 in fields seeded in the fall of 2010. Results of multiple harvests made between June 20 and July 13 in 2013 are provided in Table 6 (Gunnell 2017).
Table 6. Plant growth and seed production for 2 accessions of nakedstem sunray seeded in 2010 in Fountain Green, Utah. Plot size was 5 × 80 ft, but because of the limited ability to capture highly variable plants/area this measurement was not reported (Gunnell 2017).
|
Accession |
Plant height (in) |
No. of plants |
Clean seed yield (g) |
1000 seeds/weight (g) |
|
Crystal Peak |
14.6 |
180 |
500 |
6 |
|
Painted Pot |
16.9 |
103 |
520 |
6.3 |
At OSU MES, nakedstem sunray seed production was increased with irrigation in 2 of 6 years and maximum seed production occurred without irrigation in one year (Table 7). The maximum average seed yield was 26 lbs/ac (29 kg/ha) but in 2015 non-irrigated stands produced 105 lbs/ac (118 kg/ha) (Table 7). Stands produced low levels of harvestable seed for up to 3 years (Shock et al. 2018).
Table 7. Seed yield (lbs/ac) for showy goldeneye stands with and without growing season irrigation at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2018).
|
Year* |
0 in |
4 in |
8 in |
|
––––––––––lbs/ac––––––––– |
|||
|
2013 |
2.3 |
6.8 |
5.9 |
|
2014 |
1.5a |
34.6b |
29.1b |
|
2015 |
15.7b** |
3.2a |
4.4a |
|
2016 |
10.5a |
47.6b |
45.9b |
|
2017 |
105.0b |
43.2a |
25.0a |
|
2018 |
20.1 |
20.5 |
20.1 |
|
Mean |
25.9 |
26.2 |
21.7 |
*Seed was hand harvested weekly each year.
**Values within a year followed by different letters are significantly different (P ≤ 0.1).
Nursery Practice
Nakedstem sunray was grown from wild-collected achenes at California State Polytechnic University. Soil used was eight parts pumice, four parts commercial container soil, one part gypsum. Seedlings were grown indoors and then transferred to 8-in (20 cm) pots and kept outdoors. No other details were provided (Sanders and Clark 1987), but in a later publication, Clark (1998) reported that nakedstem sunray was difficult to cultivate.
Wildland Seeding And Planting
At the Rancho Santa Ana Botanic Garden, a relative of nakedstem sunray, Panamint daisy, was grown in the nursery from wildland seed collected in Inyo County, California. Germination was rapid, and researchers reported a high level of success in getting plants to the nursery stage, but poor levels of success in transferring plants from the nursery to outdoor gardens. Of 287 nursery-grown seedlings planted outdoors between 1937 and 1948, none were alive in 1950. Specimens from only one of the collections flowered, but because of insect damage it failed to produce seed (Everett 1957).
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Olga Kildisheva, Program Manager, The Nature Conservancy and Ann Hild, Professor Emeritus, University of Wyoming.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Association of Official Seed Analysts [AOSA]. 2016. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Beale, D.M.; Smith, A.D. 1970. Forage use, water consumption, and productivity of pronghorn antelope in western Utah. The Journal of Wildlife Management. 34(3): 570-582.
Beatley, J.C. 1976. Vascular plants of the Nevada Test Site and central-southern Nevada: Ecologic and geographic distributions. Springfield, VA: U.S. Department of Commerce, Office of Technical Information, Technical Information Center. 308 p.
Blake, S.F. 1913. A revision of Encelia and some related genera. Proceedings of the American Academy of Arts and Sciences. 49(6): 346-396.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Braun, U.; Mohan, S.K. 2013. New records and new host plants of powdery mildews (Erysiphales) from Idaho and Oregon (USA). Schlechtendalia. 27: 7-10.
Brian, N.J.; Hodgson, W.C.; Phillips, A.M. 1999. Additions to the flora of the Grand Canyon region. II. Journal of the Arizona-Nevada Academy of Science. 32(2): 117-127.
Clark, C. 1998. Phylogeny and adaptation in the Encelia alliance (Asteraceae: Heliantheae). Aliso. 17(2): 89-98.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Cronquist, A. 1972. A new variety of Enceliopsis nudicaulis (Asteraceae) from southern Nevada. Bulletin of the Torrey Botanical Club. 99(5): 246-247.
Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K. 1994. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Volume 5: Asterales. Bronx, NY: The New York Botanic Garden. 496 p.
Curtis, C. 2006. 278. Enceliopsis. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 21 Magnoliophyta: Asteridae, part 8: Asteraceae, part 3 Asterales, part 3 (Aster order). New York, NY: Oxford University Press: 112-113.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Everett, P. 1957. A summary of the culture of California plants at the Rancho Santa Ana Botanic Garden 1927-1950. Claremont, CA: The Rancho Santa Ana Botanic Garden. 223 p.
Fehlberg, S.D.; Ranker, T.A. 2007. Phylogeny and biogeography of Encelia (Asteraceae) in the Sonoran and Peninsular deserts based on multiple DNA sequences. Systematic Botany. 32(3): 692-699.
Gunnell, K. 2017. Great Basin Research Center seed increase. In: Kilkenny, F.; Edwards, F.; Irwin, J.; Barga, S., eds. Great Basin Native Plant Project: 2016 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 87-90.
Harris, J.G. 1983. A vascular flora of the San Rafael Swell, Utah. Great Basin Naturalist. 43(1): 79-87.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
Howe, G.; St, Clair, B.; Bachelet, D. 2017. Seedlot Selection Tool. Corvallis, OR: Conservation Biology Institute. https://seedlotselectiontool.org/sst/
Jensen, S. 2012. Provisional seed zone-based seed increase. In: Shaw, N.; Pellant, M., eds. 2013. Great Basin Native Plant Selection and Increase Project: 2012 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station and U.S. Department of the Interior, Bureau of Land Management: 95-102.
Jensen, S.; Stettler, J. 2012. Applying provisional seed zones to Great Basin forb production and cultural practice notes. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 70-84.
Karrfalt, R.; Vankus, V. 2012. Development of germination protocols, seed weight, purity, and seed conditioning/cleaning protocols for Great Basin grasses and forbs. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Selection and Increase Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 36-39.
Kildisheva, O.A.; Erickson, T.E.; Madsen, M.D.; Dixon, K.W.; Merritt, D.J. 2018a. Seed germination and dormancy traits of forbs and shrubs important for restoration of North American dryland ecosystems. Plant Biology. 21(2019): 458-469.
Lady Bird Johnson Wildflower Center [LBJWC]. 2020. Enceliopsis nudicaulis (A. Gray) A. Nelson. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants-main [Accessed 2020 January 3].
Love, S.L.; Akins, C.J. 2019. Second summary of the native seed germination studies of Norman C Deno: Species with names beginning with letters C through E. Native Plants Journal. 20(1): 65-97.
Mozingo, H.N.; Williams, M. 1980. Threatened and endangered plants of Nevada: An illustrated manual. Portland, OR: US Fish and Wildlife Service. 268 p.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Rawlins, J.K.; Roundy, B.A.; Davis, S.M.; Egget, D. 2012. Predicting germination in semi-arid wildland seedbeds. I. Thermal germination models. Environmental and Experimental Botany. 76 (2012): 60-67.
Reveal, J.L.; Styer, E.L. 1974. Miscellaneous chromosome counts of western American plants-I. The Southwestern Naturalist. 18(4): 397-401.
Rittenhouse, B.; Rosentreter, R. 1994. The autecology of Challis milkvetch, an endemic of east-central Idaho. Natural Areas Journal. 14(1): 22-30.
Sada, D.W. 1990. Recovery plan for the endangered and threatened species of Ash Meadows, Nevada. Reno, Nevada: US Fish and Wildlife Service. 75 p. +Appendices.
Sanders, D.L.; Clark, C. 1987. Comparative morphology of the capitulum of Enceliopsis (Asteraceae: Heliantheae). American Journal of Botany. 74(7): 1072-1086.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Wieland, K.; Shaw, N.; Kilkenny, F. 2019. Native wildflower seed yield in response to modest irrigation. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2018. OSU AES Ext/CrS161. Corvallis, OR: Oregon State University: 134-145.
Shock, C.C.; Feibert, E.B.G.; Saunders, L.D. 2014a. Direct surface seeding strategies for successful establishment of native wildflowers. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2013. OSU AES Ext/CrS149. Corvallis, OR: Oregon State University: 159-165.
Smith, A.D.; Beale, D.M. 1980. Antelope in Utah. No. 80-13. Salt Lake City, UT: Utah Division of Wildlife Resources. 88 p.
Smith, A.D.; Malechek, J.C. 1974. Nutritional quality of summer diets of pronghorn antelopes in Utah. The Journal of Wildlife Management. 38(4): 792-798.
Thorne, R.F. 1982. The desert and other transmontane plant communities of southern California. Aliso. 10(2): 219-257.
Train, P.; Henrichs, J.R.; Archer, W.A. 1941. Medicinal uses of plants by Indian tribes of Nevada: Part 1. Contributions toward a flora of Nevada. No. 33. Washington, DC: U.S. Department of Agriculture, Bureau of Plant Industry, Division of Plant Exploration and Introduction. 199 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Great Basin Native Plant Project [USFS GBNPP]. 2014. Seed weight table calculations made in-house. Report on file. Boise, ID: U.S. Department of Agriculture, Forest Service, Boise Aquatic Sciences Laboratory. Available: https://www.fs.fed.us/rm/boise/research/shrub/Links/Seedweights.pdf
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2019. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2016. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 37 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2017. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
USDI US Fish and Wildlife Service [USFWS]. 1983. Eight more Ash Meadows species proposed as endangered. Endangered Species Technical Bulletin. 8(11): 1-10.
USDI US Fish and Wildlife Service [USFWS]. 2020. Ash Meadows sunray (Enceliopsis nudicaulis var. corrugata) species profile in Environmental Conservation Online System (ECOS). U.S. Fish and Wildlife Service. https://ecos.fws.gov/ecp0/profile/speciesProfile?spcode=Q297 [Accessed 2020 February 12].
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2020. Nakedstem sunray (Enceliopsis nudicaulis). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org/species/nakedstem-sunray-enceliopsis-nudicaulis/