Authorship
Shaw, Nancy; Gucker, Corey
Publication Date
April 2018
Update
April 2024
Nomenclature
Royal penstemon (Penstemon speciosus Douglas ex Lindley) is a member of the family Plantaginaceae (the plantain family), subgenus Penstemon, section Glabri (Rydberg) Pennell (Freeman 2019; ITIS 2024). David Douglas collected the type specimen (now missing) from the banks of the Spokane River, Washington, in 1829 (Crosswhite 1967).
Family
Plantaginaceae – Plantain Family
Genus
Penstemon
Species
speciosus
NRCS Plant Code
PESP (USDA NRCS 2024).
Synonyms
Penstemon speciosum Douglas ex Lindl., P. glaber var. occidentalis A. Gray, P. glaber var. speciosus Regel, P. glaber f. speciosus Voss, P. kennedyi A. Nels., P. speciosus subsp. kennedyi Keck, P. piliferus A.A. Heller, P. speciosus var. piliferus Munz & I.M. Johnston, P. rex Nels. & Macbr, P. perpulcher var. pandus Nels & Macbr., P. pandus Nels. & Macbr., P. deserticola Piper, P. glaber sensu A. Gray, P. glaber var. utahensis sensu Jepson (Cronquist et al. 1984; Freeman 2019; ITIS 2024; USDA NRCS 2024).
Common Names
Royal penstemon, royal beardtongue, sagebrush beardtongue, sagebrush penstemon, showy penstemon (Cronquist et al. 1984; Tilley et al. 2009; ITIS 2024; LBJWC 2023; USDA NRCS 2024).
Chromosome Number
The species is diploid, 2n = 16 (Crosswhite 1967; Hickman 1993).
Hybridization
A hybrid (single plant), possibly derived from hybridization of Penstemon grinnellii (Eastw.) and P. speciosus, was discovered in Ventura County, California and was published as Penstemon x peirsonii Munz & I.M. Johnston (Munz and Johnston 1925). Stevens et al. (2020) report successful production of artificial hybrids: P. ovatus (Douglas ex Hook.) x P. speciosus (Mackaness 1967), a cross with the Flathead Lake complex (P. barbatus [Cav.] Roth x P. strictus [Benth.] materials) (Viehmeyer 1961) and crosses with P. eatonii A. Gray as the male parent.
Distribution
Royal penstemon occurs from the eastern slopes of the Cascade and Sierra Nevada Mountains eastward across central and southeastern Washington, eastern Oregon, and southwestern Idaho, and south through eastern California, Nevada, and northwestern Utah to upper elevations in mountain ranges on the northern edge of the Mojave Desert (Hitchcock and Cronquist 2018; USDA NRCS 2024).
Habitat And Plant Associations
Plants are found in openings in sagebrush (Artemisia spp.), antelope bitterbrush (Purshia tridentata), shadscale (Atriplex confertifolia), pinyon-juniper (Pinus–Juniperus spp.), juniper, and mountain brush communities. It also occurs in clearings in ponderosa pine (P. ponderosa) and subalpine forests (Wright and Mooney 1965; Munz and Keck 1973; Cronquist et al. 1984; Hickman 1993; Strickler 1997).
Elevation
Royal penstemon grows over a broad elevational range of 700 to 11,300 ft (210–3,400 m) (SEINet 2017). In Utah it occurs at elevations from 5,900 to 9,500 ft (1,800-2,900 m) (Stevens et al. 2020).
Soils
Royal penstemon grows on loamy to sandy loam soils that are near neutral to slightly alkaline (Strickler 1997; Lindgren and Wilde 2003). It frequently colonizes eroding slopes and other disturbed sites with well-drained, coarse-textured soils (LBJWC 2023). The species exhibits medium CaCO3 tolerance and soil pH of 6.4-8.0 (USDA NRCS 2024). Wright and Mooney (1965) found royal penstemon populations growing on sandstone and granite but not on dolomite substrates at elevations above 9,000 ft (2,700 m) in bristlecone pine (P. aristida) forests in the White Mountains in California.
Description
Royal penstemon is a highly variable, short-lived, perennial herb that ranges from 3 to 26 (35) in (7-65 [90] cm) tall (Hitchcock et al. 1959; Cronquist et al. 1984; Neese and Atwood 2015; Freeman 2019) and has a rooting depth of 14 in (36 cm) (USDA NRCS 2024). Stems are few to several, basally puberulent to retrorsely hairy or sometimes glabrous, and range from erect to ascending or occasionally decumbent from a branched caudex (Hitchcock et al. 1959; Cronquist et al. 1984; Freeman 2019). Leaves are opposite, entire, rather thick and somewhat leathery, more or less sinuate, and glabrous or occasionally retrorsely hairy (Hickman 1993; Neese and Atwood 2015; Freeman 2019). Basal leaves are clustered, petiolate, somewhat evergreen, linear to lanceolate, obovate, or spatulate, and up to 6 in (15 cm) long and 0.5 (0.8) in (1.2 [2] cm) wide (Hitchcock et al. 1959; Hickman 1993). Cauline leaves are reduced upward, sessile to short petiolate, linear to lanceolate, subcordate, sometimes clasping, less than 2 cm wide, and folded lengthwise or flat (Cronquist et al. 1984; Hickman 1993). Leaves and stems may be purplish tinted.
The inflorescence is an elongate, one-sided or cylindric thyrse, 1 to 14 inches (3-35 cm) long (an inflorescence with an indeterminate main axis and determinate subaxes, Fig. 1). The lower nodes may support thyrsoid branches (Cronquist et al. 1984; Hickman 1993). Flowers are produced in 2 to 14 closely spaced verticillasters (false whorls) along the inflorescence (Strickler 1997). The main floral axis, peduncles and pedicels are glabrous to puberulent and spreading to ascending. Bracts are lanceolate, 0.8 to 3 in (2-8 cm) long, and reduced upwards.

Figure 1. Royal penstemon plant in southeastern Oregon. The inflorescence is a thyrse with flowering indeterminate on the main axis and determinate on the subaxes. Photo: USFS.
Flowers are perfect and ascending to spreading. The five-toothed calyx is cuplike; the segments are lanceolate to ovate with scarious and erose margins and acute to acuminate tips (Cronquist et al. 1984; Freeman 2019). The corolla is bilabiate and tubular, formed from five fused petals. It is 1.0 to 1.5 in (2.5–3.7 cm) long and glabrous or glandular at the base. Rodriguez-Pena and Wolf (2023) found that flower size increased from east to west across the species range. The corolla tube is swollen ventrally, and there are two ventral ridges. The two upper corolla lobes are spreading, and the three lower lobes are spreading to reflexed. Color ranges from bright blue to purplish at the base, sky blue along the limb, and light blue to white in the interior. The nectar guides may be reddish-purple (Freeman 2019; Mansfield 2000; LBJWC 2023). The four fertile stamens reach the apex with the longer pair sometimes extruded. Anther cells are divergent, 1.8 to 3 mm long, and generally glabrous except for the papillate-toothed sutures. At maturity the anther cells dehisce along the distal two-thirds of their length and twist into an S-shape (Hitchcock et al. 1959; Cronquist et al. 1984; Neese and Atwood 2015). Glandular hairs near the bases of the upper stamens produce nectar (Straw 1966). The staminode is 14 to 18 mm long and glabrous or sparsely bearded at the apex. It is attached apically to the roof of the corolla but drops to the floor anterior to the ovary where it may provide some protection from insect herbivory (Straw 1966; Strickler 1997; Freeman 2019).
The fruit is a capsule, 0.2 to 0.6 in (0.6–1.5 cm) long with four valves and tips that spread as it opens apically. It becomes dry and parchment colored at maturity (Fig. 2). Seeds are brown, 2 to 2.5 (3.5) mm long, somewhat elongate and three-angled (Fig. 3; Cronquist et al. 1984; Hickman 1993; Hurd n.d.). The embryo is slightly curved and embedded in living endosperm (Hurd n.d.) (Fig. 4).


Figure 2. Royal penstemon with immature capsules (top) and mature capsule opening apically (bottom; scale in mm). Photos: USFS.

Figure 3. Royal penstemon seeds. Photo: J. Cane, USDA ARS.

Figure 4. Royal penstemon seed: cross section and longitudinal sections. The embryo is surrounded by endosperm and the seed coat. Scale in mm. Photo: USFS.
Reproduction
Royal penstemon reproduces entirely from seed. Seeds are gravity dispersed.
Phenology
Flowering may begin in the first or second year and occurs from May to early September depending on location (Hitchcock et al. 1959; Hickman 1993; Freeman 2019; LBJWC 2023). In California, Nevada, and Oregon, flowering occurs primarily in June and July but in Washington it is primarily from mid-May to late June (Freeman 2019). Flowering is indeterminate with flowers on branches near the base of the inflorescence opening first, but flowering within the side branches is mostly determinate. Flowers are protandrous (anthers mature before the stigmas) (Castellanos et al. 2002). Fruits mature about 5 to 8 weeks after flowering (Parkinson and Debolt 2005).
Castellanos et al. (2002) found the time from flower opening to corolla dehiscence averaged about 4.2 days in a royal penstemon population at 6,600 ft (2,000 m) elevation in the southern Sierra Nevada Mountains of California. They described six stages of floral phenology: flower opening to (1) anterior anther dehiscence (0.2 day), (2) posterior anther dehiscence (0.4 day), (3) style tip bent <45º (1.3 days), (4) style bent >45º (1.6 days), (5) style bent 90º (1.9 days), and (6) corolla abscised (4.2 days). Style bending was assumed to indicate stigma receptivity as it positions the stigma to receive pollen carried by foraging bees or wasps. This occurs after pollen from the same flower has been released, thus encouraging cross pollination (Chari and Wilson 2001). Nectar secretion began with corolla opening, and slowed if not visited by a pollinator. Nectar was replaced within 3 to 4 hours following visitation, indicating some, though variable, homeostatic control (Castellanos et al. 2002). The process was repeated throughout the life of the flower, encouraging repeated visitation as the anthers released pollen. With abundant pollinator activity, Castellanos et al. (2002) predicted that most pollen would be removed by the time the stigmas become receptive and that the stigmas would be loaded rapidly.
Breeding System
Cane (2006) found that royal penstemon set few seeds when pollinators were excluded. Manual pollen transfer more than doubled seed production and insect pollination increased yields by more than five times compared with self-pollination. Royal penstemon flower attributes indicate insect rather than hummingbird pollination: corollas are blue to white, relatively short and wide, and pollen and concentrated nectar are dispensed slowly (Tepedino et al. 2011).
Pollination
Cane and Love (2016) inventoried pollinator abundance and diversity at native forbs used in restoration to estimate their “pollinator friendliness.” Surveys were conducted at four Nevada sites in sagebrush steppe and its ecotones with pinyon-juniper woodlands. Solitary native mason bees (Osmia spp.) comprised about half of the observed pollinators at royal penstemon (Cane and Love 2016), while the solitary pollen wasp (Pseudomasaris vespoides), which may sleep in the flowers at night (Nold 1999), accounted for 19.8% of the observed pollinators (Fig. 5). The remaining pollinators included digger bees (Anthophora spp., 2.7%), bumblebees (Bombus spp., 8.1%), small carpenter bees (Ceratina spp., 0.9%), sweat bees (Halictus spp., 11.7%), mason bees (Hoplitis spp., 1.8%) and sweat bees (Lasioglossum spp., 3.6%). Two mason bees (Osmia brevis and O. penstemonis) and the solitary pollen wasp are pollen specialists (oligoleges) that collect pollen for their offspring from penstemon species. O. brevis, for example, visited at least 42 penstemon species (Tepedino et al. 2011).
In a study of pollen nutrition on bee-plant mutualisms in the eastern Sierra Nevada Mountains and Great Basin, Vaudo et al. (2024) reported that, similar to other penstemon species, royal penstemon produced pollen with a high protein to lipid ratio of 13.32. Pollen nutrition was related to flower morphology with bilateral (e.g. Penstemon) and complex (e.g. many Fabaceae) flowers having higher ratios.

Figure 5. Royal penstemon visited by a solitary pollen wasp. Photo: Jim Cane, USDA ARS.
Compared to the other 16 forbs examined, royal penstemon exhibited moderate abundance and low diversity of pollinators (Cane and Love 2016). Eight genera were observed at royal penstemon compared to a range of 3 to 19 for the other 16 species examined. Abundance of pollinators at royal penstemon was 8.1 bees or wasps per 100 plants compared to 1.4 to 28.4 per 100 plants for the other forbs (Cane and Love 2016). In cultivation, flowers of royal penstemon were avidly sought out by diverse pollen-foraging bumblebees and sometimes by digger bees (J. Cane, USDA ARS, Logan, UT, personal communication, August 2017). Castellanos et al. (2002) noted hummingbird visits to royal penstemon flowers, but concluded they removed little pollen.
Ecology
Royal penstemon is a drought-tolerant species. It requires full sun, at least 110 frost-free days, and 8 to 24 in (203-610 mm) of precipitation (USDA NRCS 2024). It often colonizes steep and eroding slopes, road cuts, rights-of-way, and other disturbances (Tilley et al. 2009). It is adapted to well-drained soils and the low levels of competition encountered on such sites. Flowering royal penstemon in sagebrush communities of southern Idaho has been noted in the year following wildfire (Fig. 6; M. Fisk, USGS, Boise, ID, personal communication, June 2017). Although not a strong soil stabilizer, the species does provide ground cover, and it will reseed and reestablish itself unless more competitive vegetation establishes. Royal penstemon persists in later successional communities and may be found in openings in shrublands and forested areas where adapted.
Seed And Seedling Ecology
Meyer and Kitchen (1994) examined the germination of 36 Penstemon seed collections representing 13 species in section Glabri. These were collected over an broad elevation range in Utah, Nevada, and Idaho. The collections included one royal penstemon population from a site at 5,940 ft (1,810 m) elevation in central Nevada. Seed of this collection incubated at 50 to 68 ºF (10-20 ºC) in light for 4 weeks exhibited high primary dormancy (1% germination). Prechilling at 34 ºF (1 ºC) for 12 weeks to simulate conditions experienced under a snow pack followed by 4 weeks of incubation at 50 to 68 ºF (10-20 ºC) in light increased germination to 70%. Germination increased to 90% with 24 weeks of prechilling (Meyer and Kitchen 1994).

Figure 6. Royal penstemon one year after wildfire. Photo: Matt Fisk, USGS.
For this same seed lot, a 4-week initial incubation of imbibed seeds at 50 to 68 ºF (10-20 ºC) as might occur with fall rains followed by 12 weeks of prechilling resulted in 91% germination (Meyer and Kitchen 1994). Chilling response was not cumulative if a 12-week prechill was interrupted by periods of higher temperatures. This was simulated by 4-week incubations at 50 to 68 ºF (10-20 ºC) after either 4 or 8 weeks of prechilling, as might result from warmer periods in winter. Inadequate or interrupted prechilling in some years might permit some seed to remain dormant and be carried over in the soil seed bank (Meyer and Kitchen 1994).
Germination rate (days to 50% germination) during prechilling for the 13 penstemon species was negatively correlated to habitat as described by the mean January temperature at the collection site (Meyer and Kitchen 1994). Thus seeds of species collected from sites with short winters tended to germinate more rapidly, while seed of species collected from sites with long winters germinated more slowly, possibly timing germination to early spring emergence. Dry afterripening the seed for 1 year had little effect on these chilling responses (Meyer and Kitchen 1994).
When wild-collected seed of royal penstemon and five other section Glabri species were grown in a common garden, the parent populations and their progeny were characterized by generally similar levels of primary dormancy and responses to prechilling, suggesting a genetic basis for these responses (Meyer and Kitchen 1994). The size of the chill-responsive fraction of seeds was the most phenotypically plastic trait.
Disturbance Ecology
Species Interactions. Parkinson et al. (2013) examined competitive interactions of royal penstemon with native grasses or cheatgrass (Bromus tectorum) for 12 weeks in a greenhouse study. When grown alone, royal penstemon biomass was 1.5 ± 0.3 g. Its root system was distributed laterally and vertically with considerable branching throughout. The biomass of royal penstemon was not reduced, but its relative growth rate declined by 25% when grown with a single Sandberg bluegrass (Poa secunda) or squirreltail (Elymus elymoides) plant. When grown with a single cheatgrass plant, the biomass of royal penstemon was reduced by 91% and its relative growth rate by 80% compared to its growth alone. The decreases were attributed to the similar fibrous rooting morphologies of royal penstemon and cheatgrass and the higher growth rate of cheatgrass (Parkinson et al. 2013).
In a field experiment conducted at two sites in southern Idaho, royal penstemon biomass, but not density, was decreased by cheatgrass concurrently seeded at densities as low as 1.3 or 2.6 plants/ft² (14 or 28 plants/m²) (Parkinson 2008). In addition to the above factors, the earlier germination of cheatgrass (fall or early spring) compared to royal penstemon (early April) and the reported concentration of cheatgrass root mass in the upper 8 in (20 cm) of soil in a study by Melgoza et al. (1990) may have contributed to the ability of cheatgrass to compete effectively with the later developing royal penstemon seedlings.
Wildlife And Livestock Use
Wildlife use of royal penstemon is not well documented. Foliage and stems are taken by pronghorn antelope (Antilocapra americana) (Martin 1951). Parkinson (2008) found that Piute ground squirrels (Urocitellus mollis) began consuming emerging royal penstemon seedlings in a southern Idaho seeding study in May. All seedlings were consumed by late July. Royal penstemon seeds and foliage may provide 10 to 25% of the mantled ground squirrel (Callospermophilus lateralis) diet, and they are a food source for the Merriam kangaroo rat (Dipodomys deserti) and other kangaroo rat (Dipodomys spp.) species (Martin 1951). Seeds of Penstemon species are taken by rosy finches (Leucosticte tephrocotis) (Martin 1951).
Fajemisin et al. (1996) reported that royal penstemon constituted 0.1% of the July ground cover on a Wyoming big sagebrush steppe (Artemisia tridentata subsp. wyomingensis) site in south-central Oregon that supported a diversity of grasses and forbs. During a grazing trial, royal penstemon constituted 0.5% of the bites taken by Spanish goats (Capra hircus) in July, during the period of active growth. This figure increased to 2.3% of bites taken in August when forages were cured. This was consistent with the goat’s overall greater preference for forbs over grasses (Fajemisin et al. 1996).
Although the use of royal penstemon by butterflies has not been reported, James and Nunnallee (2011) listed several Penstemon species as hosts for adults, larvae, or egg deposition of Edith’s checkerspot (Euphydryas editha) and snowberry checkerspot (E. colon) in the Pacific Northwest. They also reported that some Penstemon species as larval hosts of anicia checkerspot (Euphydryas anicia) and common buckeye (Junonia coenia) (James and Nunnallee 2011). Arachne checkerspot (Polyadryas arachne monache) has been associated with royal penstemon (Robinson et al. 2023), but its use of the species was not determined.
Horticulture
Royal penstemon can be used in gardens, parks, and urban landscaping to provide striking seasonal color. Tilley et al. (2009) and Way and James (1982) recommended that royal penstemon be treated as a biennial when used as a specimen plant for domestic landscapes. Nold (1999) noted that the lifespan of penstemon species in section Glabri, can be prolonged by providing favorable growing conditions and removing the flowering stalks immediately after flowering. Leaving a few stalks with seed capsules provides for natural reseeding. For domestic plantings it may be necessary to improve drainage of planting beds by adding sand or gravel.
There are no formal horticultural releases of royal penstemon. A dwarf form of the species found above 8,000 ft (2,440 m) in the Sierra Nevada and White Mountains of California is often grown in alpine gardens, and is sometimes referred to as P. speciosus ‘Kennedyi’ in the commercial trade (Way and James 1982; Hickman 1993; Lindgren and Wilde 2003; Stevens et al. 2020). The plants are generally less than 8 in (20 cm) tall with grey-green leaves and large, deep blue flowers (Way and James 1982). Once known as P. kennedyi or P. speciosus subsp. kennedyi, these populations are now included in P. speciosus (Hickman 1993; Lindgren and Wilde 2003).
Revegetation Use
Within its range, royal penstemon can be seeded on road cuts and other unstable sites where it functions as an early seral species. It can be used to add diversity to seed mixes for community restoration following wildfire if competition from cheatgrass and other recovering or seeded vegetation is low during its establishment period. Royal penstemon can also be used in recreation areas and low water-use landscapes to seasonal color and habitat for pollinators and other wildlife. Due to its brief flowering period, royal penstemon should be planted with species having different flowering phenologies to provide sequential pollen and nectar for its non-specialist pollinators, especially bumblebees, which will pollinate other species as well (J. Cane, USDA, ARS Logan, UT, personal communication, August 2017). Seed is commercially available (LBJWC 2023), but the seed source should be checked before purchasing to determine whether it is appropriate for the planting site.
Developing A Seed Supply
For restoration to be successful, the right seed must be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (Young et al. 2003, 2022; UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for royal penstemon, generalized provisional seed zones developed by Bower et al. (2014), may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat:moisture index). In Figure 7, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s Threat and Resource Mapping (TRM) Seed Zone application (USFS WWETAC 2024) provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Restoration Tool (Richardson et al. 2020) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Barga et al. (2020) conducted an analysis of royal penstemon and other common native forb seeds collected through the national Seeds of Success program. A binary species distribution map developed for royal penstemon indicated a range of 243,630 mi2 (631,000 km2), 15.4% of which had been affected by fire between 2000 and 2018. When climatic conditions (annual precipitation, summer precipitation, and annual maximum and minimum temperatures) at the 25 royal penstemon collection sites were compared with conditions at 500 randomly selected points across the distribution map for the species, the collection sites differed from the random points in that their average maximum temperature was greater. Compared to restoration sites within the distribution range, summer precipitation of the collection sites was lower. Selecting future collection sites with climatic conditions comparable to those of areas where burns might be expected was considered critical for meeting future restoration needs in the face of increasing wildfires, weed invasions, and climate change (Barga et al. (2020).
Occurrence Map
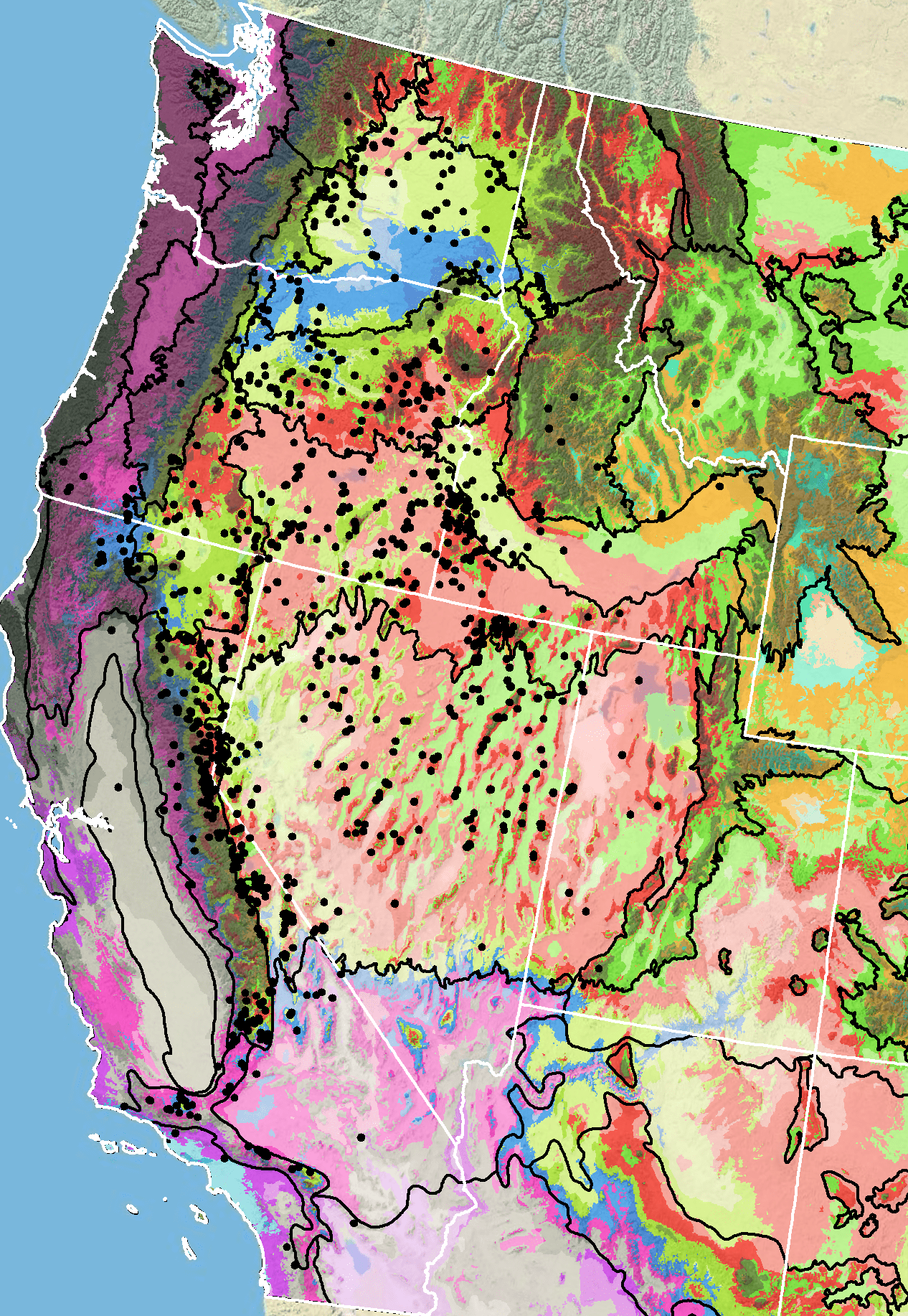
Figure 7. Distribution of royal penstemon (black circles) based on geo-referenced herbarium specimens and observational data from 1881-2016 (CPNWH 2017; SEINet 2017; USDI USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USDA FS WWETAC 2024). Map prepared by M. Fisk, USDI USGS.
Wildland Seed Collection
Seed production in wildland royal penstemon populations varies widely from year to year, but affecting factors have not been examined. Little or no seed may be present in some years. In the northern Great Basin, excellent seed crops have been noted in the year following wildfire (M. Fisk, USGS, Boise, ID, personal communication, June 2017).
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (Young et al. 2003, 2020; UCIA 2015). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Royal penstemon seed is mature and can be harvested when the stalks and capsules are dry and straw colored and the seeds are brown and firm. Seed fill should be checked with a cut or X-ray test. Variation in maturation dates occurs among plants within populations. Seed maturation within inflorescences is indeterminate. However, mature capsules open and seeds are dispersed slowly over time, so it is possible to delay collection until most or all capsules on a stalk or plant have matured. When possible, sites should be checked prior to harvest to determine whether an adequate seed crop is present for collection and to update harvest date predictions. Collection can occur over a 2- to 3-week period depending upon location and weather conditions (Parkinson and DeBolt 2005). Harvest dates for 27 collections made over a 13-year period ranged from July 10, 2012, in Payette County, Idaho, at 2,512 ft (765 m) elevation to two collections made in Harney County, Oregon, on August 16, 2010 at elevations of 2467 ft (752 m) and 4,435 ft (1,352 m) (USDI SOS 2017, 2024).
Collection Methods
Seeds are most easily harvested from wildland stands by clipping mature inflorescences into collection bags, buckets, seed hoppers, or other containers. Gloves should be worn to protect the hands from cuts and splinters because the stalks are brittle. If capsules are opening, care must be taken to avoid seed loss by holding the stalks upright until they are deposited in a container to minimize spillage. Capsules may also be stripped from the flowering stalks. Although this method simplifies seed cleaning, it is a much slower collection process and results in greater seed loss from open capsules. Small quantities of nearly pure seed may be collected by shaking partially open capsules into a container, but this technique, in addition to being extremely time consuming, might reduce genetic diversity if only a fraction of the capsules have opened (M. Fisk, personal communication).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). Collecting protocols and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2023).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2023). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of royal penstemon.
Post-Collection Management
Seed should be kept in a dry, shaded place until collections can be moved to a controlled short-term storage environment. Short-term storage should be dry, cool, and inaccessible to rodents or other seed predators. If insects are suspected in any collection, seed should be frozen for 48 hours or treated with an appropriate insecticide. The more plant material in the collection, the more ventilation and drying a seed lot will likely need (Gold n.d.; Parkinson and DeBolt 2005; Hay and Probert 2011).
Seed Cleaning
Appropriate procedures, equipment, and settings for seed cleaning vary among seed lots of a species depending on the size of the seed lot, the type and amount of debris in the collection, and seed size. Very small seed lots of partially open capsules may be cleaned simply by shaking the seed from the capsules and sieving to remove fine debris (M. Fisk, personnal communication). Small lots can be cleaned using a rubbing board to crush the capsules. The resulting material is then passed through sieves with 1.7 and 1.0mm square openings (nos. 12 and 18 USA STS) (Parkinson and DeBolt 2005). Fine debris is removed using a seed blower.
Larger samples can be cleaned using a hammermill or debearder to break up the capsules and release the seed (Fig. 6). Fine cleaning can then be accomplished using a clipper or seed blower (Meyer 2008). Personnel at the USFS Lucky Peak Nursery (USFS LPN 2017) remove debris using a scalper with a no. 6 top screen and 1/25 bottom screen. Material is then fed through a dewinger with a setting of 4 to move the stalks and capsules into the hopper, 6 for both the drum speed and drum angle. The air is kept closed.
Barner (2007) recommended cleaning royal penstemon seed lots using a Westrup Model LA-H laboratory brush machine with a no. 7 mantel set at a medium speed. Material is then air-screened with an office clipper fitted with a no. 9 round or triangle top screen and a 1/24 round bottom screen set at medium speed with medium air. A seed blower can be used to remove fine debris.
The cleanout ratio (clean seed weight:bulk collection weight) for seed lots of royal penstemon cleaned at the USFS Bend Seed Extractory and seed quality data for these lots are provided in Table 1 (USFS BSE 2017, 2023). Seeds are small, averaging more than 400,000/lb (882,000/kg) (Table 1). Other sources report values that range from 309,800 to 429,000 seeds/lb (683,100-946,000 seeds/kg) (Maquire and Overland 1959; Barner 2007; Shock et al. 2014; USFS NSL 2017; SER, INSR, RBGK 2023).
Seed Storage
Penstemon seeds are orthodox and store well in dry conditions (Meyer 2008). Data is not available for royal penstemon, but Stevens et al. (1981) found that Palmer’s penstemon (P. palmeri) seed germinated to 50% following 15 years of storage in an open warehouse. Barner (2007) recommended storage at 33 to 38 ºF (1-3 ºC) (original germination not provided). Lindgren and Schaaf (2004) monitored germination of four penstemons: foxglove penstemon (P. digitalis), large beardtongue (P. grandiflorus), Rocky Mountain penstemon (P. strictus), and ‘Prairie Splendor’ (cobea beardtongue [P. cobaea] x Heller’s beardtongue [P. triflorus]) for 11 years of storage at 61 to 64 ºF (16-18 ºC) and 35% relative humidity. Emergence percentages were maintained for 4 to 5 years under these conditions.
Seed Testing
Testing methods are available for penstemons. A viability test is often used to provide an estimate of seed quality due to the long stratification requirement for germination.
Viability Testing
Instructions for estimating the viability of family Plantaginaceae species are provided in the AOSA Tetrazolium Testing Handbook (AOSA 2010). Seeds are soaked in water overnight at 68 to 77 oF (20-25 oC), then prepared for staining by cutting them laterally across the distal end of the cotyledons and soaking them overnight in 1.0% tetrazolium chloride (TZ) at 86 to 95 oF (30-35 oC). Stained seeds are prepared for evaluation by cutting them longitudinally and removing the embryo from the seed. Alternatively, imbibed seeds may be prepared by cutting them longitudinally, leaving the distal end intact, and soaking them overnight in 0.1 to 0.5% TZ at 86 to 95 oF (30-35 oC). The two halves are then separated for evaluation. Seeds are scored viable if the embryo and endosperm are evenly stained.
Purity Testing
A minimum sample size of 2.75 g of cleaned royal penstemon seed is recommended for estimating purity and the presence of noxious weeds (USFS NSL 2017). Purity tests are conducted following Association of Official Seed Analysts procedures (AOSA 2016).
Germination Testing
The AOSA Rules for Testing Seed (AOSA 2023 includes a general rule that is used for testing penstemon species lacking specific rules. Two tests are conducted to provide an estimate of viability and nondormant seed:
- Test 1: Seeds (400) are imbibed and chilled in a solution of gibberellic acid (GA3, 500 ppm) solution for 60 days at 36 to 41 ºF (2-5 ºC) then incubated for 14 days at 10 ºC or 50 to 68 ºF (1o-20 ºC). Germinated seeds are counted and nongerminated seeds are tested for viability. The sum of the germinated seed plus nongerminating viale seed is the total viable seed percentage. As a more rapid alternative to this method, 4000 seeds can be tested for viability, which requires about 2 days.
- Test 2: Germinable (nondormant) seed percentage. Seeds are incubated in light without pretreatment for 28 days. The result is reported as percentage germination. Only nondormant seeds will germinate. The difference between the two tests (viability [Method 1] minus germinable seed [Method 2]) is the percentage of dormant seed.
Germination Biology
Germination recommendations prescribe prechilling as a pretreatment to relieve physiological dormancy in royal penstemon. Lindgren and Wilde (2003) recommended an 8 to 12 week prechill. Maquire and Overland (1959) recommended a 1 to 4 week prechill at 37 ºF (3 ºC) followed by incubation at 68 ºF (20 ºC) in the dark.
Wildland Seed Yield And Quality
Table 1. Seed yield and quality of royal penstemon seed lots collected in the Intermountain region, cleaned and evaluated by the Bend Seed Extractory. Viability was tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Characteristic |
Mean |
Range |
Samples (No.) |
|
Bulk weight (lbs) |
1.45 |
0.09-10.56 |
39 |
|
Clean weight (lbs) |
0.33 |
0.0005-3.73 |
39 |
|
Clean-out ratio |
0.20 |
0.01-0.60 |
39 |
|
Purity (%) |
93 |
57-99 |
39 |
|
Fill (%)¹ |
95 |
63-99 |
39 |
|
Viability (%)² |
91 |
76-96 |
15 |
|
Seeds/lb |
444,284 |
231,428-743,590 |
39 |
|
Pure live seeds/lb |
391,058 |
206,314–677,708 |
15 |
¹100 seed X-ray test
²Tetrazolium chloride test
Marketing Standards
Walker and Shaw (2005) recommended a minimum of 95% purity and 66% viability as standards for royal penstemon seed purchases. Acceptable purity, viability, and germination specifications vary with revegetation plans. High purity and viability are required for precision seeding equipment used in nurseries and agricultural seed fields. Lower purity may be acceptable for some wildland seeding techniques.
Agricultural Seed Production
Only limited supplies of royal penstemon are collected from wildland stands. Cultural practices for producing royal penstemon seed have been developed, and seed has been produced commercially (Fig. 8).
Crop Phenology. Adequate flowering for seed harvesting occurred in the first year following seeding at the Oregon State University’s Malheur Experiment Station in eastern Oregon (OSU MES, elevation 2,260 ft [689 m]) annual precipitation 11 in [279 mm]) (Shock et al. 2016). Over a 10-year period (2006 to 2015), flowering was initiated between April 28 and May 25 (mean: May 7), peak flowering occurred between May 5 and 30 (mean: May 17, 7 yrs of data), flowering was completed between May 30 to June 30 (mean: June 17), and seed was harvested between June 30 and July 29 (mean: July 15) (Shock et al. 2016).
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2003, 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Weed Management
Weed control was accomplished at OSU MES primarily by hand rouging and cultivation (Shock et al. 2015b). Research was conducted to identify herbicides effective for weed control and plant tolerance on local soils (Shock et al. 2007, 2008, 2009a, 2009b, 2010a, 2010b, 2011). Herbicides are not registered for this species, and the results do not constitute an endorsement of specific companies or products or recommendations for use. Results of this research, however, could contribute to future registration efforts.
Preemergent herbicide trial results (Shock et al. 2008, 2011) indicated that royal penstemon plant stand was not reduced by trifluralin (0.375 lb ai/ac), benefin (1.2 lb ai/ac), dimethenamid (0.656 lb ai/ac), or linuron (1.0 lb ai/ac) applied in fall (late November) after seeding. Plant stand was reduced by bensulide (5 lb ai/ac), pronamide (1.0 lb ai/ac), and pendimethalin (0.75 lb ai/ac). Considerable plant injury resulted from pendimethalin and to a lesser extent from benefin applications. A follow-up study indicated that activated charcoal (Gro-Safe®, Norit Americas, Atlanta, GA, 48 lbs/ac) applied over the seed rows protected emerging royal penstemon from pendimethalin (0.75 lb ai/ac) when used as a preemergent compared to its application over rows without charcoal. Royal penstemon emergence was 34.8% with charcoal compared to 3.3% without) (Shock et al. 2011).
Seven herbicides were evaluated for post-emergent weed control with treatments repeated annually from 2006, the establishment year, through 2008 (Shock et al. 2009b, 2010b, 2011). In 2006 plant stand was not reduced by any of the herbicides compared to the untreated check. Extensive plant injury resulted from prometryn (0.8 lb ai/ac) and lesser amounts from linuron (0.5 lb ai/ac) applications. Clethodim (0.125 ai/ac), oxyfluorfen (0.125 lb ai/ac), clethodim + surfactant (0.094 lb ai/ac + 1% v/v), and dimethenamid-P (0.656 lb ai/ac) caused only limited damage while pendimethalin (1.0 lb ai/ac) damage was marginal. Relative to the check, seed yields were reduced by octanoic acid ester of bromoxynil, oxyfluorfen, prometryn, and linuron applications in 2007, but none of the herbicides reduced seed yields in 2008.
Promising herbicides for development of a weed control program for native forbs were further tested at various rates and combinations (Shock et al. 2009a, 2010a, 2011). These included: 1) Clethodim (0.094 lb ai/ac); 2) Separate treatments with dimethenamid-P (0.84 or 0.98 lb ai/ac) or pendimethalin (0.95, 1.19 or 1.43 lb ai/ac), both broad spectrum soil-active herbicides; and 3) Combinations of pendimethalin at 0.95 or 1.19 lb ai/ac with dimethenamid-P at 0.66, 0.84, or 0.98 lb ai/ac. Seed yield of all treatments was similar to the control.
Establishment And Growth
Studies begun in 2005 by Shock et al. (2015a) examined cultural practices for seed production of royal penstemon at OSU MES located in southeastern Oregon (elevation: 2,260 ft [689 m], mean annual precipitation: 11 in [279 mm]. Stands were seeded in fall (late October or November) to provide for overwinter cool prechilling and early spring seedling emergence.
Establishment studies initiated in 2012 and 2013 examined seven combinations of seed treatment with fungicides (Thiram and Captan), seed covers (sawdust, sand) and mulch (N-sulate, a plastic mulch; hydroseeding, a paper mulch) (Shock et al. 2014, 2015a). Seeds were planted at a rate of about 30/linear ft (76/linear m) in November of 2012 and 2013 to provide for overwinter cool prechilling and early spring seedling emergence.
In 2013 establishment was improved (P < 0.05) relative to the control (0.1% stand) only with combinations that included row cover and fungicide seed treatment. The addition of sawdust or sawdust and sand did not result in further significant increases. The range in establishment following these treatments was 27 to 33.1% (Shock et al. 2014). In April 2014, only the combination of row cover, fungicide seed treatment, sawdust and sand (12.2% stand) improved stand compared to the control (0.7% stand) (P < 0.05) for plots planted in fall 2013 (Shock et al. 2015a).

Figure 8. Royal penstemon irrigation test plots at Oregon State University, Malheur Experiment Station, Ontario, Oregon. Photo: USFS.
At the Natural Resources Conservation Service Aberdeen Plant Material Center, in southeastern Idaho, seed fields of royal penstemon were established from greenhouse-grown container seedlings (Tilley et al. 2009). Weed barrier fabric was used to reduce weed problems. Seedlings were planted in holes 3 to 4 in (8-10 cm) in diameter spaced 9- to 16-in (23-41 cm) apart.
Irrigation
Irrigation requirements for royal penstemon were examined at OSU MES using subsurface drip irrigation (Shock et al. 2015b). This system provides precise application of water directly to the root system and away from crown tissues. This minimizes water use and reduces water availability to weed seeds. Drip lines were placed 12 in (30 cm) deep between alternate rows (30-in [76-cm] row spacing). Zero, 4, or 8 in (0, 100, 200 mm) of supplemental water was added in four equal applications at 2-week intervals beginning with flowering. From 2006 to 2015, irrigation began between April 19 and May 20 (mean: May 5) and ended between June 3 and July 5 (mean: June 18). Seed yield of royal penstemon demonstrated a quadratic response to irrigation with the maximum response with 5 in (130 mm) of irrigation. Irrigation of 4 to 8 in (100-200 mm) supplemental water was recommended for warm, dry years and none for cool, wet years (Shock et al. 2016).
Pollinator Management
In cultivated gardens, four bumblebees: Great Basin bumblebee (B. centralis), yellow bumblebee (B. fervidus), brown-belted bumblebee (B. griseocollis), and Hunt’s bumblebee as well as a digger bee, all generalist (polylectic) bees, were observed to sonicate (buzz pollinate) royal penstemon flowers for pollen (Fig. 9; Cane and Love 2016). Honey bees (Apis mellifera) placed within a royal penstemon plot did not visit its flowers. Because of the high cost of commercial bumblebee colonies, locating royal penstemon seed production fields near suitable habitat or creating suitable habitat for unmanaged bumblebees and ground nesting bees may be required to insure pollination. Careful use of insecticides and restriction of cultivation during the period of bee nesting is recommended (Cane 2008).

Figure 9. Royal penstemon flower pollinated by a bumblebee (Bombus spp.). Photo: Jim Cane, USDA ARS.
Pest Management
Two lygus bugs, (western tarnished plant bug [Lygus hesperus] and tarnished plant bug [L. lineolaris]) will attack royal penstemon (Hammon and Franklin 2012, Hammon 2014). Lygus bugs have sucking mouth parts and can cause extensive loss of flowers, developing seeds, and leaves. Black tar-like drops (penstemon tears) (Fig. 10) are produced near royal penstemon flower buds, stems, and leaf axils within a day of feeding. Because the bugs feed on flowers and developing ovaries, developing fruit is lost. This is not always apparent and results in low seed yields rather than lowered seed quality.
Lygus bugs are common in alfalfa (Medicago sativa) fields and on smotherweed (Bassia spp.) and other weeds (Hammon 2014). They can be expected to migrate into nearby fields or native populations of penstemon following alfalfa harvest. Hammon (2014) reports that sweeps capturing one or more immature lygus bugs during the pre-bloom stage followed by increased catch sizes over time can indicate a need for control measures in valuable crops such as penstemon. Several insecticides available for control of lygus bugs in alfalfa can be applied to royal penstemon before or during bloom (Hammon 2014). Pyrethroids applied pre-bloom provide residual control of immigrating lygus bugs without harming pollinators. Naled, an organophosphate, when applied at night, will kill insects, but has a short residual. Novaluron, a growth regulator, affects immature insects as they molt, but will not affect adult insects. Prompt and appropriate timing of applications is critical to effectively control lygus bugs, protect pollinators, and provide for human safety. Local individuals with expertise in producing alfalfa seed can advise on herbicide selection and treatment timing (Hammon 2014).
At the OSU MES, the first hatch of lygus bugs appears with the accumulation of 250 degree-days (52 °F [11 °C] base) (Shock et al. 2015b). From 1995 to 2010, the average first hatch date was May 18, which coincided with the period of early to peak flowering for royal penstemon. Prolonged infestation and low seed set resulted from an early hatch in 2007 that was not adequately controlled by azadirachtin treatment at 0.0062 lb ai/ha, applied in May. Bifenthrin at 0.1 lb/ac and acephate at 8 oz/ac (a contact and residual systemic) were applied during flowering or pre-flowering in subsequent years to control lygus bugs (Shock et al. 2016).
Royal penstemon has been associated with a fungus, Ramularia nivosa (Farr and Rossman 2023), an ascomycete fungus and plant pathogen. Negative impacts of the fungus on royal penstemon have not been reported.

Figure 10. Royal penstemon “tears.” These are black tar-like drops produced on stems, leaves, and flower buds following lygus bug predation. Photo: USFS.
Seed Harvesting
Seed was harvested at the OSU MES with a Wintersteiger Nursery Master small-plot combine (Salt Lake City, UT) with an alfalfa seed concave (Shock et al. 2015b). Seed was threshed by the combine and further cleaned with a clipper seed cleaner.
Yields from small plots over 10 years averaged 108.3 lbs of cleaned seed/ac (121.4 kg/ha) with no irrigation, 177.8 lbs/ac (199.3 kg/ha) with 4 in (100 mm) of applied water, and 151.1 lbs/ac (169.4 kg/ha) with 8 in (200 mm) of applied water (Shock et al. 2016). In 2007 yields were drastically reduced by lygus bug infestations. Poor vegetative growth in 2007 and 2009 resulted in very low seed set, and root rot negatively affected plants receiving the higher irrigation rates. Natural reseeding occurred within plots and increased subsequent production. However, if natural reseeding in an agricultural seed field exceeds certification limits and is not controlled in any year, the generation designation for the field must be increased. Based on 10 years of data, Shock et al. (2016) estimated stand life at 3 years with expected seed yields of 25 to 350 lbs/ac (30–390 kg/ha).
Nursery Practice
Nursery stock of royal penstemon has largely been produced for domestic horticulture and research. Seedlings could also be used to improve aesthetics in parks, along roadways, and other low-maintenance landscaping. Container seedlings of native wildflowers are beginning to see increased use for forb island plantings on wildland sites (Landeen et al. 2021). Forb islands are clusters of plants planted in favorable locations where site preparation and management are most feasible. The aim is to provide for eventual spread from limited supplies of expensive seed.
Seed used for production of container stock must first be prechilled to release dormancy. Alternatively, dormancy can be relieved by watering with 250 ppm gibberellic acid (Tilley et al. 2009). Pretreated seeds or germinants can be planted in containers filled with standard soil mixes. Cropping time is about 3 months. Seedlings can also be propagated from semi-woody cuttings (LBJWC 2023). Detailed information on propagation of native plants can be found in Landis et al. (1989–2010) and Dumroese et al. (2012). Seedlings should be carefully acclimated before outplanting. Stock may also be held outdoors until needed.
Parkinson and Debolt (2005) propagated seedlings using seed from a Malheur County, Oregon, seed source (3,870 ft [1,180 m] elevation; seed quality: 73% viability, 90% purity). Seeds were imbibed in 250 ppm gibberellic acid (GA3) for 24 hours, then chilled at 39 °F (4 °C, dark) for 60 days. Mold was treated with thiram. Prechilled seeds were then incubated in a germinator at 70 °F (21 °C, 12 hrs light/12 hrs dark, cool-white fluorescent light). Germination began after 3 days and was complete (33%) after 4 days. Germinants were sown 0.25 to 0.5 cm deep in styrofoam contetainers (50:50 peat:vermiculite mixture) and grown at a constant 81 °F (27 °C). Seedlings were irrigated when soil saturation fell below 80% and fertilized periodically. Of the emergents, 26% developed into healthy seedlings. A longer prechill was recommended for future efforts.
Wildland Seeding And Planting
Careful planning is required to develop seed supplies of royal penstemon for use in native seedings. Sources can be developed by harvesting seed from a provisional seed zone (Bower et al. 2014) or other area of interest. Seed increase via contracting for production in agricultural fields is generally required as adequate seed cannot be harvested from wildland populations, which are often limited in size and should not be over collected. Increased seed can be used directly on planned projects or stored for inclusion in seed mixes for post-fire or other unforeseen disturbances within the collection area. Past wildfire and seeding records for individual provisional seed zones can be used to aid in estimating future seed needs.
Because of the low competitive ability of royal penstemon with cheatgrass (Parkinson 2008, Parkinson et al. 2013), it should be seeded in areas where cheatgrass seed density has been reduced by wildfire or other treatments and is not expected to recover rapidly (Ott et al. 2016b). To avoid competition with more rapidly growing seeded grasses and forbs having similar root morphologies, royal penstemon should be seeded in separate rows or areas, either alone or with other slow growing species with different rooting habits (Fig. 11; Parkinson 2008). This would reduce spatial overlap and interspecific competition for resources while increasing the diversity of species and life forms. Cane et al. (2007) suggested that royal penstemon pollinators Osmia brevis and O. penstemonis may have shallow underground nests, in which case they and the surface nesting Pseudomasaris vespoides would be susceptible to soil heating by wildfires (Cane and Neff 2011) or soil disturbance resulting from mechanical site preparation practices. Loss of specialist pollinators would restrict seeding of royal penstemon to areas near native vegetation that are likely to harbor pollinators (Cane and Love 2016).

Figure 11. Royal penstemon in a post-fire seeding study plot in southern Idaho. Photo: USFS.
Fall seeding is essential to provide the prechilling required to release seed dormancy (Meyer and Kitchen 1994). The small seeds require near surface planting and firming into the soil. This can be accomplished using a Brillion-type seeder, imprinter units, or other equipment modifications that preclude planting the seeds too deep (Ott et al. 2016b). Seeding rates depend on seed availability and the nature of the seeding mix, specifically the number of other forbs included and the desired composition of the resulting community (Ott et al. 2016b).
Ott et al. (2016a, 2017) examined the use of two rangeland drills (minimum-till and conventional) for seeding diverse native seed mixes after fire in Wyoming big sagebrush communities in the northern Great Basin. Larger seeds (primarily grasses) were seeded in alternate rows in each plot. Grasses, forbs, and shrubs with small seeds were either: 1) concurrently drill seeded in the remaining rows, 2) hand broadcast immediately following fall seeding of the large-seeded species, or 3) hand broadcast in mid-winter. Broadcasting simulated aerial seeding. Royal penstemon was seeded at a rate of 1.4 pure live seeds (PLS)/ft² (15 PLS/m²) with other small-seeded species at one site in southern Idaho. For plots where small-seeded species were drill seeded, the disks of each drill were lifted and the seed was broadcast along the row through the seed drops. The minimum–till drill pressed the seeds into the surface with patterned imprinter wheels. The conventional drill covered the seed with drag chains. After 2 years, establishment of royal penstemon was greater when seeded with the minimum-till drill (0.06 ± 0.01 seedling/ft² [0.7 ± 0.1 seedlings/m²]) rather than the conventional drill (0.04 ± 0.01seedlings/ft² [0.4 ± 0.1 seedlings/m²]). Drill seeding was more effective than hand broadcast seeding in fall or winter. Broadcast seeding in fall was more effective than broadcast seeding in mid-winter (J. Ott, USFS Rocky Mountain Research Station, data on file, June 2017).
The effects of seeding treatments and environmental factors were examined for 20 native forb species in two years (2013 and 2014) at three sites (Wells and Orovada, Nevada, and Fountain Green, Utah) (Jensen et al. 2022). Treatments combinations were seeding depth (1.4, 2.6, 3.6 or 4 cm) and row cover (present or absent). Royal penstemon emergence exceeded 3% in 7 of the 12 year x site x row cover treatments, and averaged 1.5% for all treatments. Emergence improved by 78.1% with row cover at Wells in 2013. At Orovada, emergence occurred only under row cover in both years. Increasing seeding depth improved emergence at Orovada in 2013 and 2014, possibly due to the higher temperatures and sandier soils at this site. Increasing seeding depth reduced emergence at Wells and Fountain Green in 2013 (Jensen et al. 2022).
Seedlings emerge in mid-spring and develop a rosette of leaves in the first season (Fig. 12). In southern Idaho, following a cool, moist spring, some seeded plants flowered in their first season (N. Shaw, USFS, Boise, ID, data on file). Plants are not long-lived (Shock et al. 2016), but spread from seed may occur if competition from other vegetation is not excessive. Management to control seedling predation may be required, and livestock grazing should be deferred until all seeded species and recovering native plants have reached reproductive maturity. Monitoring can provide valuable data for improving management and strategies for future seedings.
If seed supplies are limited, royal penstemon seed should be planted only in the most favorable sites for establishment. As an alternative, seedlings can be planted in spots prepared to limit competition. Initial watering may be required for planted seedlings if the soil is dry.

Figure 12. Royal penstemon seedling emerging from a post-fire seeding in southern Idaho. Photo: USFS.
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI Bureau of Land Management Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. We thank Jim Cane, USDA ARS and Clint Shock, Oregon State University Malheur Experiment Station for reviews of this chapter. Marie Tolliver, USFS Lucky Peak Nursery, kindly provided seed cleaning methodology.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Association of Official Seed Analysts [AOSA]. 2023. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Barga, S.C.; Olwell, P.; Edwards, F.; Prescott, L.; Leger, E.A. 2020. Seeds of Success: A conservation and restoration investment in the future of U.S. lands. Conservation Science and Practice. 2(7): e209.
Barner, J. 2007. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Penstemon speciosus. Dougl. ex Lindl. Seeds. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://NativePlantNetwork.org [Accessed 2017 June 16].
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Cane, J.H. 2006. Pollinator and seed predator studies. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2005 progress report. Boise, ID: USDA Forest Service, Rocky Mountain Research Station: 42-44.
Cane, J.H. 2008. Pollinating bees crucial to farming wildflower seed for U.S. habitat restoration. In: James, R.; Pitts-Singer, T., eds. Bees in agricultural ecosystems. Oxford, UK: Oxford University Press: 48-64.
Cane, J.H.; Griswold, T.; Parker, F.D. 2007. Substrates and materials used for nesting by North American Osmia bees (Hymenoptera: Apiformes: Megachilidae). Annals of Entomology. 100(3): 350-358.
Cane, J.H.; Love, B. 2016. Floral guilds of bees in sagebrush steppe: Comparing bee usage of wildflowers available for postfire restoration. Natural Areas Journal. 36(4): 377-391.
Cane, J.H.; Neff, J.L. 2011. Predicted fates of ground-nesting bees in soil heated by wildfire: Thermal tolerances of life stages and a survey of nesting depths. Biological Conservation. 144(2011): 2631-2636.
Castellanos, M.C.; Wilson, P.; Thomson, J.D. 2002. Dynamic nectar replenishment in flowers of Penstemon (Scrophulariaceae). American Journal of Botany. 89(1): 111-118.
Chari, J.; Wilson, P. 2001. Factors limiting hybridization between Penstemon spectabilis and P. centranthifolius. Canadian Journal of Botany. 79(12): 1439-1448.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K. 1984. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Vol. 4: Subclass Asteridae (except Asteraceae). New York, NY: The New York Botanical Garden. 573 p.
Crosswhite, F.S. 1967. Revision of Penstemon Section Habroanthus (Scrophulariaceae). II. Series Speciosi. American Midland Naturalist. 77(1): 12-27.
Dumroese, R. K.; Landis, T.D.; Luna, T. 2012. Raising native plants in nurseries: Basic concepts. Gen. Tech. Rep. RMRS-GTR-274. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. 84 p.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Fajemisin, B.; Ganskopp, D.; Cruz, R.; Vavra, M. 1996. Potential for woody plant control by Spanish goats in the sagebrush steppe. 20(3): 229-238.
Farr, D.F.; Rossman, A.Y. 2023. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://fungi.ars.usda.gov/
Freeman 2019. Penstemon. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 17. Magnoliophyta: Tetrachondraceae to Orobanchaceae. New York, NY: Oxford University Press: 82-202.
Gold, K. n.d. Post-harvest handling of seed collections. Technical Information Sheet 04. UK: Royal Botanic Gardens Kew and Millennium Seed Bank Partnership. 4 p.
Hammon, B. 2014. Pests affecting native plant seed production. Grand Junction, CO: Colorado State University Extension, Tri-Rivers Area. http://www.wci.colostate.edu/shtml/Lygus.shtml.
Hammon, R.; Franklin, M. 2012. Some insects affecting Penstemon seed production. Native Plants Journal. 13(2): 107-110.
Hay F.R.; Probert, R.J. 2011. Chapter 20: Collecting and handling seeds in the field. In: Guarino, L.; Ramanatha, V.; Goldberg, E. Collecting plant genetic diversity: Technical Guidelines-2011 update. Rome, Italy: Bioversity International. 33 p.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
Hitchcock, C.L.; Cronquist, A. 2018. Flora of the Pacific Northwest: An illustrated manual. Second Ed. Giblin, D.E.; Legler, B.S.; Zika, P.F.; Olmstead, R.G., eds. Seattle, WA: University of Washington Press. 882 p.
Hitchcock, C.L; Cronquist, A.; Ownbey, M. 1959. Part 4: Ericaceae through Campanulaceae. In: Hitchcock, C.L.; Cronquist, A; Ownbey, M.; Thompson, J.W. Vascular plants of the Pacific Northwest. Seattle, WA: University of Washington Press: 510 p.
Hurd, E.G. n.d. Tetrazolium test for selected species. Unpublished report on file with the USDA Forest Service, Rocky Mountain Research Station, Boise, ID. 30 p.
ITIS Database. 2024. Integrated Taxonomic Information System. Available: http://www.itis.gov/index.html
James, D.G.; Nunnallee, D. 2011. Life histories of Cascadia butterflies. Corvallis, OR: Oregon State University Press. 447 p.
Jensen, S.; Christensen, W.F.; Roundy, B.; Anderson, V.J.; Kitchen, S.G.; Allphin, L. 2022. Temporal and spatial factors influence native forb emergence more than sowing depth. Rangeland Ecology and Management. 83: 41-49.
Keck, D.D. 1937. Studies in Penstemon V. The section Peltanthera. The American Midland Naturalist. 18: 790-829.
Lady Bird Johnson Wildflower Center [LBJWC]. 2023. Penstemon speciosus Douglas ex Lindl. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center, The University of Texas at Austin. https://www.wildflower.org/plants-main [Accessed 2024 March 15].
Landeen, M.; Jones, C.; Jensen, S.; Whittaker, A.; Summers, D.D.; Eggett, D.; Petersen, S.L. 2021. Establishing seed islands for native forb species on rangelands using N-Sulate ground cover fabric. Native Plants Journal. 22(1): 51-63.
Landis T.D. et al. 1989-2010. The container tree nursery manual. Volumes 1-7. Agriculture Handbook 674. Washington, DC: U.S. Department of Agriculture, Forest Service.
Lindgren, D.; Wilde, E. 2003. Growing penstemons: Species, cultivars and hybrids. American Penstemon Society. Haverford, PA: Infinity Publishing Company. 151 p.
Lindgren, D.T.; Schaaf, D. 2004. Influence of seed stratification and seed age on emergence of Penstemon. HortScience. 36(6): 1385-1386.
Mackaness, R. 1961. Reports on behavior of penstemons in gardens: Mrs. Frank. G. Makaness (Faith), Troutdale, Oregon. Bulletin of the American Penstemon Society. 20: 96-97.
Mansfield, D.H. 2000. Flora of Steens Mountain. Corvallis, OR: Oregon State University Press. 410 p.
Maquire, A.D.; Overland, A. 1959. Laboratory germination of seeds of weedy and native plants. Washington State Agricultural Experiment Station Circular 349. Pullman, WA: Washington State University. 15 p.
Martin, A.C.; Zim, H.S.; Nelson, A.L. 1951. American wildlife and plants: A guide to wildlife food habits. New York, NY: Dover Publications. 500 p.
Melgoza, G.; Nowak, R.S.; Tausch, R.J. 1990. Soil water exploitation after fire: Competition between Bromus tectorum (cheatgrass) and two native species. Oecologia. 83(1): 7-13.
Meyer, S.E. 2008. Penstemon Schmidel; penstemon, beardtongue. In: Bonner, F.T.; Karrfalt, R.P., eds. The woody plant seed manual. Agriculture Handbook 727. Washington, DC: U.S. Department of Agriculture, Forest Service: 774-777.
Meyer, S.E.; Kitchen, S.G. 1994. Habitat-correlated variation in seed germination response to chilling in Penstemon Section Glabri (Scrophulariaceae). The American Midland Naturalist. 132(2): 349-365.
Munz, P.A.; Johnston, I.M. 1925. Miscellaneous notes on plants of southern California-IV. Bulletin of the Torrey Botanical Club. 52(5): 221-228.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Neese, E.C.; Atwood, N.D. 2015. Penstemon Mitchell. In: Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University: 673-693.
Nold, R. 1999. Penstemons. Portland, OR: Timber Press. 259 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Ott, J.; Cox, R.D.; Shaw, N.L. 2017. Comparison of postfire seeding practices for Wyoming big sagebrush. Rangeland Ecology & Management. 70(5): 625-632.
Ott, J.; Halford, A.; Shaw, N. 2016b. Seeding techniques for sagebrush community restoration after fire. Great Basin Factsheet Series Number 14. Sage Grouse Initiative. 6 p.
Ott, J.E.; Cox, R.D.; Shaw, N.L.; Newingham, B.A.; Ganguli, A.C.; Pellant, M.; Roundy, B.A.; Eggett, D.L. 2016a. Post-fire drill-seeding of Great Basin plants: Effects of contrasting drills on seeded and nonseeded species. Rangeland Ecology and Management. 69(5): 373-385.
Parkinson, H.; DeBolt, A. 2005. Propagation protocol for production of container (plug) Penstemon speciosus Dougl. Plants USDA FS – Rocky Mountain Research Station, Boise, Idaho. In: Native Plant Network. US Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. https://NativePlant Network.org [Accessed 2024 April 3].
Parkinson, H.; Zabinski, C.; Shaw, N. 2013. Impact of native grasses and cheatgrass (Bromus tectorum) on Great Basin forb seedling growth. Rangeland Ecology and Management. 66(2): 174-180.
Parkinson, H.A. 2008. Impacts of native grasses and cheatgrass on Great Basin forb development. Bozeman, MT: Montana State University. Thesis. 73 p.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Robinson, G.S.; Ackery, P.R.; Kitching, I.; Beccaloni, G.W.; Hernández, L.M. 2023. HOSTS: A Database of the World’s Lepidopteran Hostplants [Data set]. Natural History Museum. https://doi.org/10.5519/havt50xw
Rodriguez-Pena, R.A.; Wolfe, A.D. 2023. Flower morphology variation in five species of Penstemon (Plantaginaceae) displaying Hymenoptera pollination syndrome. Botanical Sciences. 101: 217-232.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shock, C.; Feibert, E.; Saunders, L.; Parris, C.; Shaw, N. 2011. Evaluation of herbicides for weed control in forb seed production. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2010. OSU AES Ext/CrS 132. Corvallis, OR: Oregon State University: 186-198.
Shock, C.C.; Feibert, E.; Shaw, N. 2009a. Native perennial forb tolerance to rates and mixtures of postemergence herbicides. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2008. Special Report 1094. Corvallis, OR: Oregon State University: 177-179.
Shock, C.C.; Feibert, E.; Shaw, N. 2010a. Native perennial forb tolerance to rates and mixtures of postemergence herbicides, 2009. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2009. OSU AES Ext/CRS 131. Corvallis, OR: Oregon State University: 210-212.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D. 2015a. Direct surface seeding strategies for emergence of native plants in 2014. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2014. OSU AES Ext/CrS152. Corvallis, OR: Oregon State University: 275-279.
Shock, C.C.; Feibert, E.B.G.; Saunders, L.D.; Shaw, N. 2014. Direct surface seeding systems for successful establishment of native wildflowers. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2013. OSU AES Ext/CrS149. Corvallis, OR: Oregon State University: 159-165.
Shock, C.C.; Feibert, E.B.G.; Shaw, N.L.; Shock, M.P.; Saunders, L.D. 2015b. Irrigation to enhance native seed production for Great Basin restoration. Natural Areas Journal. 35(1): 74-82.
Shock, C.C.; Ishida, J.; Feibert, E. 2008. Native wildflowers grown for seed production show tolerance to conventional postemergence herbicides. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2007. Special Report 1087. Corvallis, OR: Oregon State University: 197-203.
Shock, C.C.; Ishida, J.; Feibert, E.; Shaw, N. 2009b. Native perennial forb tolerance to repeated annual applications of postemergence herbicides. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2008. Special Report 1094. Corvallis, OR: Oregon State University: 180-184.
Shock, C.C.; Ishida, J.; Feibert, E.; Shaw, N. 2010b. Native perennial forb tolerance to repeated annual applications of postemergence herbicides, 2009. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2009. OSU AES Ext/CRS 131. Corvallis, OR: Oregon State University: 213-217.
Shock, C.C.; Ishida, J.; Ransom, C. 2007. Tolerance of seven native forbs to preemergence and postemergence herbicides. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2006. Special Report 1075. Corvallis, OR: Oregon State University: 21-32.
Shock, CC.; Feibert, E.B.G.; Rivera A.; Saunders, L.D.; Shaw, N.; Kilkenny, F. 2016. Irrigation requirements for seed production of five native Penstemon species in a semi-arid environment. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2015, Ext/CrS 156. Corvallis, OR: Oregon State University: 211-222.
Society for Ecological Restoration; International Network for Seed Based Restoration; Royal Botanic Gardens [SER, INSR, RBGK]. 2023. Seed Information Database (SID). Available from: https://ser-sid.org/
Stevens, M.R.; Love, S.L.; McCammon, T. 2020. The heart of Penstemon country: A natural history of penstemons in the Utah region. Helena, MT: Sweetgrass Books. 393 p.
Stevens, R.; Jorgensen, K.R.; Davis, J.N. 1981. Viability of seed from thirty-two shrub and forb species through fifteen years of warehouse storage. Great Basin Naturalist. 41(3): 274-277.
Straw, R.M. 1966. A redefinition of Penstemon (Scrophulariaceae). Brittonia. 18(1): 80-95.
Strickler, D. 1997. Northwest penstemons. Columbia Falls, MT: The Flower Press. 191 p.
Tepedino, V.J; Griswold, T.L.; Freilich, J.E.; Shephard, P. 2011. Specialist and generalist bee visitors of an endemic beardtongue (Penstemon caryi: Plantaginaceae) of the Big Horn Mountains, Wyoming. Western North American Naturalist. 71(4):523-528.
Tilley, D.; Ogle, D.; St. John, L.; Shaw, N. 2009. Plant guide: Royal penstemon (Penstemon speciosus Douglas ex Lindl.). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 3 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2023. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service Bend Seed Extractory.
USDA Forest Service, Lucky Peak Nursery [USFS LPN]. 2017. Seed cleaning protocols. Boise, ID: U.S. Department of Agriculture, Forest Service, Lucky Peak Nursery. Unpublished report.
USDA Forest Service, National Seed Laboratory [USFS NSL]. 2017. Great Basin Native Plant Project Penstemon speciosus. Dry Branch, GA: U.S. Department of Agriculture, Forest Service, National Seed Laboratory. https://www.fs.usda.gov/nsl/ Penstemon_speciosus_seed_weights.pdf. [Accessed 2017 June 16].
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2024. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2024. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2023. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 45 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2024. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2017. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Vaudo, A.D.; Dyer L.A.; Leonard, A.S. 2024. Pollen nutrition structures bee and plant community interactions. PNAS. 121(3): e2317228120.
Viehmeyer, G. 1961. Penstemon in your garden. Station Circular 105. Lincoln, NE: University of Nebraska College of Agriculture, Agricultural Experiment Station. 27 p.
Walker, S.C.; Shaw, N.L. 2005. Current and potential use of broadleaf herbs for reestablishing native communities. In: Shaw, N.L.; Pellant, M.; Monsen, S.B., comps. Sage-grouse habitat restoration symposium proceedings; 2001 June 4-7; Boise, ID. RMRS-P-38. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 56-61.
Way, D.; James, P. 1982. The gardeners guide to growing penstemons. Portland, OR: Timber Press. 160 p.
Wright, R.D.; Mooney, H.A. 1965. Substrate-oriented distribution of bristlecone pine in the White Mountains of California. The American Midland Naturalist. 73(2): 257-284.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
Young, S.A.; Schrumpf, B.; Bouck, M.; Moore, M. 2020. How the Association of Official Seed Certifying Agencies (AOSCA) tracks wildland sourced seed and other plant propagating materials. Logan, UT: Utah State University, Utah Agricultural Experiment Station. 36 p.
How to Cite
Shaw, Nancy L.; Gucker, Corey L. 2024. Royal penstemon (Penstemon speciosus). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. https://westernforbs.org