Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
November 2022
Nomenclature
Scabland penstemon (Penstemon deustus) Douglas ex Lindl. belongs to the Scrophulariaceae family, Chelone tribe, Penstemon subgenus, Penstemon section, Deusti subsection (Way and James 1998; Wolfe et al. 2002; Lindgren and Wilde 2003; Freeman 2019; Stevens et al. 2020).
Family
Scrophulariaceae – Figwort family
Genus
Penstemon
Species
deustus
NRCS Plant Code
PEDE4 (USDA NRCS 2022).
Subtaxa
There are four scabland penstemon varieties currently recognized: deustus Douglas ex Lindl., pedicellatus M.E. Jones, suffrutescens L.F. Hend., and variabillis (Suksd.) Cronquist (USDA NRCS 2022).
Synonyms
P. deustus var. heterander ([Torr. & A. Gray] Pennell & D.D. Keck) is a synonym for variety pedicellatus (Freeman 2019).
Common Names
Scabland, hotrock, lava, parched, and scorched penstemon or beardtongue are common names for this species (Taylor 1992; UW Extension 2017). Parched penstemon was the common name given to this plant in its original description in the 1830 Edward’s Botanical Register (Everett 1950).
Chromosome Number
Chromosome numbers are: 2n = 16 and 32 (Hitchcock et al. 1959; Munz and Keck 1973; Freeman 2019). Chromosome number 2n = 32 pertains only to variety variabilis (Freeman 2019).
Hybridization
Available literature suggests scabland penstemon does not hybridize with other Penstemon species. Plants intermediate between varieties deustus and variabillis were reported in the Blue Mountains of northeastern Oregon and adjacent Washington (Hitchcock and Cronquist 2018).
Distribution
Scabland penstemon is a widespread species occurring as scattered populations from central Washington to western Montana and northwestern Wyoming to Nevada and California (Lindgren and Wilde 2003; St. John et al. 2009; LBJWC 2014). Populations are limited to Box Elder County in northwestern Utah (Cronquist et al. 1984; Welsh et al. 2016), the northern two-thirds of Nevada (Cronquist et al. 1984; Hitchcock and Cronquist 2018), and the Klamath, North Coast, Cascade, Sierra Nevada Ranges and Modoc Plateau regions of California (Hickman 1993).
Variety deustus occupies the largest range and occurs from Washington east to Montana and Wyoming and south to Nevada (Fig. 1). It is widespread through the Columbia Basin and Central Rocky Mountains (Nold 1999; Freeman 2019). Variety variabilis grows in Washington and Oregon with populations concentrated in the Blue Mountains. Variety suffrutescens occurs in Oregon and adjacent northern California (Nold 1999; Freeman 2019). Variety pedicillatus has the southernmost distribution and grows in Oregon, California, Nevada, and Utah (Freeman 2019).
In a climate change study that utilized scabland penstemon as a focal species, Brown et al. (2016) predicted that changes in climate would result in geographically widespread declines in genetic diversity of this species. Researchers used climatic, demographic, and genetic data to construct spatially explicit predictions of within-species genetic diversity and noted that decreases in genetic diversity would depend heavily on the continuity of population networks and deme sizes (Brown et al. 2016).
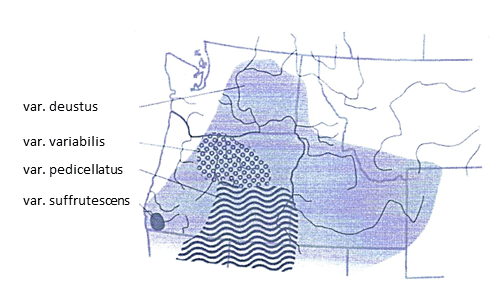 Figure 1. The distribution of scabland penstemon varieties (Strickler 1997).
Figure 1. The distribution of scabland penstemon varieties (Strickler 1997).
Habitat And Plant Associations
Scabland penstemon is commonly associated with grasslands (Johnson and Swanson 2005), sagebrush (Artemisia spp.) (Fig. 2) and chaparral shrublands, pinyon-juniper (Pinus–Juniperus spp.) and oak (Quercus spp.) woodlands (Detling 1961; Cronquist et al. 1984), and forests (Freeman 2019) growing at low to moderate elevations where conditions are dry and rocky (Hitchcock et al. 1959; Lambert 2005). It also occurs, but is less common, in high-elevation forests (Hitchcock et al. 1959).

Figure 2. Scabland penstemon growing in sagebrush and juniper habitats. Photo: USFS RMRS.
In Oregon, scabland penstemon occurs in chaparral and grassland communities. In southwestern Oregon’s Rogue watershed, scabland penstemon grows in late-seral chaparral vegetation with buckbrush (Ceanothus cuneatus), manzanita (Arctostaphylos spp.), Oregon white oak (Q. garryana), and ponderosa pine (P. ponderosa) (Detling 1961). In the Blue and Ochoco Mountains of eastern Oregon, scabland penstemon cover was up to 3% in Idaho fescue-bluebunch wheatgrass (Festuca idahoensis–Pseudoroegneria spicata) and up to 10% in bluebunch wheatgrass-Sandberg bluegrass-lupine (Poa secunda–Lupinus spp.) (Johnson and Swanson 2005).
In California, scabland penstemon grows in a variety of vegetation types including sagebrush scrub, pinyon-juniper, and montane ponderosa pine and red fir (Abies magnifica) forests (Munz and Keck 1973; Hickman 1993). In northwestern California, it is associated with western white pine (Pinus monticola) and canyon live oak (Q. chrysolepis) and in the northern Sierra Nevada Mountains, it is associated with Jeffrey pine (P. jeffreyi), incense cedar (Calocedrus decurrens), white fir (A. concolor), and huckleberry oak (Q. vacciniifolia) (Denton 1982). In the Lake Tahoe region, scabland penstemon grows in subalpine scrub or chaparral with snowbrush ceanothus (Ceanothus velutinus), greenleaf manzanita (Arctostaphylos patula), and pinemat manzanita (A. nevadensis) on dry rocky hillsides (Smiley 1915).
Vegetation associations are similar in more inland habitats. In southeastern Idaho, scabland penstemon is common in woodlands and dwarf shrublands dominated by Rocky Mountain juniper (J. scopulorum) (Rust 1999). At Craters of the Moon National Monument in south-central Idaho, scabland penstemon is a common forb on the youngest seral lava flows (Day and Wright 1985). In central Nevada, scabland penstemon is a distinguishing species within singleleaf pinyon (P. monophylla) woodland types and most common in singleleaf pinyon/mountain big sagebrush (Artemisia tridentata subsp. vaseyana)/scabland penstemon woodlands (West et al. 1998). In Utah, scabland penstemon grows in rocky pinyon-juniper woodlands (Figure 2) (Welsh et al. 2016). In Jackson Hole Wildlife Park in Wyoming, it is often abundant in big sagebrush (A. tridentata) vegetation (Reed 1952). In Montana, it occurs in sagebrush steppe and pinyon-juniper woodlands (Lesica 2012).
Elevation
Scabland penstemon grows in low (1,300 ft [400 m]) to subalpine habitats of about 10,000 ft (3,000 m) in elevation (Table 1; Cronquist et al. 1984; Hickman 1993; Strickler 1997; Blackwell 2006).
Table 1. Elevation range of scabland penstemon varieties.
| Variety | Elevation range (ft) |
| deustus | 1,300–8,530 (Freeman 2019) |
| pedicillatus | 6,560–9,800 (Welsh et al. 2016; Freeman 2019) |
| suffrutescens | 1,970–8,860 (Freeman 2019); 2,000–7,200 in CA (Hickman 1993) |
| variabilis | 1,970–8,530 (Freeman 2019) |
Soils
Scabland penstemon is common on rocky outcrops (Fig. 3), talus slopes, or other rocky soils. It grows primarily in well-drained loam to fine sandy loam soils typically derived from basalt or volcanic origins, although it is occasional on limestone (Keck 1940; Cronquist et al. 1984; Taylor 1992; Nold 1999; Ogle et al. 2014). Scabland penstemon is drought tolerant and moderately tolerant of calcium carbonate (LBJWC 2014). It grows on exposed, dry sites, in soils with a pH range of 5 to 8 (St. John et al. 2009).

Figure 3. Scabland penstemon growing in a basalt outcrop. Photo: USFS RMRS.
In bunchgrass communities in eastern Oregon’s Blue and Ochoco Mountains, scabland penstemon cover was up to 10% in bluebunch wheatgrass-Sandberg bluegrass-lupine communities. These communities occupied basalt soils with very low- to moderate-water holding capacities and 8 to 26 in (20–66 cm) depths to bedrock (Johnson and Swanson 2005).
Description
Scabland penstemon is a densely clumped, perennial, subshrub (Keck 1940; Munz and Keck 1973; Freeman 2019). It produces erect to ascending, slender, branching stems up to 24 in (61 cm) tall from a multi-branched, woody, caudex (Keck 1940; Way and James 1998; Spellenberg 2001; Welsh et al. 2016). The species is extremely variable with foliage ranging from retrorsely hairy to glabrate but not glaucous (Hickman 1993; Lindgren and Wilde 2003; Freeman 2019). Scabland penstemon produces basal and stem leaves (Cronquist et al. 1984; Freeman 2019). Leaves are opposite and often thick (Lindgren and Wilde 2003). Leaf color ranges from gray-green to bright green (Spellenberg 2001; LBJWC 2014; Stevens et al. 2020). Leaf margins range from deeply or shallowly serrate to dentate (Fig. 4) (Stevens et al. 2020). Basal leaves are 0.7 to 2 in (2–5 cm) long, 0.1 to 0.7 in (0.25–1.8 cm) wide, oblanceolate to spatulate, and taper to a petiolar base. Stem leaves are smaller (0.4 to 1 in [1–2.5 cm] long) and more reduced up the stem (Cronquist et al. 1984; Strickler 1997; Welsh et al. 2016). There are typically five to nine pairs of sessile or clasping and lanceolate- to ovate-shaped stem leaves (Cronquist et al. 1984; Freeman 2019).

Figure 4. Close-up of scabland penstemon leaves growing in Oregon. Photo: BLM OR-110 SOS.
Bisexual flowers are produced in terminal thryse inflorescences of 3 to 14 secondary inflorescences (verticillasters) (Fig. 5) (Welsh et al. 2016; Freeman 2019). Primary inflorescences are cylindric, interrupted, and 2.5 to 12 in (6–30 cm) long (Freeman 2019). Secondary inflorescences are one- to six-flowered cymes, two per node. Individual flowers are small (~0.5 in [1.2 cm] long), short campanulate, and bilaterally symmetrical (Taylor 1992; Spellenberg 2001). They have five creamy white to yellowish sepals (Keck 1940; Hickman 1993; Freeman 2019). Lower sepals spread widely and bend downward. The two upper sepals are shorter, slightly spreading, bent upward, and often shriveled (Keck 1940; Cronquist et al. 1984; Spellenberg 2001). The tube formed as the sepals attach has brownish or red-purple nectar guidelines inside (Munz and Keck 1973; Taylor 1992). Flowers have five stamens, four of which are fertile, and one that is infertile. Fertile stamens reach the orifice or are slightly exserted (Keck 1940; Strickler 1997; Blackwell 2006). Scabland penstemon produces seeds in capsules that are glabrous and 2.5 to 5.5 mm long (Cronquist et al. 1984; Freeman 2019). Seeds are brown, gray, or black, measure 0.7 to 1.2 mm long, and have sharp rhombic angles and finely reticulate surfaces (Fig. 6) (Keck 1940; Cronquist et al. 1984; Martin and Barkley 2000).

Figure 5. Scabland penstemon inflorescence. Photo: USFS RMRS.

Figure 6. Close up of scabland penstemon seed. Photo: USFS, Bend Seed Extractory.
Varieties. The four scabland penstemon varieties are marginally discrete. While extreme varietal forms are distinctive, extensive zones of contact exist and degrees of intermediacy are common, especially in Oregon where all four varieties occur (Freeman 2019).
Variety deustus produces opposite, ovate leaves with sharply serrate margins. Stem leaves measure up to 2 in (5 cm) long and 0.8 in (2 cm) wide. Flowers are white to off-white and sometimes tinged with lavender. Flowers may or may not have maroon blotches or red veins in the throat. Staminodes are glabrous (Strickler 1997; Nold 1999; Lindgren and Wilde 2003; Hitchcock and Cronquist 2018; Freeman 2019).
Variety pedicillatus is a very woody form often forming a neat small clump. It produces opposite, lanceolate, gray-green leaves with obscurely to sharply serrate or entire margins. Stem leaves measure up to 1.4 in (3.5 cm) long and 0.6 in (1.4 cm) wide. Flowers are smaller, white to pale violet, and often have a reduced or misshapen brownish upper lip. Flowers are sometimes fragrant, which is rare in the genus. Staminodes are glabrous (Munz and Keck 1973; Hickman 1993; Strickler 1997; Nold 1999; Lindgren and Wilde 2003; Hitchcock and Cronquist 2018; Freeman 2019; Stevens et al. 2020).
Variety suffrutescens produces opposite, ovate to round leaves with sharply serrate margins. Stem leaves measure up to 1.6 in (4 cm) long and 0.6 in (1.4 cm) wide. Flowers are small (<1 cm long), white to off-white and sometimes lavender tinged. Staminodes (1.5–2.5 mm) are glabrous or distally sparsely pilose with yellow hairs up to 0.4 mm long (Hickman 1993; Strickler 1997; Nold 1999; Lindgren and Wilde 2003; Freeman 2019).
Variety variabilis produces narrow oblanceolate to lanceolate whorled or sometimes opposite or subopposite leaves with entire or distally serrate margins. Stem leaves are up to 1.9 in (4.8 cm) long and 0.2 in (5 mm) wide. Flowers are white to off-white and sometimes tinged lavender. Staminodes are bearded distally with sparse white hairs of up to 0.5 mm long (Strickler 1997; Nold 1999; Lindgren and Wilde 2003; Freeman 2019).
Reproduction
Scabland penstemon reproduces by seed (Kramer et al. 2011). In a survey of sagebrush-steppe habitat in the Great Basin region of Nevada and Oregon (elevation range: 4,488–8,691 ft [1,368–2,649 m]), the size of scabland penstemon populations ranged from 100 to 500 flowering plants (Kramer et al. 2011).
Breeding System
Scabland penstemon produces protandrous flowers (male parts mature before female parts) in late spring or early summer (Strickler 1997; Kramer 2009; LBJWC 2014). Although flowers have at least some self-compatibility (Kramer 2009; Kramer et al. 2011), pollination is required for good seed production.
Pollination
Penstemon spp. are visited by honey bees (Apis spp.), bumble bees (Bombus spp.), mason bees (Osmia spp.) or leafcutter bees (Megachile spp.) and small carpenter bees (Ceratina spp.) (Cane and Kervin 2013). Small bees (many mason bee species) and bumble bees at high-elevation sites visited scabland penstemon flowers in the Great Basin in Oregon and Nevada (Kramer et al. 2011). Wilson et al. (2004) recorded visitation of scabland penstemon flowers by mason bees, bumble bees, and digger bees (Anthophora spp. or similar sized nectar-collecting bees).
Ecology
Very little is reported in the literature about the ecology of scabland penstemon. It has been described as a short-lived (St. John et al. 2009) and long-lived perennial (Kramer et al. 2011; Eldredge et al. 2013). It tolerates dry, exposed, low-nutrient and early-seral growing conditions (Day and Wright 1985) and is also reported to grow in late-seral communities (Detling 1961; West et al. 1998).
Seed And Seedling Ecology
Scabland penstemon seed (mean mass: 0.130 mg) is primarily gravity dispersed (Kramer et al. 2011). A common garden study conducted by Kramer (2009) revealed many scabland penstemon seedling growth characteristics and climate relationships (Tables 2 and 3). Seedlings grown in a greenhouse from seed collected from six different Great Basin locations were transplanted to common gardens in Boise, Idaho, and Kaysville, Utah. Transplanted seedling survival was significantly greater in Boise compared to Kaysville (P = 0.0001). This was attributed to the drier conditions and better drained soils in Boise than in Kaysville. Seedling mortality in Kaysville was primarily due to plant rot. Plants from seed collected from sites most climatically like either common garden had higher survival (P < 0.0027).
Table 2. Survival of greenhouse-grown scabland seedlings from seed collected from populations in Oregon and Nevada and transplanted at two common gardens in Idaho and Utah (Kramer 2009).
| Seed origin | Mean min temp (°F) | Mean max temp (°F) | Mean annual ppt (in) | BSU* | UBC* | ||
| No. planted | Survival (%) | No. planted | Survival (%) | ||||
| NV, 6,644 ft | 12.4 | 82.6 | 9.9 | 32 | 63 | 34 | 29 |
| NV, 8,691 ft | 8.2 | 78.4 | 16.9 | 33 | 73 | 29 | 48 |
| OR, 5,883 ft | 14.9 | 82.4 | 11.8 | 30 | 83 | 31 | 58 |
| OR, 4,488 ft | 17.4 | 88.5 | 9.3 | 31 | 81 | 33 | 58 |
| NV, 6,106 ft | 17.4 | 87.1 | 10.4 | 35 | 80 | 34 | 50 |
| NV, 6,017 ft | 16.3 | 84.4 | 11.5 | 27 | 74 | 30 | 53 |
*Common garden locations were Boise State University (BSU) in Boise, ID (2,723 ft [830 m] elevation, 13 in [330 mm] mean annual precipitation) and Utah Botanical Center (UBC) in Kaysville, UT (4,590 ft [1,400 m] elevation, 19.8 in [503 mm] mean annual precipitation).
Scabland penstemon seedlings showed variation by population origin in total and summer growth, with the low-elevation Oregon population showing more growth than most other populations. For all other measurements, scabland penstemon showed significant among-population differences. There was no interaction between common garden and population origin and a weak or insignificant correlation to plant size (Table 3). Interestingly, the largest and fastest growing population (OR, 4,488 ft [1,368 m]) came from seed collected from the same mountain range as one of the smallest and slowest growing populations (OR, 5,883 ft [1,793 m]). Population differences were also evident in germination response, where higher elevation populations required longer stratification than lower elevation populations (see Germination section).
Table 3. Mean values of scabland penstemon seedling growth traits measured at two common garden sites by population origin (Kramer 2009). See table 2 for origin population climate and precipitation characteristics.
| Origin population | Total growth (dm3) | Summer growth (dm3) | Leaf area (cm2) | Stem length (cm) | Flower height
(cm) |
Flower (%) | Fruit (no.) |
| NV, 6,644 ft | 10.2bc | 0.22c | 1.3bc | 23.2bc | 8.6ab | 9.6ab | 36.4a |
| NV, 8,691 ft | 15.2bc | 0.31c | 1.6ab | 22.9bc | 9.8a | 12.0a | 36.7a |
| OR, 5,883 ft | 11.1bc | 0.39bc | 1.2c | 20.9c | 8.4ab | 2.7cd | 24.9b |
| OR, 4,488 ft | 27.7a | 1.39a | 1.7a | 31.4a | 10.3a | 0.3d | 33.8ab |
| NV, 6,106 ft | 17.6b | 0.82ab | 1.4abc | 25.5b | 9.1ab | 4.3bc | 41.9a |
| NV, 6,017 ft | 9.2c | 0.34c | 1.6abc | 20.9c | 7.4b | 5.7abc | 37.0a |
Values within a column followed by different letters are significantly different (P < 0.05).
Disturbance Ecology
Scabland penstemon stems regrow from a woody base each growing season, which suggests it would may following top-killing disturbances.
Wildlife And Livestock Use
Scabland penstemon is a food source for pronghorn (Antilocapra americana) (Ferrel and Leach 1952 cited in Gullion 1964), deer (Odocoileus spp.) (Trout and Thiessen 1973 cited in Stevens et al. 1985), small mammals, birds (Martin et al. 1951), and insects (James and Nunnallee 2011). Insects that utilize scabland penstemon are important to birds, which may explain why scabland penstemon is often a component of habitats used by greater sage-grouse (Centrocercus urophasianus) (Dalke et al. 1963; Lambert 2005).
Studies in California revealed that pronghorn fed on scabland penstemon heavily in spring but only lightly in fall (Ferrel and Leach 1952 cited in Gullion 1964). In a review of plant foods used by North American wildlife (Martin et al. 1951), seeds of penstemon species were important to western rodents. They comprised 10 to 25% of the diets of ground squirrels (Sciuridae), wood rats (Neotoma spp.), and golden-mantled ground squirrels (Callospermophilus lateralis). Finches (Haemorhous spp.) also consumed seeds of penstemon species.
Scabland penstemon is a host plant for moths (Sparganothis senecionana and S. tunicana) (Gilligan and Epstein 2012), and for Edith’s checkerspot butterfly (Euphydryas editha) (James and Nunnallee 2011).
Ethnobotany
Research conducted by Train et al. (1941) revealed that Shoshone and Paiute Indians used and traded scabland penstemon as a medicine plant to treat many diseases, illnesses, and wounds. For trading, the plant was dried and ground into a fine powder. Scabland penstemon was used in various ways both internally and externally to treat venereal diseases (Train et al. 1941; Van Allen Murphey 1990). Powder and poultice treatments were preferred by most Shoshone and Paiute people for chronic sores and skin ailments, like itching and eczema. A Shoshone medicine woman used a liquid scabland penstemon concoction to cure serious ear infections. Poultices made from green or dry plants were used to treat swellings. A scabland penstemon solution was used as an eyewash and a tea was used to treat stomachaches, colds, and rheumatic disorders (Train et al. 1941). An ethnobotanical review indicates that Paiute people used a poultice of mashed, fresh leaves to treat boils, mosquito bites, tick bites, and open sores and a poultice of dry, ground leaves to soothe chapped and cracked skin (Moerman 2003).
Current Medicinal Use
Penstemide, a cytotoxic compound showing some effectiveness against lymphocyte leukemia, was isolated from scabland penstemon (Inouye 1978 cited in Béjar et al. 2000; Jolad et al. 1980).
Horticulture
Scabland penstemon and other penstemon species are commercially available in local garden centers and catalogs (Parkinson 2003). They are recommended for creating waterwise and wildlife- and pollinator-friendly native gardens (Skinner et al. 2005; Hovis and Lohr 2019) in USDA hardiness zones 6 to 8 in areas receiving at least 8 in (203 mm) annual precipitation (St. John et al. 2009).
Scabland penstemon grows well in dry conditions and nutrient-poor, gravelly, shallow soils (Way and James 1998; Lindgren and Wilde 2003; UW Extension 2017). It can be propagated from semi-woody cuttings or seed (LBJWC 2014; UW Extension 2017). Growth from seed requires on average an 8-week stratification period (Lindgren and Wilde 2003). Plants are easily transplanted and growth is slow to moderate (UW Extension 2017).
Penstemon species are short-lived under cultivation but self-seed well in sunny locations with gravel mulch. Gravel mulch is considered better than bark mulch to promote successful self-seeding since bark holds more moisture and can cause root rot (Parkinson 2003). The appearance of scabland penstemon changes with soils. A small compact plant is produced in poor, sterile soils. Plants produce weaker stems and are less attractive in richer soils or shady sites (Everett 1950).
Revegetation Use
Scabland penstemon is a good addition to revegetation seed mixes. It provides fair to good forage for pronghorn, antelope, and small mammals and is attractive to pollinators and other insects that are a food source for birds and other invertebrates making it a good addition to revegetation seed mixes (St. John et al. 2009; Ogle et al. 2011). Scabland penstemon is recommended for rangeland restoration and roadside and other low-maintenance beautification plantings in dry, rocky, well-drained soils (Ley et al. 2007; St. John et al. 2009). It is best adapted to sites receiving at least 8 in (203 mm) of annual precipitation with soils derived from basalt parent materials (Ogle et al. 2014).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
In a Great Basin study evaluating how landscape features interacted with life history traits to influence population genetic structure of scabland penstemon, thickleaf beardtongue (P. pachyphyllus), and Bridge penstemon (P. rostriflorus), scabland penstemon had high genetic structure and low inferred gene flow. Researchers cautioned against broad-scale movement and mixing of populations for restoration (Kramer et al. 2011).
Because empirical seed zones are not currently available for scabland penstemon, generalized provisional seed zones developed by Bower et al. (2014) may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat to moisture index). In Figure 7, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seed zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors. The Western Wildland Environmental Threat Assessment Center’s (USDA FS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Restoration Tool (Richardson et al. 2020) can also guide revegetation planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
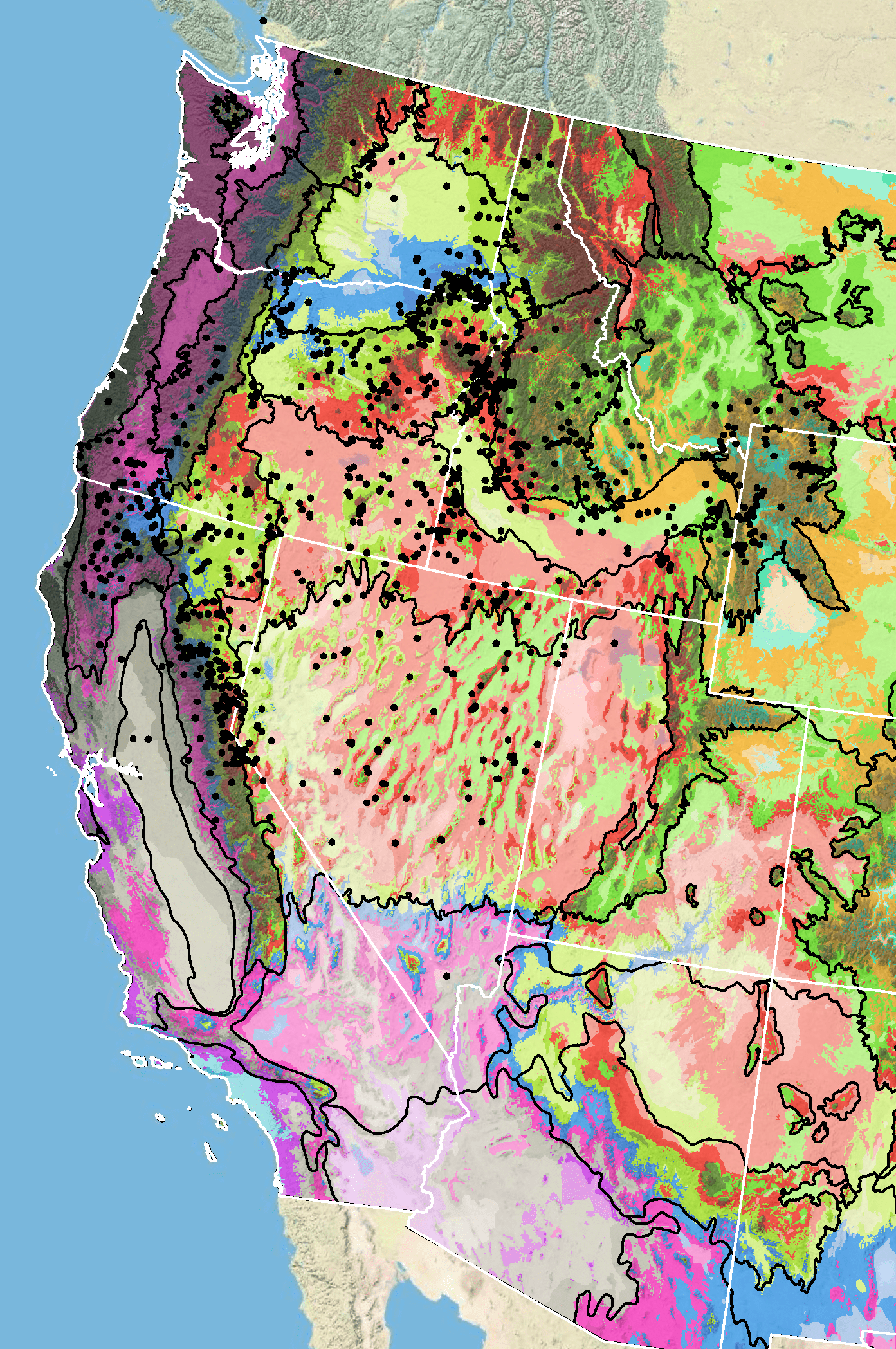
Figure 7. Distribution of scabland penstemon (black circles) based on geo-referenced herbarium specimens and observational data from 1875-2016 (CPNWH 2017; SEINet 2017; USDI USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USDA FS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USDI USGS.
Releases
As of 2022, there were no scabland penstemon germplasm releases.
Wildland Seed Collection
Scabland penstemon seed is mature when the capsule turns straw colored to brown and begins to open (Fig. 8) (Parkinson and DeBolt 2005). This is commonly in August (USDI BLM SOS 2017) and typically 5 to 8 weeks after flowering. Seed persists in the capsules for several weeks (Parkinson and DeBolt 2005).

Figure 8. Scabland penstemon with dry stems and inflorescences ready for harvest. Photo: USFS RMRS.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (Young et al. 2020; UCIA 2015). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Reproductive phenology of scabland penstemon varies by elevation, aspect, and seasonal weather patterns (Parkinson and DeBolt 2005). Seed is mature and ready for harvest when the capsules turn brown and begin to split, typically in August (Fig. 8) (Skinner 2004; USDI BLM SOS 2017). Seed is mature about 5 to 8 weeks after flowering. Seed persists in the capsules for several weeks (Parkinson and DeBolt 2005).
The USDI Bureau of Land Management, Seeds of Success collection crews made 41 total collections of scabland penstemon seed from 2000 to 2014 in Oregon (11), Idaho (20), Nevada (6), and California (4) (USDI BLM SOS 2017). Most harvests occurred in August (27), but some were made in July (6), September (3), October (3), November (1), and June (1). The earliest overall collection date was June 9, 2012, in Boise County, Idaho, at 3,264 ft (995 m) in elevation. The latest overall collection date was October 20, 2010, in Jackson County, Oregon at 3,674 ft (1,120 m) in elevation. In the single year (2010) with the greatest number of collections (14), the earliest harvest was made on July 1, 2010, in Elmore County, Idaho, at 5,119 ft (1,560 m) in elevation, and the latest harvest was on October 20, 2010, in Jackson County, Oregon at 3,674 ft (1,120 m) in elevation (USDI BLM SOS 2017).
Collection Methods
Seed can be collected by hand-stripping the inflorescence (Skinner 2004) or by clipping the entire inflorescence into containers (Parkinson and DeBolt 2005).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021). It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of scabland penstemon.
Post-Collection Management
Scabland penstemon seed collections should be stored in breathable containers in an area free of rodents and insects until they can be cleaned (Skinner 2004). Parkinson and DeBolt (2005) recommend drying collections thoroughly and then freezing them for 48 hours to kill insect pests (Parkinson and DeBolt 2005).
Seed Cleaning
Cleaning scabland penstemon is time consuming because of the small seed size (Fig. 6). Seeds are first removed from the inflorescences by coarse sieving and crushing the capsules on a rubbing board (Skinner 2004; Parkinson and DeBolt 2005). Coarse material can be separated by hand screening (Skinner 2004). Seed cleaning is finished using a small clipper, air-screen cleaner, or air column separator (Skinner 2004; St. John et al. 2009). Larger seed lots can be threshed with a hammermill and finished using air-screen cleaners (Skinner 2004).
Parkinson and DeBolt (2005) used the following procedures to clean a small lot of scabland penstemon seed: 1) Coarse sieving to remove stems, 2) Hand crushing capsules on a rubbing board, 3) Passing material through 1.7- and 1.15-mm sieves with square openings (No. 12 and 16 USA STS), 4) Cleaning with a seed blower, 5) Passing remaining debris through square openings (No. 18 USA STS) four times (Parkinson and DeBolt 2005).
Seed Storage
Scabland penstemon seed is orthodox. After 5.4 years of storage at -4 °F (-20 °C), seed viability was 83% (SER, INSR, RBGK, SID 2023, original viability unknown). The USDA Pullman, Washington, Plant Materials Center stored scabland penstemon seed at 40 °F (4 °C) and 40% relative humidity (Skinner 2004). Neither short- nor long-term seed viability was tested after these storage conditions.
Seed Testing
Researchers have developed guidance and procedures for testing the purity, viability, and germination of penstemon seed.
Viability Testing
The Association of Official Seed Analysts (AOSA) provides a procedure for testing the seed viability of penstemon species (AOSA 2010). Seed is imbibed on moist media overnight at 68 to 77 °F (20–25 °C) then cut longitudinally leaving just enough seed coat at the distal end to keep the halves attached. Cut seeds are soaked in a 0.1 to 0.5% tetrazolium chloride (TZ) for 24 to 48 hours at 86 to 95 °F (30–35 °C). Seed is considered viable if the entire embryo and endosperm stain evenly. Seed is nonviable when any essential part of the embryo or endosperm is unstained (AOSA 2010).
Purity Testing
To test the purity and noxious weed contamination in penstemon seed, Vankus (2006) suggests a minimum of 0.07 oz (2.1 g) of seed is needed.
Germination Testing
To test the germination of penstemon species, the AOSA (2016) provides two methods. For method one, place 400 seeds on blotters moistened with gibberellic acid (GA3 500 ppm) and prechill for 60 days at 36 to 41 °F (2–5 °C). Germinate the seed for 14 days at 50 to 68 °F (10–20 °C). The first germination count is made at 7 days and the last at 14 days. Post-testing viability of ungerminated seed is required. For method two, conduct a TZ test on 400 seeds and then place them on water-moistened blotters and germinate in light for 28 days. The first germination count is made at 14 days and the last at 28 days (AOSA 2016).
Germination Biology
Most literature indicates that scabland penstemon seed requires cold-moist stratification to germinate (Skinner 2004; Kramer 2009; Karrfalt and Vankus 2012). Duration of stratification for germination varies by seed source, with seed from colder, upper-elevation locations requiring longer cold stratification than those from warmer, low-elevation locations (Table 4; Kramer 2009). GA3 treatments can reduce the duration of stratification needed for germination (SER, INSR, RBGK, SID 2023), but St. John et al. (2009) reports that GA3 treatments produced low vigor seedlings.
In preliminary germination testing conducted at the National Tree Seed Laboratory (USFS), scabland penstemon seed germinated best at 50 °F (10 °C) after 12 weeks of moist chilling at 50 °F (10 °C) (Karrfalt and Vankus 2012).
Table 4. Climate conditions and days to germination of scabland penstemon seed from various collection sites with cold stratification of 8 hours light at 50 °F (10 °C) and 16 hours dark at 39 °F (4 °C) (Kramer 2009).
| Origin | Origin climate conditions | Days to germinate with cold stratification | ||
| Elevation | Mean min temp (°F) | Mean max temp (°F) | Mean annual ppt (in) | |
| NV, 6,644 ft | 12.4 | 82.6 | 9.9 | 90.4a |
| NV, 8,691 ft | 8.2 | 78.4 | 16.9 | 87.1a |
| OR, 5,883 ft | 14.9 | 82.4 | 11.8 | 92.3a |
| OR, 4,488 ft | 17.4 | 88.5 | 9.3 | 62.3c |
| NV, 6,106 ft | 17.4 | 87.1 | 10.4 | 59.9c |
| NV, 6,017 ft | 16.3 | 84.4 | 11.5 | 70.2b |
Days to germination followed by different letters are significantly different (P < 0.05).
Meyer et al. (1995) evaluated several Penstemon spp., one of which was scabland penstemon, and found a highly significant negative correlation (P < 0.0001) between average January temperature of the seed collection site and mean germination at 34 °F (1 °C). Seed collected plants growing in areas with severe winters (low temperatures and prolonged snow cover) had long chilling requirements for germination. Seed collected from plants growing in areas with mild winters had short chilling requirements for germination (Meyer et al. 1995).
Scabland penstemon seed collected near Colfax, Washington, germinated at 95% after 90 days of stratification (40 °F [4 °C]). Seed failed to germinate without stratification, and just 18% of seed germinated with shorter duration stratification (45 days). Some seed that failed to germinate after short-duration stratification treatments germinated in the second year outdoors (Skinner 2004).
Total germination was 97% for stratified scabland penstemon seed collected from Boise County, Idaho (3,858 ft [1,176 m]) (Parkinson and DeBolt 2005). Seed was soaked in 250 ppm GA3 for 24 hours and chilled in the dark for 60 days at 39 °F (4 °C) before being put in an incubator. The incubator was kept at a constant 70 °F (21 °C) under a 12:12 hr light:dark cycle. Germination began after three days in the incubator and was 97% by day four (Parkinson and DeBolt 2005).
Wildland Seed Yield And Quality
Post-cleaning seed yield and quality of scabland penstemon seed lots collected in the Intermountain region are provided in Table 5 (USDA FS BSE 2017). Results indicate that professional cleaning results in high levels of purity, seed fill, and viability.
Table 5. Seed yield and quality of scabland penstemon seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USDA Forest Service National Seed Laboratory (USDA FS BSE 2017).
| Seed lot characteristic | Mean | Range | Samples (no.) |
| Bulk weight (lbs) | 2.50 | 0.05–38 | 42 |
| Clean weight (lbs) | 0.57 | 0.0005–13.8 | 42 |
| Clean-out ratio | 0.12 | 0.002–0.68 | 42 |
| Purity (%) | 90 | 38–99 | 42 |
| Fill (%)1 | 91 | 70–99 | 42 |
| Viability (%)2 | 92 | 78–98 | 40 |
| Seeds/lb | 3,467,478 | 2,061,800–5,670,000 | 42 |
| Pure live seeds/lb | 2,897,138 | 1,207,017–5,063,310 | 40 |
¹100 seed X-ray test
²Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well. Walker and Shaw (2005) suggest seed viability of 90% and seed purity of 95% for contracted harvests of scabland penstemon seed.
Agricultural Seed Production
Scabland penstemon was grown for seed production at the Oregon State University Malheur Experiment Station (OSU MES) (Fig. 9). It produced harvestable seed by year 2, and seed yields averaged close to 250 lbs/ac (280 kg/ha) in many years of evaluation (Shock et al. 2021).
 Figure 9. Scabland penstemon seed production plots growing at Oregon State University, Malheur Experiment Station in Ontario, OR. Photo: USDA, Forest Service, Rocky Mountain Research Station.
Figure 9. Scabland penstemon seed production plots growing at Oregon State University, Malheur Experiment Station in Ontario, OR. Photo: USDA, Forest Service, Rocky Mountain Research Station.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Site Preparation
At OSU-MES, scabland penstemon stands were fuller when seed was pretreated with a fungicide and row cover was used to protect emerging seedlings (Table 6; Shock et al. 2012). Plots were disked before seeding on November 3, 2010. Treatments were then applied to protect seeds and seedlings from dry soils, soil crusting, and bird damage. These included row cover (N-sulate, DeWitt Co., Sikeston, MO), fungicide (mix of Captan and Ridomil) seed pretreatments, sawdust (0.26 oz/ft of row), sand (0.65 oz/ft of row), and mulch (hydroseed mulch applied dry at 7.5 g/ft of row in 1.2 in [3 cm] bands over seeded row) (Shock et al. 2012).
Table 6. Scabland penstemon seed production field establishment (%) using various seed protection techniques at Oregon State University Malheur Experiment Station in Ontario, OR. Average seed viability was 70%. Treatments and planting occurred on November 3, 2010 (Shock et al. 2012).
| Row cover | Fungicide | Sawdust | Sand | Mulch | % stand on March 23 | % stand on May 13 |
| x | x | x | 30.5 | 32.1 | ||
| x | x | 55.3 | 46.5 | |||
| x | x | 18.1 | 15.3 | |||
| x | x | 0 | 14.7 | |||
| x | x | x | x | 37.9 | 31.3 | |
| x | x | 0 | 20.0 | |||
| 11.7 | 5.7 |
Weed Management
Research conducted at the NRCS Aberdeen, Idaho, Plant Materials Center (IDPMC) showed that weeds can be controlled using weed-barrier fabric. Planting holes should be 3 to 4 in (8–10 cm) in diameter and spaced 9 to 18 in (23–46 cm) apart (St. John et al. 2009).
At OSU-MES, weeds in scabland penstemon seed production plots were controlled in year 1 (2010) by hand. In subsequent years, weeds were controlled with annual fall and spring applications of pendimethalin (1 lb ai/ac [1.1 kg/ha] broadcast), annual spring applications of clethodim (0.24–0.35 lb ai/ac [0.27–0.39 kg/ha] broadcast) and hand weeding. In year 6 (March 2, 2016), a one-time, grass-specific herbicide application of sethoxydim (0.35 lb ai/ac [0.39 kg/ha] broadcast) was applied (Shock et al. 2021).
On other scabland penstemon plots at OSU-MES, the effects of preemergent and post-emergent herbicides were evaluated. There was little to no emergence of scabland penstemon following preemergent herbicide treatments. Initial post-emergent herbicide treatments in spring 2007 of octanoic acid ester of bromoxynil (0.125 lb ai/ac), oxyfluorfen (0.125 lb ai/ac), prometryn (0.8 lb ai/ac), and linuron (0.5 lb ai/ac) resulted in lower seed yields than untreated plots, but when reapplied the following spring, seed yields showed no difference between herbicide-treated and untreated plots for any of the herbicides tested: octanoic acid ester of bromoxynil [0.125 lb ai/ac], oxyfluorfen [0.125 lb ai/ac], prometryn [0.8 lb ai/ac], linuron [0.5 lb ai/ac], clethodim [0.094 lb ai/ac], pendimethalin [1 lb ai/ac], or dimethenamid-P [0.656 lb ai/ac]) (Shock et al. 2011).
Seeding
Spring planting of cold-stratified seed resulted in poor establishment of scabland penstemon (Shock et al. 2021); therefore, it is recommended to seed at depths of up to 0.6 mm in the fall to allow for natural seed stratification (St. John et al. 2009). Successful fall seeding techniques included soil surface placement of 30 seeds/ft (100 seeds/m) in 30-in (76-cm) rows, covering with sawdust (0.26 oz/ft) and then row cover at OSU-MES. Surface-seeded plots were irrigated for 24 hours in early December due to very dry soil conditions (Shock et al. 2021).
Establishment And Growth
At OSU-MES, scabland penstemon was disked out and reseeded several times (Table 7; Shock et al. 2021). High plant mortality was caused by root rot. Plots seeded in 2005 were harvested for 2 years before being disked out because of substantial stand loss. Plant mortality was high again in winter 2011 and spring 2012. Only undamaged parts of the plots were harvested in 2012. In fall 2012, dead plants were removed, and empty row lengths were hand planted by placing seed on the soil surface and covering it with sawdust and row cover. Replanted stands did not flower sufficiently to harvest seed in 2013. Substantial die off occurred again at the end of 2015 following 2 years of harvests (Shock et al. 2021).
Seed production plots at OSU-MES received moderate levels of fertilization: 50 lbs phosphorus/ac (56 kg/ha) and 2 lbs zinc/ac (2.2 kg/ha) was applied on October 27, 2006; 50 lbs nitrogen/ac (56 kg/ha), 10 lbs phosphorus/ac (11 kg/ha), and 0.3 lb iron/ac (0.3 kg/ha) was applied on April 13, 2012 (Shock et al. 2021).
Irrigation
Seed yield and plant water relations were evaluated for scabland penstemon at OSU MES for over 10 years (2006–2020) (Shock et al. 2021). Studies tested additions of 0, 4, and 8 in (0, 100, and 200 mm) of subsurface drip irrigation delivered 12 in (30 cm) deep in four bi-weekly increments starting at the time of flower bud formation (Table 7).
Table 7. Flowering timing as related to timing of irrigation of scabland penstemon seed production plots growing at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2021).
| Year* | Flowering | Irrigation | ||||
| Start | Peak | End | Start | End | Harvest | |
| 2006 | 10 May | 19 May | 30 May | 19 May | 30 June | 4 Aug |
| 2007 | 5 May | 25 May | 25 June | 19 Apr | 24 June | |
| 2008 | 5 May | —- | 20 June | 18 Apr | 31 May | |
| 2011 | 23 May | 20 June | 14 July | 13 May | 23 June | 16 Aug |
| 2012 | 16 May | 30 May | 4 July | 27 Apr | 7 June | 7 Aug |
| 2013 | 3 May | 18 May | 15 June | 24 Apr | 5 June | |
| 2014 | 10 May | 20 May | 19 June | 29 Apr | 10 June | 21 July |
| 2015 | 1 May | —- | 10 June | 21 Apr | 3 June | 23 July |
| 2016 | No flowering | 18 Apr | 31 May | |||
| 2017 | 15 May | 7 June | 30 June | 2 May | 20 June | 1 Aug |
| 2018 | 3 May | —- | 20 June | 3 May | 13 June | 26 July |
| 2019 | 10 May | 28 May | 28 June | 3 May | 13 June | 30 July |
| 2020 | 7 May | 22 May | 25 June | 9 May | 18 June | 29 July |
* Replanted in fall 2009, fall 2012, and fall 2015 (Shock et al. 2021).
In 10 years of study, seed yield of scabland penstemon rarely responded to irrigation (Table 8). In 2006, 2011, and 2014 where irrigated and non-irrigated treatment seed yield differences were the greatest (150–200 lbs/ac [168–224 kg/ha]), relationships were not reliably related to growing season length or precipitation levels. Overall, irrigation at 8 in (200 mm)/season resulted in more stand loss than 4 in (100 mm)/season or non-irrigated treatments (Shock et al. 2021).
Table 8. Seed yield for scabland penstemon in response to no additional irrigation and supplemental irrigation of 4 and 8 in at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2021).
| Year | Supplemental irrigation (in/season*) | ||
| 0 | 4 | 8 | |
| ——-Seed yield (lbs/acre)—— | |||
| 2006 | 1246 | 1201 | 1069 |
| 2007 | 120 | 188 | 148 |
| 2008 | Disked out | ||
| 2011 | 638 | 478 | 453 |
| 2012 | 309 | 292 | 300 |
| 2013 | No flowering | ||
| 2014 | 356 | 505 | 463 |
| 2015** | 20a | 77b | 67b |
| 2017 | 205 | 259 | 248 |
| 2018 | 111 | 85 | 95 |
| 2019 | 113 | 154 | 144 |
| 2020 | 82 | 122 | 94 |
| Average | 266 | 286 | 262 |
*Replanted in fall 2009, fall 2012, and fall 2015.
**Seed yields significantly (P < 0.1) different in 2015.
Pollinator Management
Pollination is essential for commercial seed production. Bees and wasps that pollinate scabland penstemon include sweat bees (Halictidae), bumble bees, honey bees, and leafcutter bees (St. John et al. 2009). Bumble bees and other native bees (e.g., Osmia spp.) have been excellent pollinators of penstemon species in cultivated plots. Management practices that encourage native bee populations in and around native seed farms could increase seed yields (Cane 2008).
Pest Management
Though it is possible to produce penstemon seed crops without apparent insect damage, when pests are present crop damage can be “catastrophic” (Hammon 2014). Penstemon weevils (Hesperobarus ovulum) or borer larvae (Barus spp.) are limited to southwestern Colorado, but the penstemon clearwing borer (Penstemonia spp.) is widespread and attacks multiple penstemon species (St. John et al. 2009; Hammon 2014). Clearwing borer larvae feed in the stems of the root crown and lower above-ground portions of the plant. A pheromone is available to monitor for adult clearwing borers. Other potentially significant pests include lygus bugs (Lygus spp.) and raceme-boring moths (Crambidae) (St. John et al. 2009). Many penstemon species are short-lived plants. Hammon (2014) hypothesized that the short-life cycle may be attributed to borers and other insects and pathogens.
Scabland penstemon is also susceptible to soil-borne fusarium and rhizoctonia root rot. These pathogens and their damage are most severe in poorly drained loam or clay soils (St. John et al. 2009). Plants are also hosts to the following fungi: Puccinia palmeri, P. pentastemonis, Ramularia nivosa, Sphaeropsis spp. (Farr and Rossman 2017). Damage from or prevalence of these fungi was not reported.
Seed Harvesting
Seed is mature, and stands are ready for harvest when stems and capsules begin to dry, turn brown, and start to open, which occurs about 5 to 8t weeks after flowering. Because seeds can persist in the capsules for several weeks, harvest does not have to occur immediately once capsules senesce. Seed can be harvested by hand or direct combining (St. John et al. 2009). At OSU MES, scabland penstemon seed was harvested using a small-plot combine (Wintersteiger, Salt Lake City, UT). This harvest method did not separate seed from the capsules (Shock et al. 2021).
Seed Yields And Stand Life
Based on research conducted at OSU MES, scabland penstemon plants produced harvestable seed in year 2. Stands rarely persisted in a full and productive state beyond 3 years, and seed yields averaged 266 lbs/ac (298 kg/ha) and ranged from 20 to 1,246 lbs/ac (22–1,397 kg/ha) in 10 years of study (Table 8; Shock et al. 2021).
Nursery Practice
Several Plant Materials Centers (PMC) developed protocols for propagating scabland penstemon. Researchers at the IDPMC reported that scabland penstemon seed can be fall-seeded directly into cones or flats and kept outdoors (St. John et al. 2009) or artificially stratified for 8 to 12 weeks (see Germination Biology section for details). Treating seeds with liquid smoke can reduce the duration of stratification. Seed germination can be improved with a weak GA3 solution (250 ppm), though seedlings treated with GA3 tend to be less vigorous than non-treated (St. John et al. 2009).
The Pullman, WA PMC, produced scabland penstemon seedlings with a tight root plug in 10 in3 (164 cm³) Ray Leach Super cell cone-tainers (Skinner 2004). They sowed locally collected seed in cells filled with Sunshine #4 soil mix. Seed was lightly covered with soil and a thin layer of pea gravel to prevent seeds from floating. Cells were watered and placed outside in October, where they remained until being moved into a greenhouse in early January. Researchers noted that artificial seed stratification (35–40 °F [2–4 °C] for 90 d) could replace outdoor natural stratification. Seed germination began within 6 days in the greenhouse and was complete in 10 to 12 days. Seedlings were considered established in about 2 weeks. Seedlings were watered every other day and fertilized once each week with a complete, water-soluble fertilizer containing micronutrients. Plants were moved to a cold frame in late March or early April where watering occurred every other day in cool weather and every day in dry, hot weather. The hardening phase was 2 weeks. No insect or disease problems were encountered (Skinner 2004).
The Dorena Genetic Resource Center in Cottage Grove, Oregon, reported procedures to produce 10 in3 plugs (164 cm³) (Riley and Klocke 2018). Locally collected scabland penstemon seed was sown in trays with stabilized medium plugs (Q-plugs). Plugs were thoroughly moistened with a GA3 solution (250 mg/L) and lightly covered with nursery grit. Trays were placed in plastic bags and refrigerated at 34 to 37 °F (1–3 °C) for 60 days. Trays were kept moist throughout the stratification period. If mold formed, trays were treated with 1% hydrogen peroxide. Seedlings were transplanted to target containers about 3 weeks after removal from the refrigerator. Plugs were transplanted in a growing medium mix (40 peat: 20 composted fir bark: 20 perlite: 20 pumice) that included a controlled release fertilizer (NPK 18-P2O5-K2O with minor nutrients; 0.8 g/164 ml container). During the growing season, seedlings were fertilized (NPK 17–20:5–19:18–24, 100–150 ppm) weekly throughout growing season. Seedlings were moved to an outdoor growing area in mid-September. The hardening phase was 2 to 3 weeks. Plants were irrigated before shipping, and seedlings were outplanted in fall (Riley and Klocke 2018).
Wildland Seeding And Planting
To date (2022), use of scabland penstemon in wildland revegetation has been limited, although it has the potential to improve pollinator habitat at sites with medium to coarse-textured volcanic soils receiving at least 8 in (203 mm) annual precipitation (Ogle et al. 2014). Broadcast or drill seeding up to 3 mm deep with the desired portion of its full-stand rate of 1 lb/ac (1.1 kg/ha) is recommended (Walker and Shaw 2005; Ogle et al. 2014). In wildland revegetation, scabland penstemon would be a minor component of the seed mix and establishing community. It can also be seeded in patches with other forb species. Post-seeding management should be based on key species in the establishing community. Grazing should be deferred for at least 2 years in newly seeded areas (St. John et al. 2009).
On the west slope of the Sierra Nevada Range in Plumas County, California, scabland penstemon did not establish from seed; however, seedlings transplanted in rocky, sandy, silt, and loam soils survived up to 5 years. Seedlings were outplanted in rocky, sandy, silt, loam soils (Everett 1957). No other details were provided.
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Byron Love, USDA Pollinating Insect Research Unit and Matt Fisk, US Geological Survey.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Association of Official Seed Analysts [AOSA]. 2016. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Béjar, E.; Reyes-Chilpa, R.; Jiménez-Estrada, M. 2000. Bioactive compounds from selected plants used in the XVI Century Mexican traditional medicine. Studies in Natural Products Chemistry. 24(E): 799-844.
Blackwell, L.R. 2006. Great Basin wildflowers: A guide to common wildflowers of the high deserts of Nevada, Utah, and Oregon. Helena, MT: Morris Book Publishing. 288 p.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Brown, J.L.; Weber, J.J.; Alvarado-Serrano, D.F.; Hickerson, M.J.; Franks, S.J.; Carnaval, A.C. 2016. Predicting the genetic consequences of future climate change: The power of coupling spatial demography, the coalescent, and historical landscape changes. American Journal of Botany. 103(1): 153-163.
Cane, J.H. 2008. Pollinating bees crucial to farming wildflower seed for U.S. habitat restoration. In: James, R.; Pitts-Singer, T., eds. Bees in agricultural ecosystems. Oxford, UK: Oxford University Press: 48-64.
Cane, J.H.; Kervin, L. 2013. Gardening for native bees in Utah and beyond. ENT-133-09. Logan,UT: Utah State University Cooperative Extension and Utah Plant Pest Diagnostic Laboratory. 13 p.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Agoseris aurantiaca. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/ [Accessed 2020 October 25].
Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K. 1984. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Vol. 4: Subclass Asteridae (except Asteraceae). New York, NY: The New York Botanical Garden. 573 p.
Dalke, P.D.; Pyrah, D.B.; Stanton, D.C.; Crawford, J.E.; Schlatterer, E.F. 1963. Ecology, productivity, and management of sage grouse in Idaho. The Journal of Wildlife Management. 27(4): 811-841.
Day, T.; Wright, R.G. 1985. The vegetation types of Craters of the Moon National Monument. Bull. 38. Moscow, ID: University of Idaho, College of Forestry, Wildlife, and Range Sciences, Forest, Wildlife and Range Experiment Station. 6 p.
Denton, M.F. 1982. Revision of Sedum section Gormania (Crassulaceae). Brittonia. 34(1): 48-77.
Detling, L.E. 1961. The chaparral formation of southwestern Oregon, with considerations of its postglacial history. Ecology. 42(2): 348-357.
Eldredge, E.; Novak-Echenique, P.; Heater, T.; Mulder, A.; Jasmine, J. 2013. Plants for pollinator habitat in Nevada. Tech. Note NV 57. Reno, NV: U.S. Department of Agriculture, Natural Resources Conservation Service. 65 p.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Everett, P. 1957. A summary of the culture of California plants at the Rancho Santa Ana Botanic Garden 1927-1950. Claremont, CA: The Rancho Santa Ana Botanic Garden. 223 p.
Everett, P.C. 1950. The Californian Penstemons. El Aliso. 2(2): 155-198.
Farr, D.F.; Rossman, A.Y. 2017. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://nt.ars-grin.gov/fungaldatabases/.
Freeman 2019. Penstemon. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 17. Magnoliophyta: Tetrachondraceae to Orobanchaceae. New York, NY: Oxford University Press: 82-202.
Gilligan, T.M.; Epstein, M.E. 2012. TortAI, Tortricids of agricultural importance to the United States (Lepidoptera: Tortricidae). Fort Collins, CO: Identification Technology Program (ITP), USDA/APHIS/PPQ/CPHST. http://idtools.org/id/leps/tortai/.
Gullion, G.W. 1964. Wildlife uses of Nevada plants. CR-24-64. Beltsville, MD: U.S. Department of Agriculture, Agricultural Research Service, National Arboretum Crops Research Division. 170 p.
Hammon, B. 2014. Penstemon seed production pests. Grand Junction, CO: Colorado State University Extension, Tri-Rivers Area. http://www.wci.colostate.edu/shtml/PenstemonPests.shtml.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
Hitchcock, C.L.; Cronquist, A. 2018. Flora of the Pacific Northwest: An illustrated manual. Second Ed. Giblin, D.E.; Legler, B.S.; Zika, P.F.; Olmstead, R.G., eds. Seattle, WA: University of Washington Press. 882 p.
Hitchcock, C.L; Cronquist, A.; Ownbey, M. 1959. Part 4: Ericaceae through Campanulaceae. In: Hitchcock, C.L.; Cronquist, A; Ownbey, M.; Thompson, J.W. Vascular plants of the Pacific Northwest. Seattle, WA: University of Washington Press: 510 p.
Hovis, H.; Lohr, K. 2019. Native garden guide for southwestern Idaho. Boise, ID: U.S. Department of the Interior, Bureau of Land Management and U.S. Fish and Wildlife Service. 60 p.
Hurd, E.G. n.d. Tetrazolium test for selected species. Unpublished report on file with the USDA Forest Service, Rocky Mountain Research Station, Boise, ID. 30 p.
James, D.G.; Nunnallee, D. 2011. Life histories of Cascadia butterflies. Corvallis, OR: Oregon State University Press. 447 p.
Johnson, C.G.; Swanson, D.K. 2005. Bunchgrass plant communities of the Blue and Ochoco Mountains: A guide for managers. Gen. Tech. Rep. PNW-GTR-641. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. 119 p.
Jolad, S.D.; Hoffmann, J.J.; Cole, J.R.; Tempesta, M.S.; Bates, R.B. 1980. Cytotoxic agent from Penstemon deustus (Scrophulariaceae): Isolation and stereochemistry of Liriodendrin, a symmetrically substituted furofuranoid lignan diglucoside. Journal of Organic Chemistry. 45(7): 1327-1329.
Karrfalt, R.; Vankus, V. 2012. Development of germination protocols, seed weight, purity, and seed conditioning/cleaning protocols for Great Basin grasses and forbs. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Selection and Increase Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 36-39.
Keck, D.D. 1940. Studies in Penstemon. VII. The subsections Gairdneriani, Deusti, and Arenarii of the Graciles, and miscellaneous new species. The American Midland Naturalist. 23(3): 594-616.
Kramer, A.T. 2009. Ecological genetics of Penstemon in the Great Basin, U.S.A. Chicago, IL: University of Illinois at Chicago. Dissertation. 111 p.
Kramer, A.T., Fant, J.B., and Ashley, M.V. 2011. Influences of landscape and pollinators on population genetic structure: Examples from three Penstemon (Plantaginaceae) species in the Great Basin. American Journal of Botany. 98(1): 109-121.
Lady Bird Johnson Wildflower Center [LBJWC]. 2014. Penstemon deustus Douglas ex Lindl. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants-main [Accessed 2022 June 21].
Lambert, S. 2005. Guidebook to the seeds of native and non-native grasses, forbs and shrubs of the Great Basin. Boise, ID: U.S. Department of the Interior, Bureau of Land Management, Idaho State Office. 136 p.
Lesica, P. 2012. Manual of Montana vascular plants. Fort Worth, TX: Botanical Research Institute of Texas (BRIT) Press. 779 p.
Ley, E.L.; Buchmann, S.; Stritch, L.; Soltz, G. 2007. Selecting plants for pollinators: A regional guide for farmers, land managers, and gardeners in the Northern Rocky Mountain Steppe, Coniferous Forest, and Alpine Meadow Province. San Francisco, CA. Pollinator Partnership. 24 p.
Lindgren, D.; Wilde, E. 2003. Growing penstemons: Species, cultivars and hybrids. American Penstemon Society. Haverford, PA: Infinity Publishing Company. 151 p.
Martin, A.C.; Barkley, W.D. 2000. Seed identification manual. Caldwell, NJ: The Blackburn Press. 221 p.
Martin, A.C.; Zim, H.S.; Nelson, A.L. 1951. American wildlife and plants: A guide to wildlife food habits. New York, NY: Dover Publications. 500 p.
Meyer, S.E.; Kitchen, S.G.; Carlson, S.L. 1995. Seed germination timing patterns in Intermountain Penstemon (Scrophulariaceae). American Journal of Botany. 82(3): 377-389.
Moerman, D. 2003. Native American ethnobotany: A database of foods, drugs, dyes, and fibers of Native American peoples, derived from plants. Dearborn, MI: University of Michigan. http://naeb.brit.org
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Nold, R. 1999. Penstemons. Portland, OR: Timber Press. 259 p.
Ogle, D.; St. John, L.; Stannard, M.; Holzworth, L. 2014. Conservation plant species for the Intermountain West. Tech. Note 24. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 72 p.
Ogle, D.; Tilley, D.; Cane, J.; St. John, L.; Fullen, K.; Stannard, M.; Pavek, P. 2011. Plants for pollinators in the Intermountain West. Plant Materials Tech. Note 2A. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 40 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Parkinson, H. 2003. Landscaping with native plants of the Intermountain Region. Tech. Ref. 1730-3. Boise, ID: U.S. Department of the Interior, Bureau of Land Management. 46 p.
Parkinson, H.; DeBolt, A. 2005. Propagation protocol for production of container (plug) Penstemon deustus Dougl. ex Lindl. Plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2020 June 5].
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Riley, L.E.; Klocke, A. 2018. Propagation protocol for production of container (plug) Penstemon deustus Dougl. ex Lindl. plants 164 ml (10 in³) container. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2020 June 5].
Rust, S.K. 1999. Pinyon-juniper woodland classification and description in Research Natural Areas in southeastern Idaho. In: Monsen, S.B.; Stevens, R. comps. Proceedings – Ecology and management of pinyon-juniper communities within the Interior West: Sustaining and restoring a diverse ecosystem; 1997 September 15-18; Provo, UT. Proc. RMRS-P-9. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 82-93.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shock, C.C.; Feibert, E.B.; Saunders, L.D.; Parris, C.; Shaw, N.; Johnson, D.; Bushman, S. 2012. Seed production of Great Basin native forbs subsurface drip irrigation 2006-2011 new plantings, seeding practices, legume seed scarification. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 93-132.
Shock, C.C.; Feibert, E.B.; Saunders, L.D.; Parris, C.; Shaw, N.; Sampangi, R. 2011. Seed production of Great Basin native forbs- Subsurface drip irrigation (SDI) for stable, efficient native forb seed production using small amounts of supplemental irrigation water; weed control; and seeding practices. In: Shaw, N.L.; Pellant, M., eds. Great Basin Native Plant Selection and Increase Project: 2010 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 93-128.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Wieland, K.D.; Shaw, N.; Kilkenny, F. 2021. Irrigation requirements for seed production of five native Penstemon species. In: Reitz, S. R., et al. 2020. Malheur Experiment Station Annual Report 2020, OSU AES Ext/CrS165. Corvallis, OR: Oregon State University: 234-246.
Skinner, D.M. 2004. Propagation protocol for production of container (plug) Penstemon deustus Dougl. ex Lindl. plants. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2020 June 5].
Skinner, D.M.; Warnick, P.; French, B.; Fauci, M. 2005. Characteristics and uses of native Palouse forbs in landscaping. Available from http://www.palouseprairie.org.
Smiley, F.J. 1915. The alpine and subalpine vegetation of the Lake Tahoe region. Botanical Gazette. 59(4): 265-286.
Society for Ecological Restoration; International Network for Seed Based Restoration; Royal Botanic Gardens [SER, INSR, RBGK]. 2023. Seed Information Database (SID). Available from: https://ser-sid.org/
Spellenberg, R. 2001. National Audubon Society field guide to North American wildflowers: Western region, revised edition. New York, NY: Alfred A. Knopf, Inc. 862 p.
St. John, L.; Ogle, D.; Shaw, N.L. 2009. Plant guide: Hotrock penstemon (Penstemon deustus Douglas ex Lindl). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 3 p.
Stevens, M.R.; Love, S.L.; McCammon, T. 2020. The heart of Penstemon country: A natural history of penstemons in the Utah region. Helena, MT: Sweetgrass Books. 393 p.
Stevens, R.; Shaw, N.; Howard, C.G. 1985. Important nonleguminous forbs for Intermountain ranges. In: Proceedings: Selected papers presented at the 38th Annual Meeting of the Society for Range Management; 1985 February 11-15; Salt Lake City, UT. Denver, CO: Society for Range Management: 102-112.
Strickler, D. 1997. Northwest penstemons. Columbia Falls, MT: The Flower Press. 191 p.
Taylor, R.J. 1992. Sagebrush country: A wildflower sanctuary. Missoula, MT: Mountain Press Publishing Company. 211 p.
Train, P.; Henrichs, J.R.; Archer, W.A. 1941. Medicinal uses of plants by Indian tribes of Nevada: Part 1. Contributions toward a flora of Nevada. No. 33. Washington, DC: U.S. Department of Agriculture, Bureau of Plant Industry, Division of Plant Exploration and Introduction. 199 p.
University of Wyoming Extension [UW Ext.] 2018. Native plants for the Intermountain West. Laramie, WY: University of Wyoming. http://www.wyoextension.org/westernnativeplants
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2022. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2021. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 44 p.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2017. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Van Allen Murphey, E. 1990. Indian uses of plants. Glenwood, IL. Meyerbooks. 81 p.
Vankus, V. 2006. Association of official seed certifying agencies (AOSCA). https://www.aosca.org/.
Walker, S.C.; Shaw, N.L. 2005. Current and potential use of broadleaf herbs for reestablishing native communities. In: Shaw, N.L.; Pellant, M.; Monsen, S.B., comps. Sage-grouse habitat restoration symposium proceedings; 2001 June 4-7; Boise, ID. RMRS-P-38. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 56-61.
Way, D.; James, P. 1982. The gardeners guide to growing penstemons. Portland, OR: Timber Press. 160 p.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
West, N.E.; Tausch, R.J.; Tueller, P.T. 1998. A management-oriented classification of pinyon-juniper woodlands of the Great Basin. Gen. Tech. Rep. RMRS-GTR-12. Ogden, UT: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. 42 p.
Wilson, P.; Castellanos, M.C.; Hogue, J.N.; Thomson, J.D.; Armbruster, W.S. 2004. A multivariate search for pollination syndromes among penstemons. Oikos. 104(2): 345-361.
Wolfe, A.D.; Datwyler, S.L.; Randle, C.P. 2002. A phylogenetic and biogeographic analysis of the Cheloneae (Scrophulariaceae) based on ITS and matK sequence data. Systematic Botany. 27(1): 138-148.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2022. Scabland penstemon (Penstemon deustus). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org/species/scabland-penstemon-penstemon-deustus/