Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
July 2018
Update
December 2024
Nomenclature
Crepis acuminata Nutt., commonly called tapertip hawksbeard, belongs to the Chichorieae tribe (USFS 1937) of the Asteraceae or Aster family (Bogler 2006; USDA NRCS 2017).
Family
Asteraceae – Aster family
Genus
Crepis
Species
acuminata
NRCS Plant Code
CRAC2 (USDA NRCS 2017).
Subtaxa
No subspecies are recognized by the Flora of North America (Bogler 2006).
Synonyms
Crepis acuminata subsp. pluriflora Babcock & Stebbins, C. angustata Rydberg, C. sesselifolia Rydberg, C. acuminata Nutt. subsp. acuminata (Bogler 2006; Chambers 2020).
Common Names
Tapertip hawksbeard, longleaf hawksbeard, mountain hawksbeard, tall hawksbeard (Applegate 1938; Taylor 1992; Welsh et al. 2015; USDA NRCS 2017; Chambers 2020).
Chromosome Number
Chromosome number varies: 2n = 22, 33, 44, 55, 88 (Bogler 2006; Chambers 2020) and distinguishes sexual and apomictic plant types. Diploid plants (2n = 22) are sexual, and polyploid plants are apomictic autopolyploids (Babcock and Stebbins 1938; Cronquist et al. 1994; Majeský et al. 2017). Plant forms with 33 or 44 chromosomes are found practically throughout the species range. Forms with 55 chromosomes are rare and derived from hybridization with nakedstem hawksbeard (C. pleurocarpa) (Babcock and Stebbins 1938). Ploidy is discussed more in the Distribution and Reproduction sections below.
Distribution
Tapertip hawksbeard occurs in all states west of Montana, Wyoming, Colorado, and New Mexico (Bogler 2006). It is common in and east of the Cascade and Sierra mountain ranges and found in the northern parts of Arizona and New Mexico (Cronquist et al. 1994; LBJWC 2018). Populations are most common in the Columbia Plateau and Great Basin provinces (Babcock and Stebbins 1938) and exist in most, if not all, counties in Utah (Welsh et al. 2015). In general, tapertip hawksbeard occurs in regions with continental climates that experience dryness in all seasons (Babcock and Stebbins 1938).
In a thorough study of hawksbeards (Crepis spp.), Babcock and Stebbins (1938) mapped the distribution of the diploid sexual and polyploid apomictic tapertip hawksbeard forms (Fig. 1). The diploid sexual form is most widespread. It predominates in the northern part of the species range and overlaps the distribution of the polyploid apomictic forms in many areas (Babcock and Stebbins 1938).
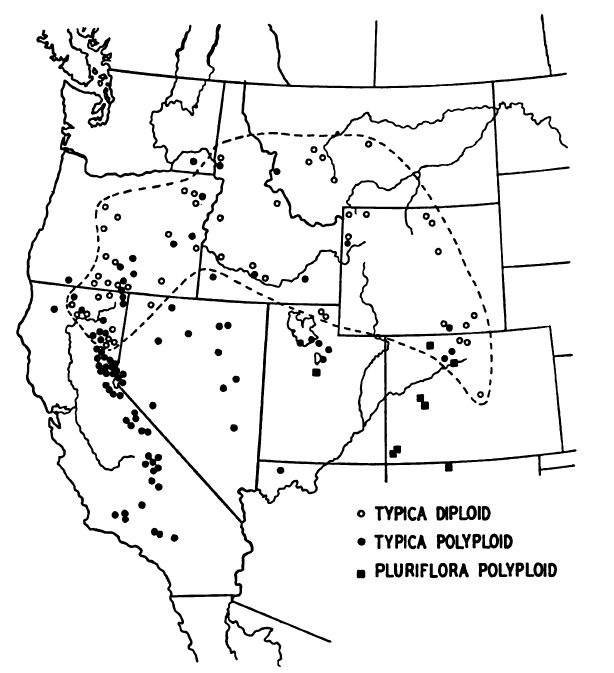
Figure 1. Distribution of tapertip hawksbeard and its sexual diploid (typica diploid) and apomictic polyploid forms (typica polyploid and pluriflora polyploid) (Babcock and Stebbins 1938).
Habitat And Plant Associations
Tapertip hawksbeard is widespread but rarely found in great abundance (USFS 1937). It occurs on open, dry flats, gravelly to rock slopes and ridges, and roadsides in sagebrush (Artemisia spp.) grasslands, mountain brush communities, pinyon-juniper (Pinus–Juniperus spp.) and quaking aspen (Populus tremuloides) woodlands, Douglas fir (Pseudotsuga menziesii) and ponderosa pine (Pinus ponderosa) forests (Craighead et al. 1963; Munz and Keck 1973; Goodrich and Neese 1986; Bogler 2006; Welsh et al. 2015; Hitchcock and Cronquist 2018; Bates et al. 2020). It is especially frequent in mid-elevation grassland and sagebrush vegetation adjoining the lower edge of the ponderosa pine zone (Babcock and Stebbins 1938; Hermann 1966). Populations are also found in upper elevation spruce-fir (Picea–Abies spp.) and white fir (Abies concolor) forests (Cronquist et al. 1994; Welsh et al. 2015). In the Sweetwater Mountains of California and Nevada it is common from pinyon-juniper woodlands to lodgepole pine (Pinus contorta) forests (Lavin and Wojciechowski 2002). Tapertip hawksbeard is commonly found with the following forbs and grasses: arrowleaf balsamroot (Balsamorhiza sagittata), buckwheats (Eriogonum spp.), lupines (Lupinus spp.), Sandberg bluegrass (Poa secunda), and other bunchgrasses (Pseudoroegneria, Agropyron, Elymus spp.) (Fig. 3)(USFS 1937).
Shrublands. Tapertip hawksbeard is commonly associated with sagebrush (Fig. 2) and mountain brush communities. In a 10-year study of plant communities, soil, and vegetation relationships in near climax sagebrush communities in northern Utah, southern Idaho, northeastern Nevada, and west-central Wyoming, tapertip hawksbeard occurred in 47 of 85 sagebrush-grassland plots (Passey et al. 1982). In descriptions of rangeland cover types of the US, tapertip hawksbeard was described as frequent in basin big sagebrush (Artemisia tridentata subsp. tridentata), mountain big sagebrush (A. t. subsp. vaseyana), threetip sagebrush (A. tripartita), and more mesic parts of Wyoming big sagebrush (A. t. subsp. wyomingensis), and black sagebrush (A. nova) communities (Shiflet 1994). In the Blue and Ochoco mountains of Oregon, tapertip hawksbeard was frequent but cover was low in antelope bitterbrush/Idaho fescue-bluebunch wheatgrass (Purshia tridentata/Festuca idahoensis–Pseudoroegneria spicata) and mountain big sagebrush/Idaho fescue-prairie Junegrass (Koeleria cristata) communities (Johnson and Swanson 2005).
In the Owyhee Mountains of southern Idaho, tapertip hawksbeard was considered a common perennial forb in a Wyoming big sagebrush community at 1221 m elevation with 264 mm annual precipitation and sandy loam soils and at a mountain big sagebrush site with annual precipitation of 624 mm and loam soils (Schachtschneider et al. 2024. Clark et al. (2018) described tapertip hawksbeard as a typical forb in a mountain big sagebrush-mountain snowberry vegetation type encroached by western juniper in the Owyhee Mountains that was typical of mid and higher elevation portions of the sagebrush steppe. Elevation on the site ranged from 1542 – 1763 m. Soils were derived from granite and composed of coarse, loamy, mixed, frigid Typic Haploxerolls and fine, loamy, mixed, frigid Typic Argixerolls. Precipitation at both sites occurs primarily in winter and spring. In Box Elder County, Utah, tapertip hawksbeard was a characteristic forb on a mountain big sagebrush study site between 1670 and 2560 m (Montez-Sánchez et al. 2017). Soils were Xerolls with well-developed A-horizons and texture ranging from stony, cobbly, or gravelly loam to clayey.

Figure 2. Tapertip hawksbeard growing in Oregon. Photo: USDI BLM OR030 Seeds of Success (SOS).
In southern Idaho, tapertip hawksbeard was reported in sagebrush kipukas, islands of vegetation isolated by lava flows (Passey and Hugie 1963; Tisdale et al. 1965). Near Craters of the Moon National Monument, tapertip hawksbeard was most frequent in threetip sagebrush/Idaho fescue kipukas on moist, cool, north and east slopes with relatively deep and fertile soils. It also occurred, but was poorly represented, in big sagebrush/Idaho fescue-bluebunch wheatgrass vegetation on warm, dry, southern and western slopes (Tisdale et al. 1965).
Bates et al. (2022) reported tapertip hawksbeard growing on low sagebrush (Artemisia arbuscula subsp. arbuscula) sites in south central Oregon on west-facing aspects with 1 to 10% slopes at elevations of 1430–1560 m and average crop-year precipitation of 280 mm.
Tapertip hawksbeard often occurs in mountain brush communities. In 60 stands from 18 northeastern Nevada mountain ranges, tapertip hawksbeard constancy (percentage of plots within a vegetation type on which tapertip hawksbeard occurred) was nearly 100% in mountain snowberry (Symphoricarpos oreophilus) and mountain big sagebrush communities. Cover of tapertip hawksbeard ranged from 0.1 to 0.4% in these communities (Tueller and Eckert 1987). Constancy was high (>20%) and cover ranged from 1 to 4% in Saskatoon serviceberry (Amelanchier alnifolia) mountain brush sites in eastern Idaho (Major and Rejmanek 1992).
Tapertip hawksbeard was a representative forb on medusahead-infested mountain big sagebrush in Box Elder County, Utah. The site was at 5,480 to 8,400 ft (1,670-2,560 m) elevation with 5 to 70% slopes (Montes-Sánchez et al. 2017). Soils were Xerolls with a well-developed organic-rich A-horizon. Textures were stony, cobbly or gravelly loams upslope and clayey downslope.
Woodlands/Forests. Tapertip hawksbeard was considered a major associate species in western juniper (J. occidentalis) woodlands characteristic of shallow soils in Oregon’s Blue Mountains (Franklin and Dyrness 1973). It also occurred in Utah juniper/low sagebrush (J. osteosperma/A. arbuscula)/Sandberg bluegrass on north and east slopes with very fine soils in Nevada’s Cow Creek Watershed (Blackburn et al. 1969). In Underdown Canyon, Lander County, Nevada, tapertip hawksbeard was found in singleleaf pinyon (Pinus monophylla) woodlands on 7,200- to 7,300-ft (2,195–2,225 m) north-facing slopes with intermediate levels of tree cover (MontBlanc et al. 2007).
When riparian and terrestrial ecosystems were surveyed for an entire catchment in Glacier National Park, tapertip hawksbeard was restricted to low-elevation forests and not found in riparian areas, floodplains, or subalpine or alpine forests (Mouw and Alaback 2003). In Jackson Hole Wildlife Park, Wyoming, tapertip hawksbeard occurred along streams and was also found in the understory of lodgepole pine forests (Reed 1952).
Elevation
Tapertip hawksbeard occurs in habitats at elevations of 700 to 10,800 ft (200–3,300 m) (Munz and Keck 1973; Cronquist et al. 1994; Bogler 2006; Welsh et al. 2015; Chambers 2020), but it is most frequent at mid-elevations (Babcock and Stebbins 1938; Smith 1960; Hermann 1966). It occurs from 700 to 7,900 ft (200 to 2,400 m) in Oregon (Chambers 2020). In the Steens Mountain area of Oregon it is scattered in sagebrush scrub below 5,600 ft (1700 m) and is found up to 8,200 ft (2,500 m) (Mansfield 2000).
Soils
In tapertip hawksbeard habitats, soils are typically well–drained, neutral to slightly alkaline (pH 6-7.8), and non–saline with medium to coarse sandy or stony textures (USFS 1937; Stubbendieck et al. 1997; Taylor 1992; Eldredge et al. 2013; LBJWC 2018). Plants have low fertility requirements and are somewhat tolerant of calcareous soils (USDA NRCS 2017). In Grand Teton National Park, Wyoming, tapertip hawksbeard occurred in 60% of mountain big sagebrush and antelope bitterbrush stands and 17 to 20% of low sagebrush or mixed sagebrush stands, where soils were coarsely textured (Sabinske and Knight 1978). In the Wasatch Mountains of Utah, the importance value (a product of constancy and average cover) for tapertip hawksbeard was greater in Utah serviceberry (Amelanchier utahensis) foothills than in Gambel oak (Quercus gambelii)-dominated vegetation when 25 stands were compared (Yake and Brotherson 1979). Soil pH levels, sand content, and bare ground were significantly greater and clay content significantly lower in Utah serviceberry foothills than Gambel oak stands (P ≤ 0.05). Soil pH averaged 7.9 and composition was 64% sand and 25% clay in the Utah serviceberry foothill stands (Yake and Brotherson 1979).
Although tapertip hawksbeard is common in dry habitats with coarse–textured soils, it is also found on some sites with fine–textured soils. In a greenhouse study, tapertip hawksbeard emergence was significantly greater in sandy loam than clay loam soils (P = 0.004) (Rawlins et al. 2009). In a big sagebrush/ bluebunch wheatgrass kipuka north of Blackfoot, Idaho, soils were well–drained silt loams leached of carbonates in the top 14 to 26 in (36–66 cm). In this kipuka, western hawksbeard (Crepis occidentalis) and tapertip hawksbeard made up 5 to 8% of the species composition (Passey and Hugie 1963). In contrast, tapertip hawksbeard was associated with grassland sites on northeastern aspects on soils with high water–holding capacities and clay levels in central Idaho (Mueggler and Harris 1969).
In the Reynolds Creek watershed, Owyhee County, Idaho, tapertip hawksbeard was a common forb in a mountain big sagebrush community on a loamy ecological site at an average elevation of 5,200 ft (1,600 m) that received 48 in (624 mm) of annual precipitation, primarily in winter and spring (Schachtschneider et al. 2024). In the same watershed it was also common on a Wyoming big sagebrush site on a sandy loam ecological site at an average elevation of 2,050 ft (1,221 m) that received 263 mm of annual precipitation, primarily in winter and spring.
Revegetation guidelines recommend tapertip hawksbeard for fine silt loam, silt, and sandy to coarse-textured soils on sites receiving 7 to 20 in (18–50 cm) of precipitation and a minimum of 110 frost free days (Walker and Shaw 2005; Ogle et al. 2012; USDA NRCS 2017). Recommendations for restoration are discussed in the Wildland Seeding and Planting section.
Research examining titanium (Ti) as a soil contamination indicator of plant samples collected for trace element analysis found Ti in tapertip hawksbeard seedling shoots resulting from root uptake averaged 5 mg/kg-1, milling increased Ti by 4 mg/kg-1, and Ti in reagents and on labware added 5 to 6 mg/kg-1 (Cook et al. 2009). Dusting seedlings with soil resulted in elevenfold increases in Ti. Washing procedures failed to remove all soil from the seedlings.

Figure 3. Tapertip hawksbeard growing in Nevada. Photo: USDI BLM NV020 SOS.
Description
Tapertip hawksbeard is a perennial plant that produces a milky sap (USFS 1937; Lambert 2005a; LBJWC 2018). One to five erect stems develop from a caudex and deep, tough, branching taproot (Fig. 3) (USFS 1937; Bogler 2006; Welsh et al. 2015). Stems are stout (2-4 mm diameter), moderately tall (8–33 in [20–85 cm]), and covered with dense, short, soft hairs, especially when young. Upper stem parts become hairless with age (Babcock and Stebbins 1938; Cronquist et al. 1994; Stubbendieck et al. 1997; Welsh et al. 2015). Stems branch near or above the midpoint and end in erect inflorescences with yellow flowers. Rosettes of coarse, pinnately lobed leaves surround the stem bases (Bogler 2006). Plants are described as short-lived (Eldredge et al. 2013) and long-lived (Ogle et al. 2012; Tilley et al. 2013), but no study directly investigating life span was found.
Plants produce basal and stem leaves that are 3 to 16 in (8–40 cm) long and 0.4 to 4 in (1–10 cm) wide, alternate, and petiolate. Basal and lower stem leaves are pinnately lobed with 5 to 10 pairs of lobes and are more numerous (2–7) and larger than the reduced upper stem leaves that have only one to three lobes (USFS 1937; Babcock and Stebbins 1938; Hermann 1966; Munz and Keck 1973; Bogler 2006; Welsh et al. 2015). Lobes of the lower leaves are toothed or cleft about halfway to the midvein (Cronquist et al. 1994). Leaves, especially the lower ones, are gray-green, long-tapered at the tips, and coated with cottony pubescence (Craighead 1963; Lesica 2012; Hitchcock and Cronquist 2018).
Tapertip hawksbeard produces an abundance of flower heads (20–100 or more) (Fig. 4) arranged in flat-topped, compound corymbiform inflorescences with numerous flower heads (Munz and Keck 1973; Spellenberg 2001; Bogler 2006). Flower heads are narrow, erect, and made up of 5 to 12 petal–like, perfect, ray flowers with five-toothed tips (USFS 1937; Babcock and Stebbins 1938; Hermann 1966; Anderson and Holmgren 1976; Welsh et al. 2015; LBJWC 2018). Flower heads are enclosed by conspicuous, smooth, equal inner bracts that are at least twice as long as the few very short glabrous to slightly pubescent outer bracts (USFS 1937; Spellenberg 2001; Lesica 2012). Fruits are 6 to 9 mm long, nearly cylindric cypselas (false achenes) with pointed tips and a white pappus with barbed bristles. Fruits are yellow, buff, or brown with 10 to 12 longitudinal ribs. Length of the ribs equals or exceeds pappus length (USFS 1937; Babcock and Stebbins 1938; Munz and Keck 1973; Cronquist et al. 1994; Bogler 2006). Tapertip hawksbeard fruits are cypselas, but they are often referred to as achenes or seeds because seeds are not removed from the fruits in the cleaning process. Throughout this review, the term seed will refer to the fruit or cypsela and the seed it contains.

Figure 4. Tapertip hawksbeard inflorescences, note the indeterminate flowering. Photo: USDI BLM OR030 SOS.
Diploid and polyploid plants are not always distinguishable morphologically (Babcock and Stebbins 1938). Flower heads of diploid plants typically contain five, or rarely six, individual flowers from bases measuring 0.9 to 1.2 cm tall. Polyploid plants have as many as 8 to 12 individual flowers from bases up to 1.5 cm tall. In areas where the distributions of diploid forms overlap those of polyploid forms and limestone hawksbeard (Crepis intermedia), distinguishing the forms and species can be difficult (Babcock and Stebbins 1938; Hitchcock et al. 1955). Diploid and polyploid plants can quite reliably be distinguished through stomata and pollen comparisons. Stomates are larger and pollen is absent or irregular in polyploid forms (Babcock and Stebbins 1938).
Reproduction
Tapertip hawksbeard reproduces from seed (Fig. 5), which is produced sexually in diploid plant forms and asexually through apomixis in polyploid forms (Babcock and Stebbins 1938).
For individual plants, the flowering window is quite narrow, but flowers are produced from May to August across the species’ range (Munz and Keck 1973; Anderson and Holmgren 1976; Cronquist et al. 1994; Bogler 2006; Tilley et al. 2012). Phenology varies from year to year depending on weather conditions. Blaisdell (1958) monitored tapertip hawksbeard for 9 to 16 years in threetip sagebrush at the USFS Sheep Experiment Station (USSES) near Dubois, Idaho. Seasonal development of tapertip hawksbeard was slow. Blooms generally appeared in mid-June, and seeds dispersed in early August. Over the 9-year period, the average date for growth initiation was April 12, flower stalk appearance May 11, first bloom appearance June 18, full flowering July 1, seed dispersal began July 16 and ended August 9, and plants were dry on August 3. The variation in date range for these stages was 42 days for appearance of flower stalks, 40 days for first blooms, 43 days for full blooming, and 20 days for ripe seed (Blaisdell 1958).

Figure 5. Open and closed tapertip hawksbeard seed heads. Photo: USDI BLM CA370 SOS.
Heavy spring precipitation may severely limit tapertip hawksbeard reproduction (Bates et al. 2006). In field experiments, tapertip hawksbeard failed to reproduce when 80% of total moisture was delivered in the spring (April-July) at a northern Great Basin site. After 6 years of the experiment, reproductive success was greater for winter precipitation (≥60%) than for normal (≤30%) and spring precipitation (0%) treatments. Plant density was significantly greater (P < 0.05; about 0.2 plant/ft² [2 plants/m²]) where 80% of total moisture was delivered in winter (October-March) than for spring moisture treatments (< 0.09 plant/ft² [1 plant/m²]) or when moisture timing and amount matched the area’s 50-year average (Bates et al. 2006).
Breeding System
Sexual diploid plants are widely distributed and highly variable morphologically. Polyploid plants are often apomictic. They fail to produce pollen or they produce highly abnormal pollen the further they are from diploid plants. New apomictic forms may occasionally be adapted to new habitats and allow for “aggressive” colonization of open or new sites (Babcock and Stebbins 1938).
Pollination
Sexual tapertip hawksbeard plants are likely insect pollinated. Twenty–eight bees were collected from 169 plants growing at three sites in Utah and Idaho (Cane and Love 2016). Of these, 40% were mason bees (Osmia spp.), 11% mining bees (Andrena spp.), 11% bumblebees (Bombus spp.), 10% furrow bees (Halictus spp.), 8% sweat bees (Agapostemon spp.), 8% Lasioglossum spp., and less than 5% of each of the following genera were collected: plasterer bees (Colletes spp.), long–horned bees (Eucera spp.), mason bees (Hoplitis spp.), leafcutter bees (Megachile spp.), and cuckoo bees (Nomada spp.) (Cane and Love 2016). Tapertip hawksbeard also serve as nectar sources for butterflies (Tilley et al. 2019).
Ecology
Tapertip hawksbeard tolerates early seral to climax conditions (Eggler 1941) but is often more abundant in mid-seral communities (Daubenmire 1975; Koniak 1985; Walker and Shaw 2005). It has been described as a short–lived (Eldredge et al. 2013) and as a long–lived perennial (Ogle et al. 2012), but no study directly investigating its lifespan was found. Plants require full sun or partial shade and soils with rooting depths of at least 10 inches (USDA NRCS 2017).
In early succession on volcanic deposits at Craters of the Moon National Monument, tapertip hawksbeard occurred on cinder cones before any shrub established (Eggler 1941). When mountain big sagebrush and western juniper stands were compared in west–central Owyhee County, Idaho, tapertip hawksbeard occurred in both early to mid-seral and climax stands. It was present in 5 of 11 seral stands where tree age ranged from 33 to 88 years and crown cover averaged 22% and in 1 of 7 climax stands where tree age ranged from 185 to 365 years and crown cover averaged 47%. Frequency of tapertip hawksbeard was 15% in seral stands and 12% in the climax stand (Burkhardt and Tisdale 1969). In the Shoshone Mountains of central Nevada, tapertip hawksbeard occurred in mountain big sagebrush where cover of singleleaf pinyon and Utah juniper was low to moderate, but it was absent from where woodland cover was high (Allen and Nowak 2008).
Because tapertip hawksbeard is rarely present in high abundance, study area size and sampling effort may influence ecological findings. Tapertip hawksbeard was restricted to mid-seral conditions when a single burned area and adjacent habitats were evaluated in the Sweetwater Mountains of California (Koniak and Everett 1982), but it occurred across the range of early, mid-, and late seral communities when 21 burned sites in California and Nevada were evaluated (Table 1; Koniak 1985). Tapertip hawksbeard was found only in mid-seral shrub-tree communities in the Sweetwater Mountains near Walker, California. Plants were absent from early seral sites dominated by grasses, forbs, and juvenile shrubs.
Table 1. Occurrence and cover of tapertip hawksbeard across a post–fire chronosequence in Nevada and California (Koniak 1985).
|
Seral stage, yrs since fire |
Early, 1 |
Early-Mid, 4–8 |
Mid, 15–17 |
Mid-Late, 22–60 |
Unburned |
|
Frequency (%) |
40a* |
24b |
29b |
36ab |
13c |
|
Cover (%) |
5ab |
6ab |
16a |
<5b |
<5b |
*Values in the same row followed by a different letter are significantly different (P <0.05).
and absent from late seral tree-shrub sites with high densities of singleleaf pinyon and limited understories. Cover of tapertip hawksbeard was 0.3% in mid-seral sites with good shrub growth and scattered trees (Koniak and Everett 1982). In a study of 21 burns in Nevada and California, tapertip hawksbeard was found in early to late-seral communities burned 1 to 60 years prior and in long unburned singleleaf pinyon-Utah juniper (Table 1). Frequency was greatest in the earliest seral communities, but cover was greatest in mid- to late-seral communities (Koniak 1985).
Seed And Seedling Ecology
It is common to find seeds destroyed by insect larvae in nearly every tapertip hawksbeard flower head (Babcock and Stebbins 1938), and reductions in seed yield can be substantial (Tilley et al. 2012).
Tapertip hawksbeard fruits contain pappi with barbed bristles (Bogler 2006) that facilitate long-distance wind dispersal. Hawksbeard (Crepis spp.) seed (0.8 seed/liter) was collected in September from ant mounds that were 6.6 feet (2 m) or further from hawksbeard plants. Seeds were recovered from mounds in 3-year old burned big sagebrush-yellow rabbitbrush (Chrysothamnus viscidiflorus) shrublands in southeastern Idaho (Nowak et al. 1990).
Dormancy varies among seed lots, which may be a product of population differences, site differences, seed age, storage conditions, or other factors. Some sources suggest seed is non-dormant (Young and Young 1986; Karrfalt and Vankus 2012), but others indicate that cold stratification is needed for germination (Jensen and Stettler 2012). Regardless of stratification needs, it is unlikely that tapertip hawksbeard banks seed. Studies conducted by the Great Basin Research Center in Ephraim, Utah, found that most tapertip hawksbeard seed imbibed water quickly when available. When bagged seeds were buried in September and retrieved in late November, about 7% had germinated, about 80% were fully imbibed, and the rest were dead (Vernon et al. 2007).
Establishment of tapertip hawksbeard from seed requires seed burial or protection from the elements (Eckert et al. 1986; Rawlins et al. 2009). In experimental seedings in big sagebrush communities north of Elko, Nevada, establishment was best (47-52 seedlings/9 m²) when seeds were placed in naturally occurring trenches or cracks in the soil surface and seedlings were protected from cattle for 1 year. Establishment was significantly higher for seeds in natural cracks and trenches (25-26 seedlings/9 m²) than on the soil surface (7-9 seedlings/9 m2) even without protection from cattle (P ≤ 0.05) (Eckert et al. 1986). Establishment is considered rapid from wind-dispersed seed (Laycock 1967), but seedling vigor is considered low and spread from seed slow (USDA NRCS 2017).
Disturbance Ecology
Tapertip hawksbeard is common in early post-disturbance communities. It often recovers to pre-fire abundance levels within the first few years following fire (Poreda and Wullstein 1994). It may take longer to re-colonize abandoned crop fields. In southeastern Washington, tapertip hawksbeard was found in older abandoned crop fields (abandoned 39 years) but not in younger fields (abandoned 1–12 years) (Daubenmire 1975). Tapertip hawksbeard is sensitive to grazing, especially spring grazing by sheep (Mueggler 1950). It may also have reduced abundance in drought years. At the US Sheep Experiment Station (USSES) near Dubois, Idaho, density of tapertip hawksbeard in a severe drought year (1934) was just 9.4% of that in a normal precipitation year, a significant decrease (P < 0.01) (Pechanec et al. 1937).
Fire Response. In general, fires do not cause dramatic changes in tapertip hawksbeard abundance. Frequency of tapertip hawksbeard was greatest in the earliest seral communities, but cover was greatest in mid- to late-seral communities in a 60-year post-fire chronosequence in California and Nevada (Koniak 1985).
In the small number of studies available, summer fires seemed to do more damage to tapertip hawksbeard abundance than spring fires. Spring fires favored tapertip hawksbeard establishment and growth at sites in California and Nevada. At Lava Beds National Monument, northern California, spring prescribed fires favored post-fire colonization by tapertip hawksbeard in areas where pre-fire mountain big sagebrush vegetation was dominated by native species (Ellsworth and Kauffman 2017). Biomass and nitrogen concentration of plant tissue were increased for tapertip hawksbeard after a spring prescribed fire in mountain big sagebrush and singleleaf pinyon vegetation in the Shoshone Mountains of central Nevada. This suggests that fire increased nutrient availability, which was in turn utilized by tapertip hawksbeard to increase growth by the second post-fire year. The fire was set in May when soil and fuel moistures were high, resulting in a patchy burn (Rau et al. 2008).
The fire response was more variable following summer fires. Frequency and cover of tapertip hawksbeard were higher on unburned than on 1-year-old burned sites near Heber Valley, Utah (Poreda and Wullstein 1994). The August fire was described as severe. In Gambel oak vegetation, tapertip hawksbeard frequency was 47.1% on unburned and 3.6% on burned and cover was 1.4% on unburned and 0.02% on burned plots. In big sagebrush-grassland vegetation, tapertip hawksbeard frequency was 54.5% on unburned and 4.9% on burned and cover was 2.2% on unburned and 0.07% on burned plots one year after fire (Poreda and Wullstein 1994). Cover of tapertip hawksbeard (averaging 1%) was not different before, 1 year after, or 5 years after a moderate- to low-severity August fire in late-seral Idaho fescue-bluebunch wheatgrass vegetation in northeastern Oregon (Johnson 1998). Following a summer fire in big sagebrush/Thurber’s needlegrass (Achnatherum thurberiana) vegetation north of Reno, Nevada, frequency of tapertip hawksbeard was 3% in the first post-fire year but 0% in the third and fourth post-fire years (Young and Evans 1978).
Although slight damage to tapertip hawksbeard has been reported from fall fires in sagebrush (Wright cited in Britton and Ralphs 1979), frequency of tapertip hawksbeard was not significantly different (P = 0.6) on unburned and 1- to 2-year-old burned plots following a fall prescribed fire on South Steens Mountain in Oregon (McDowell 2000).
Bates and Davies (2022) conducted a low severity fall burn in an intact low sagebrush (Artemisia arbuscula var. arbuscula)/Idaho fescue steppe in Harney County, Oregon in 2012. Burned and control sites were at elevations of 1,670 to 2,560 ft (,1430-1,540 m) with a west aspect and 1-10% slope. Soils were extremely cobbly loam (18 in [45 cm] to fracked bedrock) with silty clay loam inclusions (25 in [62 cm] to fracked bedrock). Most precipitation at the site occurred from winter through mid-May, with crop year precipitation averaging11 in (280 mm), but precipitation was below average during much of the 2012 to 2020 study period. From 2012 to 2017 tapertip hawksbeard cover (P=0.047) and density (P=0.010) on the burned site were twice that of the control, but yield did not differ significantly (Bates and Davies 2022).
Spring and fall prescribed burns were conducted in 1988 and 1989 at the Hart Mountain National Antelope Refuge in Lake county, Oregon, in an effort to reduce shrub cover, primarily sagebrush and bitterbrush, and improve brood-rearing habitat for greater sage-grouse (Pyle 1992). Cichorieae species, including tapertip hawksbeard, were among the primary foods of sage-grouse chicks at this site. Their frequency did not vary among treatments following the 1988 burns but was greater on fall- than spring-burns or controls in 1989 (P = 0.02).
With increasing time since fire, tapertip hawksbeard abundance was greater on burned than unburned plots following summer fires in two southern Idaho studies. Cover of tapertip hawksbeard was greater on 6-year-old burned (1.9%) than unburned (0.6%) plots in sagebrush steppe and western juniper woodlands that burned on July 6 on the Owyhee Plateau (Weiner et al. 2016). Production of tapertip hawksbeard was greater on burned than unburned plots 12 years after an August fire in big sagebrush-grasslands on the Upper Snake River Plain in Clark County, Idaho (Blaisdell 1953). Production of tapertip hawksbeard was 0.3 lb/ac (0.6 kg/ha) on unburned, 1.4 lbs/ac (1.6 kg/ha) on lightly burned, 0.9 lb/ac (1 kg/ha) on moderately burned, and 3 lbs/ac (3.4 kg/ha) on severely burned plots. Light burns consumed only big sagebrush leaves, moderate burns consumed leaves and small stems, and severe burns consumed everything including the trunk or main stem of big sagebrush plants (Blaisdell 1953).
Wildlife And Livestock Use
Tapertip hawksbeard is utilized by wildlife and livestock. It is an important greater sage-grouse (Centrocercus urophasianus) food and is highly preferred by domestic sheep.
Bighorn sheep (Ovis canadensis), elk (Cervus canadensis), mule deer (Odocoileus hemionus), and white–tailed deer (O. virginianus) utilize tapertip hawksbeard at low levels (Robinson 1937; Smith 1960; Schallenberger 1966; Lauer and Peek 1976). In big game winter range in the Sun River Canyon of west-central Montana, tapertip hawksbeard made up less than 3% of total observed instances of winter (January-March) feeding by bighorn sheep, elk, mule deer, and white-tailed deer. In this area, frequency of tapertip hawksbeard was 15% or less (Schallenberger 1966). Tapertip hawksbeard was described as a decreaser when bluebunch wheatgrass-needle and thread (Hesperostipa comata) grasslands near Jackson Hole, Wyoming, receiving elk winter use were compared with exclosures. Sites were compared in the summer and only used by native ungulates, primarily elk (Smith 1960). In northwestern Wyoming grasslands and shrublands, year-round elk and summer cattle grazing was evaluated through the use of exclosures. Tapertip hawksbeard was significantly reduced (P < 0.05) on elk-grazed but not significantly changed on cattle-grazed sites (Jones 1965). Bighorn sheep, mule deer, and cattle utilized tapertip hawksbeard at low levels from December to June along the East Fork of the Salmon River in Idaho. In this study area, tapertip hawksbeard abundance was low (Lauer and Peek 1976).
Tapertip hawksbeard is also utilized somewhat by small mammals. It was found in hay piles or food caches constructed by American pikas (Ochotona princeps) in Washoe County, Nevada (Beever et al. 2008). It was also found in 6% of the stomachs of Townsend’s ground squirrel (Spermophilus townsendii) collected in March from a Sandberg bluegrass-dominated site in southwestern Idaho, but not from collections made in May or June (Yensen and Quinney 1992).
Like other Asteraceae forbs with milky juice, tapertip hawksbeard is an important food source for greater sage–grouse hens and chicks (Klebenow and Gray 1968; Drut et al. 1994; ; Dumroese et al. 2015; Luna et al. 2018) and is often found in utilized habitats (Wik 2002; Lambert 2005a). Tapertip hawksbeard was commonly found in greater sage-grouse brood-rearing habitats in south-central Owyhee County, Idaho, and north-central Elko County, Nevada (Wik 2002).
Frequency of tapertip hawksbeard in greater sage-grouse diets was high in studies conducted in Idaho and Oregon. Tapertip hawksbeard made up 25% of the volume and frequency of crops from 2-week old chicks (n=4), 50% frequency and a trace of the volume from crops of 3-week-old chicks (n=2), and 14% frequency and a trace of the volume of crops of 5-week-old chicks (n=7) taken from big sagebrush-grasslands in Clark County, Idaho. Researchers noted that decreased use with increasing chick age may have been related to plant phenology. Tapertip hawksbeard in this area matured and dried within a short period (Klebenow and Gray 1968). Hawksbeard leaves were preferentially consumed by pre-laying greater sage–grouse hens in southeastern Oregon from March 4 to April 8 (Barnett and Crawford 1994). Hawksbeard frequency was 37 to 62% and dry weight was 3 to 14% in crops depending on the vegetation type and year. Hawksbeards were the top or second most selected food by greater sage-grouse in big sagebrush and low sagebrush vegetation (Barnett and Crawford 1994). Hawksbeards were also selected by greater sage-grouse chicks in big sagebrush and bitterbrush communities in southeastern Oregon. Chicks ranged from 1 to 11 weeks old but most were 3 to 10 weeks old. In crops, the frequency of hawksbeards was 29 to 69% and dry mass was 4 to 5% depending on the vegetation type. In the study area, the frequency of hawksbeards was 1 to 2% (Drut et al. 1994).
In brood-rearing habitat at the Hart Mountain National Antelope Refuge in Lake County, Oregon, tapertip hawksbeard and other members of the Cichorieae tribe were primary selected foods of greater sage-grouse chicks in sagebrush and grassland communities (Pyle 1992). Annual forbs were significantly more frequent in the diet of chicks 1-5 weeks old, which consumed greater (P<0.05) amounts of annual forbs compared to chicks 6-10 weeks old (36% compared to 14% of diet by mass). Perennials received greater use by chicks 6-10 weeks old compared to chicks 1-5 weeks old (55% compared to 30% by mass) though not significantly so. Tapertip hawksbeard provided 4.1% of the mass of sage-grouse chicks diet with a frequency of 34%. It was not among the nine most used forbs for 1–5-week-old chicks, but was one of the six forbs consumed in the greatest amounts by 6-10 week old chicks, providing 11.9% (SE 3.7%) of the diet with a frequency of 1.1% (SE=0.6%) in the community (Pyle 1992).
Livestock. Cattle and sheep feed on tapertip hawksbeard, but preference is highest for sheep, although it has been said to be relished by both cows and sheep (Hermann 1966). Palatability of plants is highest in spring and early summer (USFS 1937). Palatability of tapertip hawksbeard is considered poor to good for horses, fair to good for cattle, and excellent for sheep (USFS 1937; Craighead et al. 1963; Stubbendieck et al. 1997).
Decreases in tapertip hawksbeard with sheep grazing were commonly reported (Ellison 1954; Mueggler and Stewart 1980). At the USSES, tapertip hawksbeard decreased dramatically or was lost ith heavy continuous spring grazing paired with late fall grazing and heavy early spring grazing paired with late fall grazing by sheep. Palatability of tapertip hawksbeard to sheep was highest of all forbs listed (Craddock and Fording 1938). In other studies at the USSES, sheep use of tapertip hawksbeard was 33 to 56% in threetip sagebrush-grasslands grazed heavily in the summer. Use was greater in early summer than late summer (Harniss and Wright 1982).
Tapertip hawksbeard is especially sensitive to spring grazing by sheep. In several studies conducted at the USSES, spring sheep grazing resulted in reduced tapertip hawksbeard abundance (Mueggler 1950; Laycock 1967; Bork et al. 1998; Roselle et al. 2010). Production of tapertip hawksbeard decreased from 24 to 3 lbs/ac (27 to 3.4 kg/ha) when sites at the USSES fall grazed for 20 years were switched to spring sheep use for 13 years (Laycock 1967). Production was also much lower when spring and fall and fall only grazed sites were compared. Prior to initiation of the study, abundance of tapertip hawksbeard was very similar on the two sites, but after 25 years of grazing, production was 2.5 lbs/ac (2.8 kg/ha) in the spring and fall grazed area and 15 lbs/ac (17 kg/ha) in the fall only grazed area. Stocking rate in the fall-grazed area averaged 42.6 sheep d/ac and in the spring and fall grazed area averaged 19.2 spring and 10 fall sheep d/ac (Mueggler 1950).
Tapertip hawksbeard abundance was also often lower on cattle-grazed than ungrazed sites. Density was significantly greater (P < 0.05) in a 16-year-old exclosure (0.9 plant/ft² [6.9/m²]) than in a cattle-grazed semi-arid rangeland (0.1 plant/ft² [1.3/m²]) in northern Nevada (Bethlenfalvay and Dakessian 1984). The area was heavily grazed for 58 years, but in the 6 years prior to evaluating effects, grazing was changed to a 3-pasture rest-rotation regime. This regime included a year of rest, a year of April to December use, and a year of mid-July to January use at a stocking rate of 9.5 ac (3.85 ha) per animal unit month (AUM) (Bethlenfalvay and Dakessian 1984). Tapertip hawksbeard frequency was not significantly different (P > 0.05) on 1- to 3-year rested or immediately spring or summer grazed post-fire sites in the Owyhee Mountains of southwestern Idaho (Clark et al. 2016, 2018). Frequency differences for tapertip hawksbeard on rested and ungrazed sites were not statistically significant. Summer post-fire grazing was light with about 30 days of use occurring mostly in July at a stocking rate of 11 ha/AUM (Clark et al. 2018). Spring post-fire grazing use was mostly in May at stocking rates of 45 ha/AUM for the first 15 days and 23 ha/AUM for the second 15 days (Clark et al. 2016).
Nutritional Value
Several studies have evaluated the nutritive value of tapertip hawksbeard as a forage over time and with site changes. Hawksbeard plants collected in March or early April in sagebrush habitats in southeastern Oregon averaged 30% crude protein, 0.8% calcium, and 0.5% phosphorus (Barnett and Crawford 1994). At the USSES, crude protein decreased and calcium increased as tapertip hawksbeard plants aged. Phosphorus fluctuations were more variable but tended to increase from mid-May through the end of June (Table 2; Blaisdell et al. 1952).
Table 2. Seasonal changes in chemical composition of tapertip hawksbeard growing at the USSES near Dubois, Idaho (Blaisdell et al. 1952).
|
1942 Date |
Crude protein (%) |
Ca (%) |
P (%) |
|
April 30 |
18.8 |
0.93 |
0.39 |
|
May 6 |
18.2 |
0.87 |
0.23 |
|
May 15 |
16.0 |
1.15 |
0.30 |
|
May 27 |
12.6 |
1.76 |
0.46 |
|
June 4 |
12.1 |
1.85 |
0.48 |
|
June 29 |
6.6 |
2.23 |
0.56 |
When 1- and 2-year-old burned and unburned plots were compared during the late greater sage-grouse brood rearing period, crude protein was greater for flowers and leaves of burned plants than unburned plants (Table 3; McDowell 2000).
Table 3. Average nutritional and energy content of tapertip hawksbeard plants on burned and unburned sites sampled during the late greater sage-grouse brood-rearing period (late-July–early August) on South Steen Mountain, Oregon (McDowell 2000).
|
Treatment |
Ca(%) |
P (%) |
Crude protein (%) |
Gross energy (cal/g) |
|
Flowers¹ |
||||
|
Unburned |
0.85c |
0.40b |
11.3a |
4155.4a |
|
1-year-old burn |
0.65a |
0.46a |
11.8b |
4266.4a |
|
2-year-old burn |
0.73b |
0.46a |
12.5c |
4145.7a |
|
Leaves |
||||
|
Unburned |
2.09a |
0.26a |
10.4a |
3847.7a |
|
1-year-old burn |
2.24a |
0.24a |
12.8b |
3918.8c |
|
2-year-old burn |
2.07a |
0.29b |
14.3c |
3896.2b |
¹Values for flowers or leaves within a column followed by different letters are significantly different (P = 0.1).
Ogle and Brazee (2009) provided dry weight conversion factors for tapertip hawksbeard in Idaho based on phenological stage at the date of harvest:
Green before flowering: 20-25%
Full bloom, petals falling: 30-40%
Fruit ripening: 35%
Fruit ripe or fall dormancy: 40%
Seed dissemination or winter dormancy: 50%
Ethnobotany
Limited food and medicinal uses of tapertip hawksbeard are reported in the literature. The Karuk people indigenous to California peeled tapertip hawksbeard stems and ate them raw. The Shoshoni sprinkled pulverized root pieces into the eye to dislodge foreign objects and sooth eye inflammations. They also used a poultice of seeds or entire plants to relieve breast pain and induce milk flow following child birth (Moerman 2003). The Shoshone name for tapertip hawksbeard, yhambahwuhr, translates to standing carrot. It was used medicinally and for ceremonies (Friday and Scasta 2020).
Horticulture
The bright yellow clusters of flowers are attractive (Welsh et al. 2015).
Revegetation Use
To improve forage habitat for pollinators and wildlife, tapertip hawksbeard is a restoration species to consider (Eldredge et al. 2013; Dumroese et al. 2016). Although seedling growth is often slow, plants are considered long–lived by those with experience growing the species (Ogle et al. 2012). Once established, long-distance wind dispersal is encouraged by the production of light-weight seeds with pappi (Bogler 2006). Tapertip hawksbeard has been identified as an important species for seed development because of its importance to greater sage-grouse (Lambert 2005a). Current seeds supplies of tapertip hawksbeard are limited and generally from wildland collections.
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Barga et al. (2020) analyzed collections of tapertip hawksbeard and other priority forb species harvested by the BLM Seeds of Success program. Collected seeds are used for seed banking, immediate restoration, or seed increase in agricultural seed fields. The range size for tapertip hawksbeard was estimated to be 899,186 km2 with 11.15 % impacted by fire from 2000 to 2018. Climate at the collection sites was compared to climate across the species range and to restoration sites within the species range. For the 35 tapertip hawksbeard collections, minimum and maximum temperatures and annual precipitation were similar at the collection sites and ecological sites, but summer precipitation was less. When collection sites were compared to the restoration sites, minimum temperature and summer precipitation were similar, but annual precipitation was greater and maximum temperature lower at the collection sites. Barga et al. (2020) emphasized the importance of carefully selecting populations for seed collection to provide supplies of adapted seed for disturbed areas.
Seed Sourcing
Because empirical seed zones are not currently available for tapertip hawksbeard, generalized provisional seed zones developed by Bower et al. (2014), may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat:moisture index). In Figure 6, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site–specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Climate Smart Restoration Tool (Richardson et al. 2024) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
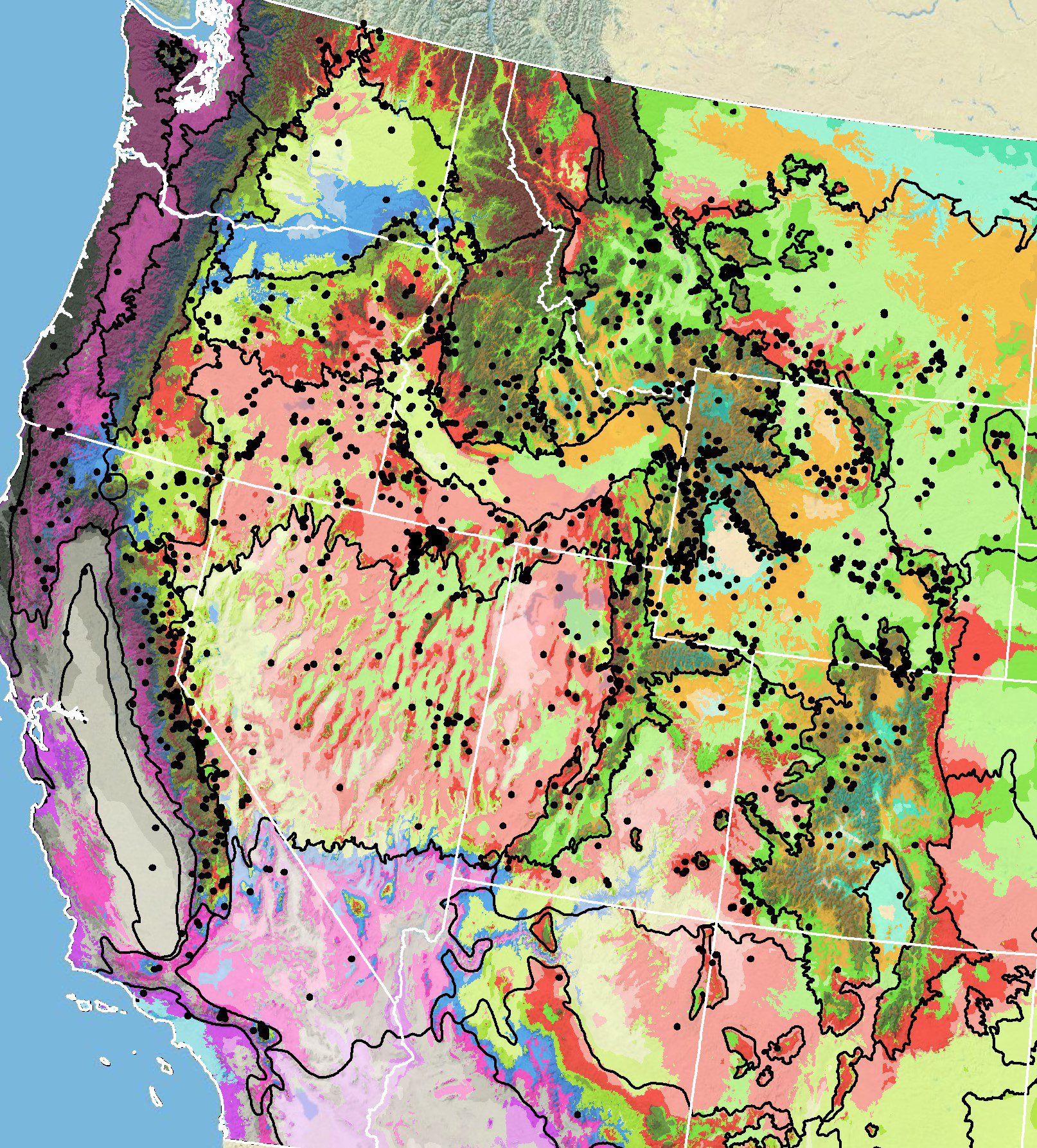
Figure 6. Distribution of tapertip hawksbeard (black circles) based on geo-referenced herbarium specimens and observational data from 1881 to 2023 (CPNWH 2017; SEINet 2017; USGS 2017). Generalized provisional seed zones (colored regions; Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USGS.
Releases
As of 2018, there had been no tapertip hawksbeard germplasm releases.
Wildland Seed Collection
Mature tapertip hawksbeard seed is available from mid-June through mid-July (Fig. 7). Presence of a fluffy white pappus is a good indication that seed is ripe. Harvesting seed before the pappus opens dramatically increases the amount of non-viable seed collected (Tilley et al. 2012). Because insect damage to seeds is common, evaluating the damage in potential collection areas can help to plan efforts for maximum seed yield. In wildland stands in central Utah, the percentage of insect-infested flower heads ranged from 0.9 to 80%, and the percentage of seed damage ranged from 0.7 to 22% depending on the site and year (Anderson 2009). Common seed predators included tephritid flies (Campiglossa spp.) and snout moths (Phycitodes albatella) (Anderson 2009). In 2012 it was reported that wildland collections were not typically available from commercial sources, and contract collecting costs exceeded $100/lb (Tilley et al. 2012).

Figure 7. Uncleaned tapertip hawksbeard seed collected from wildland stands. Photo: USFS, Bend Seed Extractory.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (UCIA 2015; Young et al. 2020). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre–collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow–outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
For individual tapertip hawksbeard plants, the flowering and seed ripening window is quite narrow, but phenology timing varies from year to year depending on weather conditions (Blaisdell 1958; Bates et al. 2006). Presence of a fluffy white pappus is a good indication that seed is ripe (Tilley et al. 2012). For maximum seed viability of wildland harvests, seed should be collected gently by hand, taking seed only from the open heads, and leaving seed in unopened heads for later collection (S. Jensen, USFS, personal communication, July 2018).
Based on 44 wildland seed collection records from 8 consecutive years (2009–2016), the earliest collection made by BLM SOS field crews was June 6, 2013 from a 3,900-ft (1,200 m) elevation site in Malheur County, Oregon. The latest collection was made September 26, 2016 from a 5,700-ft (1,700 m) site in Washoe County, Nevada. The date range for the largest collection year (17 collections in 2010), was July 7 at 5,200 ft (1,600 m) in Humboldt County, Nevada, and August 4 at 5,180 ft (1,580 m) in Malheur County, Oregon (USDI BLM SOS 2017). Phenology variations by year and weather conditions are also discussed in the Reproduction section.
Collection Methods
Staff and researchers at the USDA FS Provo Shrub Sciences Laboratory (PSSL) in Utah, developed several tools and techniques for collecting small lots of tapertip hawksbeard (Jensen 2004). They built seed collection hoppers by sewing heavy-duty nylon cloth to a round frame such that the midpoint of the cloth dropped to form a basket to capture seeds. Hoppers also included adjustable straps to be worn around the collector’s neck and back to distribute the weight and bulk of the hopper. For plants with open seed heads, badminton racquets were used to sweep ripe seeds into the hopper. If conditions were windy however, seeds were hand–plucked from the heads (Jensen 2004).
Because tapertip hawksbeard seeds are easily wind-dispersed, collecting seed heads early in the day or early in the ripening process when heads are closed is an option (Fig. 8). However, Jensen (2004) found that viability of closed seed heads was 40% and of open seed heads was 61%. Comparisons of the collections were made on the same site on the same day.

Figure 8. Tapertip hawksbeard producing seed heads in the Ruby Mountains of Nevada. Photo: S. Jensen, USFS.
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of tapertip hawksbeard.
Post-Collection Management
Because seed insects are common, seed lots should be frozen or pesticide treated as soon as possible following thorough drying and before storing or processing.
Seed Cleaning
Tapertip hawksbeard seed can be cleaned of chaff and seed contaminants without any mechanical equipment, but if pappus removal is necessary, some mechanical processing is likely necessary (Fig. 9).

Figure 9. Cleaned tapertip hawksbeard seed. Photo: USFS PSSL, SOS.
At the PSSL, staff cleaned small seed lots of tapertip hawksbeard of chaff and seed contaminants by tossing seed against a steeply inclined board covered in felt material. The contaminant grass seeds, which were commonly squirreltail (Elymus elymoides) and cheatgrass (Bromus tectorum) for Utah collections, stuck to the felt while the tapertip hawksbeard seed bounced off or quickly fell when the board was shaken or tapped. This cleaning process did not remove the pappus, which was left intact for laboratory seed studies (Jensen 2004).
For internal seed studies and uses requiring pappus removal, the following two guidelines were described for small seed lots:
Process seed using a brush machine with the gate closed to remove the pappus. Final cleaning can be done using a multi–deck air screen cleaner with a 4-mm hole screen on top, 1.4-mm slot screen on the bottom, and light air (Tilley et al. 2012).
Process seed through a brush machine (Westrup Model LA-H, Westrup A/S, Slagelse, Denmark, in this case) using a #10 mantel and medium speed. Air-screen the seed using an office clipper with a 1/14 X 1/2 slot top screen, 1/18 round bottom screen, and medium speed and air (Barner 2007).
Seed Storage
Tapertip hawksbeard seed is orthodox, meaning seed can be dried and stored. Seed longevity increases with reductions in both moisture content and storage temperature (RGB Kew 2018). No reports of seed viability retention with duration of storage were available.
Seed Testing
There is no Association of Official Seed Analysts (AOSA) rule for testing germination of tapertip hawksbeard or any members of the Crepis genus (AOSA 2023).
Viability Testing
The Association of Official Seed Analysts does not provide guidelines for testing viability of tapertip hawksbeard (AOSA 2010). A tetrazolium chloride viability testing procedure, however, was developed by the International Seed Testing Association for the Crepis genus (Moore 1985). Their method prescribed imbibition for 6-18 hrs, followed by cutting the seed completely longitudinally through the midsection of the distal half or cutting the seed along its entire length through the midsection while avoiding cutting into the embryonic axis. Seeds are then stained in TZ for 6-24 hours. The embryo is then exposed by pressing it through the distal opening. Viable embryos are completely stained, except for potential necrosis of the distal one-third of the radical, or the distal one-third to one-half of the cotyledons.
Germination Biology
Chilling requirements for germination vary among seed lots, which may reflect population variation, seed lot age, storage conditions, or other factors. Some indicate that tapertip hawksbeard seed germinates without pretreatment (Young and Young 1986; Karrfalt and Vankus 2012), but several others report that cold-stratification is necessary for good germination (Vernon et al. 2007; Jensen and Stettler 2012; Tilley et al. 2012). In a review of available germination protocols, Karrfalt and Vankus (2012) reported that tapertip hawksbeard germinated well at 50, 59, and 68 ºF (10, 15, 20 ºC) with or without 4 weeks of prechilling. Substantial cold stratification may be required for some seed lots. Researchers at the PSSL reported that germination largely failed without cold stratification (Jensen and Stettler 2012). At the NRCS Plant Materials Center (IDPMC) in Aberdeen, Idaho, germination of tapertip hawksbeard was 0 to 11% without stratification and as high as 75% after 8 months of cold stratification 34 ºF (1 ºC) in moist peat moss (Tilley et al. 2012).
Kildisheva et al. (2019b) examined the germination of tapertip hawksbeard and other forbs considered high priority species for restoration in the Great Basin. Tapertip hawksbeard seed was collected from Lassen County, California, at an elevation of 5,600 ft (1,707m). Seed was maintained at 59 oF (15 oC) and 15% relative humidity during 3-4 weeks of drying followed by 3 months of storage in sealed laminated foil bags. Seeds were permeable to water, fill was 95% (cut test), and seed mass 3.0 ± 0.03 mg. Embryos were linear and fully developed with embryo/seed length (fresh seeds) 0.7 ± 0.02. Constant temperature germination percentage based on viability (28-day incubation on a 12-hr light/dark cycle) was improved by incubation at 10, 15, 20 (P<0.001), and 25 oC (P<0.01) compared to 5 oC. Based on embryo type, permeability and germination response over the temperature range, dormancy was classified as physiological (Kildisheva 2019b).
Incubation in 1 mM GA3 at each of the five temperatures improved germination (P<0.05) and reduced the time to germination by 4.8 days across all temperatures compared to germination on water agar (Kildisheva 2019b). Germination was not improved by incubation in 1 µM karrikinolide. Results indicated that although treatment with GA3 could improve germination at higher temperatures, establishment during warmer periods would require adequate soil moisture for seedling survival.
When incubated at alternating temperature regimes of 59/41, 68/50, and 77/59 oF (15/5, 20/10, and 25/15 oC) on water agar or water agar plus 1 mM GA3, germination was greatest at 20/10 oC, and was improved by 77% averaged across the three temperatures (Kildisheva et al. 2019a).
When seeds were stratified at 3 +2C with low light for 1, 2, 3, or 6 months, then incubated at the three temperature alternations, dormancy loss was first noted at 2 months. Near complete germination resulted after 3 months at all three temperature regimes. The results indicate that dormancy loss widened the temperature range supporting germination, enabling stratified seeds to germinate more rapidly in spring as temperatures increase, but seeds germinating prematurely following shorter periods of stratification may not survive (Kildisheva et al. 2019a).
Afterripening effects were examined by storing seeds at RH 50%, 25/15oC in low light (12 h:12 h) for 0, 1, 2, 3, 6, and 12 mo to simulate seedbed conditions (Kildisheva et al. 2019a). Seeds were then incubated at the three temperature regimes. Germination varied, but was generally higher at 20/10oC, but generally remained below the baseline (20/10 oC at 0 months) at 15/5 or 25/15 oC. The authors hypothesized that such responses might spread recruitment events within a growing season.
The effectiveness of cold, dry storage for conserving viability was examined by drying seeds at 15% RH and storing them in sealed laminated bags at -18 oC for 0, 1,2,3,6, and 12 months followed by incubation at the three temperature alternations (Kildisheva 2019a) . At 20/10 oC, baseline germination (0 months storage) was 53% and germination ranged from 38 to 68%. Germination at the other two regimes remained below the baseline and did not exceed 45%. Changes in germination over time indicated the difficulty in predicting germination behaviour of seeds that have been stored for long periods.
Wildland Seed Yield And Quality
Post–cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 4. The results indicate that tapertip hawksbeard seed can generally be cleaned to high levels of purity and that viability and fill of fresh seed can be variable (USFS BSE 2017).
Seeds are small, averaging about 150,000/lb (333,000/kg) (Table 4). Other sources, including the IDPMC with more than 20 accessions, report similar values ranging from 85,800 to 261,000 seeds/lb (188,900–575,000 seeds/kg) (Tilley et al. 2012; USFS GBNPP 2014; SER, INSR, RBGK 2023).
Table 4. Seed yield and quality of tapertip hawksbeard seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Seed lot characteristic |
Mean |
Range |
Samples (no.) |
|
Bulk weight (lbs) |
1.41 |
0.02-24.86 |
76 |
|
Clean weight (lbs) |
0.15 |
0.003-1.85 |
76 |
|
Clean–out ratio |
0.16 |
0.006-0.70 |
76 |
|
Purity (%) |
96 |
28-99 |
76 |
|
Fill (%)¹ |
88 |
12-99 |
74 |
|
Viability (%)² |
93 |
61-98 |
48 |
|
Seeds/lb |
171,177 |
94,382–294,500 |
76 |
|
Pure live seeds/lb |
147,918 |
90,600–218,079 |
48 |
¹100 seed X–ray test
² Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment is able to handle less clean seed.
Wildland–collected seed viability can be highly variable (Tilley et al. 2012); 80 to 90% viability and 90% purity are recommended marketing standards (Walker and Shaw 2005).
Agricultural Seed Production
Some initial investigations related to establishment and maintenance of tapertip hawksbeard stands for seed production have been made (Fig. 10). The species has proven to be difficult to produce as it is difficult to establish from seed, the seedlings are weak, seed ripening is indeterminate, and seed production is often reduced due to insect predation (Tilley et al. 2012).

Figure 10. Tapertip hawksbeard growing in research plots at Oregon State University’s Malheur Experiment Station in Ontario, OR. Photo: N. Shaw, USFS.
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Seeding
Greenhouse studies indicate that seeds must be buried (Rawlins et al. 2009). Recommended seeding depths ranged from 0.13 to 0.75 in (0.3–1.9 cm) (Vernon et al. 2005; Rawlins et al. 2009). Stand emergence and persistence is unlikely in clay soils and better in sandy loam soils (Rawlins et al. 2009).
Establishment And Growth
Stand establishment is reportedly difficult (Tilley et al. 2012). Direct fall seeding of tapertip hawksbeard in fields in central Utah in 2005 resulted in some emergence and spotty stand establishment, but some plants were flowering in 2006 (Vernon et al. 2007). Jones and Whittaker (2009) reported good emergence following direct fall seeding to establish seed fields in central Utah. Flea beetle (Chrysomelidae) infestations in the establishment year, however, decimated the crop.
Pollinator Management
In studies at the USDA ARS Bee Biology and Systematics Lab in Logan, Utah, researchers increased managed populations of mason bees (Osmia montana) on tapertip hawksbeard (Cane 2005). This mason bee is an easily managed pollinator and can be transported to field locations in portable ground nests or hives to encourage pollination of sexual tapertip hawksbeard stands (Cane 2008; Cane et al. 2012).
Pest Management
Tapertip hawksbeard vegetation, flowers, and seeds host to a variety of pests.
The following fungi utilize tapertip hawksbeard as a host: Aecidium crepidicola, Cercospora stromatis, Erysiphe cichoracearum, Phyllosticta eximia, Puccinia crepidis-acuminatae, P. crepidis-montanae, P. hieracii, and P. stipae (Reed 1913; Wehmeyer 1947; Farr and Rossman 2024).
Insect larvae (tephritid flies [Campiglossa spp.] and snout moths [Phycitodes albatella] are frequent in seed heads, and insect damage can significantly reduce seed yields (Anderson 2009; Tilley et al. 2012). Indian rock buprestid (Acmaeodera idahoensis) was collected from tapertip hawksbeard flowers in Crook County, Oregon (MacRae and Basham 2013). In central Utah, a systemic pesticide application of imidacloprid was effective in reducing seed damage on one of three sites (Anderson 2009).
Seed Harvesting
Although some tapertip hawksbeard plants flowered the year after planting in central Utah fields (Vernon et al. 2007), most report that it takes 3 to 5 years before seeded plants produce seed (Jensen 2011; Tilley et al. 2012). Because flowering and seed ripening are indeterminate, multiple seed harvests may be necessary for maximum seed yield and to obtain maximum genetic diversity (Tilley et al. 2012).
Because seed is easily wind-dispersed and the seed ripening window for plants and populations can be narrow, close monitoring is necessary and harvesting should begin before all seed heads have expanded if harvesting by flail vac, suction, or racquet and hopper methods (S. Jensen, USFS, personal communication, July 2018).
Nursery Practice
Success has been limited in production of tapertip hawksbeard nursery stock. Researchers reported that tapertip hawksbeard failed to produce the mass of roots necessary to hold the plant media and make for easy transplanting (Vernon et al. 2005). Tilley et al. (2012) reported that the long delicate taproot produced by tapertip hawksbeard seedlings was easily damaged during transplanting and that outplanting of bare root and container stock had variable success. Studies did not allow researchers to evaluate the time to seed production from container stock (Tilley et al. 2012).
Seed mixes. Tapertip hawksbeard is typically recommended as part of a wildland seed mix, often making up a minor proportion (<10%). Full seed rates reported were: 18 seeds/ft² (198/m²) at the 1lb (0.45 kg) pure live seed (PLS) rate/ac, and a pure stand seeding rate of 3 lbs PLS/ac (3.4 kg/ha) (Ogle et al. 2012), and for 25 to 30 PLS/ft (100/m) at 12-in row spacing is 7 PLS lb/ac (8 kg/ha) (Tilley et al. 2012). For a seed mixture, adjust these rates according to the desired proportion. In big sagebrush seed mixes, tapertip hawksbeard was recommended at 0.2 PLS lb/ac (0.2 kg/ha) where 7.8 to 8.4 PLS lbs/ac (8.7–9.4 kg/ha) of shrubs, grasses, and forbs were to be drill seeded (Lambert 2005b).
Seeding. Greenhouse studies conducted to develop seeding depth recommendations for seed fields and wildlands indicated that seed must be covered, but depth recommendations varied [0.13–0.75 in (0.3–2 cm)] (Vernon et al. 2005; Rawlins et al. 2009). Shallower recommendations [0.13–0.5 in (0.3–1.3 cm)] were more common in the literature (Rawlins et al. 2009; Ogle et al. 2012; Tilley et al. 2012). When seeding depths of 0 to 1 in (25 mm) were evaluated in sandy loam and clay loam soils, emergence was greatest at 0 to 0.6 cm, but survival to 45 days was significantly lower with surface seeding (P = 0.017) (Rawlins et al. 2009). Emergence was greater in sandy loam than clay loam soils (P = 0.004). At the conclusion of the study, researchers recommended planting tapertip hawksbeard 0.3 to 0.6 cm deep on sandy soils and avoiding seeding in clay soils, where emergence was less than 12% (Rawlins et al. 2009). In germination trials conducted by the Great Basin Research Center, seeding depths of 0.25 to 0.75 in (0.6–2 cm) were considered best (Vernon et al. 2005).
Seed burial and protection from trampling was best for seedling establishment in experimental seedings in big sagebrush communities north of Elko, Nevada (Eckert et al. 1986). Seedling establishment was best (0.47–0.52 seedlings/ft² [5.2–5.8/m²]) when seeds were placed in naturally occurring trenches or cracks in the soil surface on sites protected from cattle for 1 year. Establishment was significantly higher (2.7–2.9 seedlings/m²) for seeds in natural cracks and trenches than for those on the soil surface (0.8–1 seedling/m²) even with cattle use. Some natural protection of seedlings, which came from seeds falling into natural soil crevices, was most important to establishment (Eckert et al. 1986). At least two full growing seasons of grazing protection are recommended on newly seeded sites to allow for full stand establishment (Tilley et al. 2012).
Establishment largely failed and no seedlings survived a year when tapertip hawksbeard was seeded into Wyoming big sagebrush vegetation in Toole County, Utah. Tapertip hawksbeard was broadcast seeded (season not reported) at 0.5 PLS/ft² (5.2/m²) and had less than 1% establishment when seeding was followed by Ely chaining (Gunnell and Summers 2016).
Planting. In one study, outplanting of nursery stock was unsuccessful in north Logan, Utah. Whitcomb (2011) had difficulty producing tapertip hawksbeard seedlings in the greenhouse (see Nursery Practice section above). When the few greenhouse-grown seedlings were planted in plots at the Utah State University Green Canyon Ecology Center in May, all died within the 3 weeks of discontinuing watering, which occurred in mid-June (Whitcomb 2011).
Wildland Seeding And Planting
Fall seeding tapertip hawksbeard as part of a seeding mixture is recommended for fine sandy loams, silts, coarse-textured, or gravelly soils receiving at least 8 to 20 in (200–500 mm) of precipitation (Walker and Shaw 2005; Vernon et al. 2007; Ogle et al. 2012; Tilley et al. 2012; 2013).
Tilley et al. (2013) recommend seeding at a depth of one-fourth to one-half inch. Thet recommended that the full rate for seeding (3 lbs/acre) be adjusted depending upon the other components of the seed mix. Seedling development was rated weak.

Figure 11. Tapertip hawksbeard transplant growing in research plots near Nephi, UT. Photo: S. Jensen, USFS.
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers, Robert Johnson, Brigham Young University and Hilary Parkinson.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Allen, E.A.; Nowak, R.S. 2008. Effect of pinyon-juniper tree cover on the soil seed bank. Rangeland Ecology and Management. 61(1): 63-73.
Anderson, B.A.; Holmgren, A.H. 1976. Mountain plants of northeastern Utah. Logan, UT: Utah State University, Extension Services. 148 p.
Anderson, V.J. 2009. Impacts of predispersal seed predation on seed production of Wyethia amplexicaulis, Agoseris glauca, and Crepis acuminata. In: Shaw, N.L.; Pellant, M., eds. Great Basin Native Plant Project: 2008 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 71-75.
Applegate, E.I. 1938. Plants of the Lava Beds National Monument, California. The American Midland Naturalist. 19(2): 334-368.
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Association of Official Seed Analysts [AOSA]. 2023. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Babcock, E.B.; Stebbins, G.L. 1938. The American species of Crepis: Their interrelationships and distribution as affected by polyploidy and apomixis. Pub. No. 504. Washington, DC: Carnegie Institution of Washington. 199 p.
Barga, S.C.; Olwell, P.; Edwards, F.; Prescott, L.; Leger, E.A. 2020. Seeds of Success: A conservation and restoration investment in the future of U.S. lands. Conservation Science and Practice. 2(7): e209.
Barner, J. 2007. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Crepis acuminata Nutt. seeds. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2018 April 2].
Barnett, J.K.; Crawford, J.A. 1994. Pre-laying nutrition of sage grouse hens in Oregon. Journal of Range Management. 47(2): 114-118.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Bates, J.D.; Boyd, C.S.; Davies, K.W. 2020. Longer-term post-fire succession on Wyoming big sagebrush steppe. International Journal of Wildland Fire. 29(3): 229-239.
Bates, J.D.; Davies, K.W. 2022. Early succession following prescribed fire in low sagebrush (Artemisia arbuscula var. arbuscula) steppe. 82(1): 50-66.
Bates, J.D.; Svejcar, T.; Miller, R.F.; Angell, R.A. 2006. The effects of precipitation timing on sagebrush steppe vegetation. Journal of Arid Environments. 64(4): 670-697.
Beever, E.A.; Wilkening, J.L.; McIvor, D.E.; Weber, S.S.; Brussard, P.F. 2008. American pikas (Ochotona princeps) in northwestern Nevada: A newly discovered population at a low-elevation site. Western North American Naturalist. 68(1): 8-14.
Bethlenfalvay, G.J.; Dakessian, S. 1984. Grazing effects on mycorrhizal colonization and floristic composition of the vegetation on a semiarid range in northern Nevada. Journal of Range Management. 37(4): 312-316.
Blackburn, W.H.; Eckert, R.E.J.; Tueller, P.T. 1969. Vegetation and soils of the Cow Creek watershed. R-48. Reno, NV: University of Nevada, Agricultural Experiment Station. 80 p.
Blaisdell, J.P. 1953. Ecological effects of planned burning of sagebrush-grass range on the upper Snake River Plains. Tech. Bull. 1975. Washington, DC: U.S. Department of Agriculture. 39 p.
Blaisdell, J.P. 1958. Seasonal development and yield of native plants on the Upper Snake River Plains of Idaho and their relation to climatic factors, especially precipitation and temperature. Tech. Bull. 1190. Washington, DC: U.S. Department of Agriculture. 68 p.
Blaisdell, J.P.; Wiese, A.C.; Hodgson, C.W. 1952. Variations in chemical composition of bluebunch wheatgrass, arrowleaf balsamroot, and associated range plants. Journal of Range Management. 5(5): 346-353.
Bogler, D.J. 2006. Crepis. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 19 Magnoliophyta: Asteridae, part 6: Asteraceae, part 1. New York, NY: Oxford University Press: 214-228.
Bork, E.W.; West, N.E.; Walker, J.W. 1998. Cover components on long-term seasonal sheep grazing treatments in three-tip sagebrush steppe. Journal of Range Management. 51(3): 293-300.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Britton, C.M.; Ralphs, M.H. 1979. Use of fire as a management tool in sagebrush ecosystems. In: The sagebrush ecosystem: A symposium: Proceedings; 1978 April; Logan, UT. Logan, UT: Utah State University, College of Natural Resources: 101-109.
Burkhardt, J.W.; Tisdale, E.W. 1969. Nature and successional status of western juniper vegetation in Idaho. Journal of Range Management. 22(4): 264-270.
Cane, J.H. 2005. Pollinator and seed predator studies. In: Shaw, N.L.; Pellant, M., eds. Great Basin Native Plant Project: 2006 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 23-26.
Cane, J.H. 2008. Pollinating bees crucial to farming wildflower seed for U.S. habitat restoration. In: James, R.; Pitts-Singer, T., eds. Bees in agricultural ecosystems. Oxford, UK: Oxford University Press: 48-64.
Cane, J.H.; Love, B. 2016. Floral guilds of bees in sagebrush steppe: Comparing bee usage of wildflowers available for postfire restoration. Natural Areas Journal. 36(4): 377-391.
Chambers, K.L. 2020. Dicots: Asteraceae. In: Meyers, S.C.; Jaster, T; Mitchell, K.E.; Harvey, T.; Hardison, L.K., eds. Flora of Oregon. Volume 2: Dicots A-F. Fort Worth, TX: Botanical Research Institute of Texas (BRIT): 152-370.
Clark, P.E.; Williams, C.J.; Kormos, P.R.; Pierson, F.B. 2018. Postfire grazing management effects on mesic sagebrush-steppe vegetation: Mid-summer grazing. Journal of Arid Environments. 151(2018): 104-112.
Clark, P.E.; Williams, C.J.; Pierson, F.B.; Hardegree, S.P. 2016. Postfire grazing management effects on mesic sagebrush-steppe vegetation: Spring grazing. Journal of Arid Environments. 132(2016): 49-59.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Cook, L.L.; McGonigle, T.P.; Inouye, R.S. 2009. Titanium as an indicator of residual soil on arid-land plants. Journal of Environmental Quality. 38(1): 188-190.
Craddock, G.W.; Fording, C.L. 1938. The influence of climate and grazing on spring-fall sheep range in southern Idaho. Tech. Bull. 600. Washington, D.C.: U.S. Department of Agriculture, Forest Service. 43 p.
Craighead, J.J.; Craighead, F.C. Jr.; Davis, R.J. 1963. A field guide to Rocky Mountain wildflowers from northern Arizona and New Mexico to British Columbia. Boston, MA: Houghton Mifflin Company. 277 p.
Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K. 1994. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Volume 5: Asterales. Bronx, NY: The New York Botanic Garden. 496 p.
Daubenmire, R. 1975. Plant succession on abandoned fields, and fire influences, in a steppe area in southeastern Washington. Northwest Science. 49(1): 36-48.
Drut, M.S.; Pyle, W.H.; Crawford, J.A. 1994. Technical note: Diets and food selection of sage grouse chicks in Oregon. Journal of Range Management. 9(1): 90-93.
Dumroese, R.K.; Luna, T.; Pinto, J.R.; Landis, T.D. 2016. Forbs: Foundation for restoration of monarch butterflies, other pollinators, and greater sage-grouse in the western United States. Native Plants Journal. 36(4): 499-511.
Dumroese, R.K.; Luna, T.; Richardson, B.A.; Kilkenny, F.F.; Runyon, J.B. 2015. Conserving and restoring habitat for greater sage-grouse and other sagebrush-obligate wildlife: The crucial link of forbs and sagebrush diversity. Native Plants Journal. 16(3): 276-299.
Eckert, R.E.; Peterson, F.F.; Meurisse, M.S.; Stephens, J.L. 1986. Effects of soil-surface morphology on emergence and survival of seedlings in big sagebrush communities. Journal of Range Management. 39(5): 414-420.
Eggler, W.A. 1941. Primary succession on volcanic deposits in southern Idaho. Ecological Monographs. 11(3): 277-298.
Eldredge, E.; Novak-Echenique, P.; Heater, T.; Mulder, A.; Jasmine, J. 2013. Plants for pollinator habitat in Nevada. Tech. Note NV 57. Reno, NV: U.S. Department of Agriculture, Natural Resources Conservation Service. 65 p.
Ellison, L. 1954. Subalpine vegetation of the Wasatch Plateau, Utah. Ecological Monographs. 24(2): 89-184.
Ellsworth, L.M.; Kauffman, J.B. 2017. Plant community response to prescribed fire varies by pre-fire condition and season of burn in mountain big sagebrush ecosystems. Journal of Arid Environments. 144(2017): 74-80.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Farr, D.F.; Rossman, A.Y. 2024. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://fungi.ars.usda.gov/
Franklin, J.F.; Dyrness, C.T. 1973. Natural vegetation of Oregon and Washington. Gen. Tech. Rep. PNW-8. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station. 452 p.
Friday, C.; Scasta, J.D. 2020. Eastern Shoshone and Northern Arapaho traditional ecological knowledge (TEK) and ethnobotany for Wind River Reservation rangelands. Ethnobiology Letters. 11(1): 14-24.
Goodrich, S.; Neese, E. 1986. Uinta Basin flora. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Region. 320 p.
Gunnell, K.; Summers, D. 2016. Native forb increase and research at the Great Basin Research Center. In: Kilkenny, F.; Edwards, F.; Malcomb, A., eds. Great Basin Native Plant Project: 2015 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 96-108.
Harniss, R.O.; Wright, H.A. 1982. Summer grazing of sagebrush-grass range by sheep. Journal of Range Management. 35(1): 13-17.
Hermann, F. 1966. Notes on western range forbs: Cruciferae through Compositae. Agric. Handb. 293. Washington, DC: U.S. Department of Agriculture, Forest Service. 365 p.
Hitchcock, C.L.; Cronquist, A. 2018. Flora of the Pacific Northwest: An illustrated manual. Second Ed. Giblin, D.E.; Legler, B.S.; Zika, P.F.; Olmstead, R.G., eds. Seattle, WA: University of Washington Press. 882 p.
Hitchcock, C.L.; Cronquist, A.; Ownbey, M.; Thompson, J.W. 1955. Vascular plants of the Pacific Northwest. Part 3: Saxifragaceae to Ericaceae. Seattle, WA: University of Washington Press. 614 p.
Jensen, S. 2004. Racquets, hoppers, and felt boards – Low-tech devices for processing seeds. Native Plants Journal. 5(1): 50-51.
Jensen, S. 2011. Selecting and growing Great Basin natives. In: Shaw, N.L.; Pellant, M., eds. Great Basin Native Plant Project: 2010 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 64-68.
Jensen, S.; Stettler, J. 2012. Applying provisional seed zones to Great Basin forb production and cultural practice notes. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 70-84.
Johnson, C.G. 1998. Vegetation response after wildfires in national forests of northeastern Oregon. R6-NR-ECOL-TP-06-98. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Region. 128 p.
Johnson, C.G.; Swanson, D.K. 2005. Bunchgrass plant communities of the Blue and Ochoco Mountains: A guide for managers. Gen. Tech. Rep. PNW-GTR-641. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. 119 p.
Jones, C.; Whittaker, A. 2009. Native plant material development and seed and seeding technology for native Great Basin forbs and grasses. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Selection and Increase Project: 2008 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 45-48.
Jones, W.B. 1965. Response of major plant species to elk and cattle grazing in northwestern Wyoming. Journal of Range Management. 18(4): 218-220.
Karrfalt, R.; Vankus, V. 2012. Development of germination protocols, seed weight, purity, and seed conditioning/cleaning protocols for Great Basin grasses and forbs. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Selection and Increase Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 36-39.
Kildisheva, O.A.; Erickson, T.E.; Kramer, A.T.; Zeldin, J.; Merritt, D.J. 2019a. Optimizing physiological dormancy break of understudied cold desert perennials to improve seed-based restoration. Journal of Arid Environments. 170: 104001.
Kildisheva, O.A.; Erickson, T.E.; Madsen, M.D.; Dixon, K.W.; Merritt, D.J. 2019b. Seed germination and dormancy traits of forbs and shrubs important for restoration of North American dryland ecosystems. Plant Biology. 21: 458-469.
Klebenow, D.A.; Gray, G.M. 1968. Food habits of juvenile sage grouse. Journal of Range Management. 21(2): 80-83.
Koniak, S. 1985. Succession in pinyon-juniper woodlands following wildfire in the Great Basin. Great Basin Naturalist. 45(3): 556-566.
Koniak, S.; Everett, R.L. 1982. Seed reserves in soils of successional stages of pinyon woodlands. The American Midland Naturalist. 108(2): 295-303.
Lady Bird Johnson Wildflower Center [LBJWC]. 2018. Crepis acuminata Nutt. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants-main [Accessed 2018 April 2].
Lambert, S. 2005. Guidebook to the seeds of native and non-native grasses, forbs and shrubs of the Great Basin. Boise, ID: U.S. Department of the Interior, Bureau of Land Management, Idaho State Office. 136 p.
Lambert, S.M. 2005b. Seeding considerations in restoring big sagebrush habitat. In: Shaw, N.L.; Pellant, M.; Monsen, S.B., eds. Sage-grouse habitat restoration symposium proceedings; 2001 June 4-7; Boise, ID. Proc. RMRS-P-38. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 75-80.
Lauer, J.L.; Peek, J.M. 1976. Big game-livestock relationships on the bighorn sheep winter range, East Fork Salmon River, Idaho. Bull. 12. Moscow, ID: University of Idaho, Idaho Forest Wildlife and Range Experiment Station. 44 p.
Lavin, M.; Wojciechowski, M.F. 2002. Vascular flora of the Sweetwater Mountains of California and Nevada. 24 p. http://bristleconecnps.org/native_plants/checklists/SweetwatersChecklistJun2002.pdf [Accessed 2023 October 10].
Laycock, W.A. 1967. How heavy grazing and protection affect sagebrush-grass ranges. Journal of Range Management. 20(4): 206-213.
Lesica, P.; Lavin, M.; Stickney P.F. 2012. Manual of Montana vascular plants. Fort Worth: Botanical Research Institute of Texas. 779 p.
Luna, T.; Mousseaux, M.R.; Dumroese, R.K. 2018. Common native forbs of the northern Great Basin important for greater sage-grouse. Gen. Tech. Rep. RMRS-GTR-387. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station; Portland, OR: U.S. Department of the Interior, Bureau of Land Management, Oregon-Washington Region. 76 p.
MacRae, T.C.; Basham, J.P. 2013. Distributional, biological, and nomenclatural notes on Buprestidae (Coleoptera) occurring in the U.S. and Canada. The Pan-Pacific Entomologist. 89(3): 125-142.
Majesky, L.; Krahulec, F.; Vasut, R.J. 2017. How apomictic taxa are treated in current taxonomy: A review. TAXON. 66(5): 1017-1040.
Major, J.; Rejmanek, M. 1992. Amelanchier alnifolia vegetation in eastern Idaho, USA and its environmental relationships. Vegetatio. 98(2): 141-156.
Mansfield, D.H. 2000. Flora of Steens Mountain. Corvallis, OR: Oregon State University Press. 410 p.
McDowell, M.K.D. 2000. The effects of burning in mountain big sagebrush on key sage grouse habitat characteristics in southeastern Oregon. Corvallis, OR: Oregon State University. Thesis. 62 p.
Moerman, D. 2003. Native American ethnobotany: A database of foods, drugs, dyes, and fibers of Native American peoples, derived from plants. Dearborn, MI: University of Michigan. http://naeb.brit.org
MontBlanc, E.; Chambers, J.C.; Brussard, P.F. 2007. Variation in ant populations with elevation, tree cover, and fire in a pinyon-juniper-dominated watershed. Western North American Naturalist. 67(4): 469-491.
Montez-Sanchez, J.J.; Miegroet, J.J.; Van, H.; Villalba, J.J. 2017. Effects energy supplementation and time on use of medusahead by grazing ewes and their lambs. Rangeland Ecology and Management. 70(3): 380-387.
Moore, R.P. 1985. Handbook on tetrazolium testing. Zurich, Switzerland: International Seed Testing Association. 99 p.
Mouw, J.E.B.; Alaback, P.B. 2003. Putting floodplain hyperdiversity in a regional context: An assessment of terrestrial-floodplain connectivity in a montane environment. Journal of Biogeography. 30(1): 87-103.
Mueggler, W.F. 1950. Effects of spring and fall grazing by sheep on vegetation of the Upper Snake River Plains. Journal of Range Management. 3(4): 308-315.
Mueggler, W.F.; Harris, C.A. 1969. Some vegetation and soil characteristics of mountain grasslands in central Idaho. Ecology. 50(4): 671-678.
Mueggler, W.F.; Stewart, W.L. 1980. Grassland and shrubland habitat types of western Montana. Gen. Tech. Rep. INT-66. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station. 154 p.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Nowak, R.S.; Nowak, C.L.; DeRocher, T.; Cole, N.; Jones, M.A. 1990. Prevalence of Oryzopsis hymenoides near harvester ant mounds: Indirect facilitation by ants. Oikos. 58(2): 190-198.
Ogle, D.; Brazee, B. 2009. Estimating initial stocking rates. Range Tech. Note 3. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 11 p.
Ogle, D.; St. John, L.; Stannard, M.; Holzworth, L. 2012. Conservation plant species for the Intermountain West. Plant Materials Technical Note 24. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 57 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Passey, H.B.; Hugie, V.K. 1963. Some plant-soil relationships on an ungrazed range area of southeastern Idaho. Journal of Range Management. 16(3): 113-118.
Passey, H.B.; Hugie, V.K.; Williams, E.W.; Ball, D.E. 1982. Relationships between soil, plant community, and climate on rangelands of the Intermountain West. Tech. Bull. No. 1669. Washington, D.C. U.S. Department of Agriculture, Soil Conservation Service. 123 p.
Pechanec, J.F.; Pickford, G.D.; Stewart, G. 1937. Effects of the 1934 drought on native vegetation of the upper Snake River Plains, Idaho. Ecology. 18(4): 490-505.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Poreda, S.F.; Wullstein, L.H. 1994. Vegetation recovery following fire in an oakbrush vegetation mosaic. Great Basin Naturalist. 54(4): 380-383.
Pyle, W.H. 1992. Response of brood-rearing habitat of sage grouse to prescribed burning in Oregon. Corvallis, OR: Oregon State University. Thesis. 47 p.
Rau, B.M.; Chambers, J.C.; Blank, R.R.; Johnson, D.W. 2008. Prescribed fire, soil, and plants: Burn effects and interactions in the central Great Basin. Rangeland Ecology and Management. 61(2): 169-181.
Rawlins, J.K.; Anderson, V.J.; Johnson, R.; Krebs, T. 2009. Optimal seeding depth of five forb species from the Great Basin. Native Plants Journal. 10(1): 33-42.
Reed, G.M. 1913. The powdery mildews: Erysiphacea. Transactions of the American Microscopical Society. 32(4): 219-258.
Reed, J.F. 1952. The vegetation of the Jackson Hole Wildlife Park, Wyoming. The American Midland Naturalist. 48(3): 700-729.
Richardson, B.; Kilkenny, F.; St. Clair, B.; Stevenson-Molnar, N. 2020. Climate Smart Restoration Tool. https://climaterestorationtool.org/csrt/
Robinson, C.S. 1937. Plants eaten by California mule deer on the Los Padres National Forest. Journal of Forestry. 35(3): 285-292.
Roselle, L.; Seefeldt, S.S.; Launchbaugh, K. 2010. Delaying sheep grazing after wildfire in sagebrush steppe may not affect vegetation recovery. International Journal of Wildland Fire. 19(1): 115-122.
Sabinske, D.W.; Knight, D.H. 1978. Variation within the sagebrush vegetation of Grand Teton National Park, Wyoming. Northwest Science. 52(3): 195-204.
Schachtschneider, C.L.; Strand, E.K.; Launchbaugh, K.L.; Jensen, S. 2024. Targeted cattle grazing to alter fuels and reduce fire behavior metrics in shrub-grasslands. Rangeland Ecology and Management. 96: 105-116.
Schallenberger, A.D. 1966. Food habits, range use and interspecific relationships of bighorn sheep in the Sun River area, west-central Montana. Bozeman, MT: Montana State University. Thesis. 44 p.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shiflet, T.N. 1994. Rangeland cover types of the United States. Denver, CO: Society for Range Management. 152 p.
Smith, D.R. 1960. Description and response to elk use of two mesic grassland and shrub communities in the Jackson Hole region of Wyoming. Northwest Science. 34(1): 25-36.
Society for Ecological Restoration; International Network for Seed Based Restoration; Royal Botanic Gardens [SER, INSR, RBGK]. 2024. Seed Information Database (SID). Available from: https://ser-sid.org/
Spellenberg, R. 2001. National Audubon Society field guide to North American wildflowers: Western region, revised edition. New York, NY: Alfred A. Knopf, Inc. 862 p.
Stubbendieck, J.; Hatch, S.L.; Butterfield, C.H. 1997. North American range plants. 5th ed. Lincoln, NE: University of Nebraska Press. 501 p.
Taylor, R.J. 1992. Sagebrush country: A wildflower sanctuary. Missoula, MT: Mountain Press Publishing Company. 211 p.
Tilley, D.; Jensen, S.; St.John, L. 2012. Plant guide: Tapertip hawksbeard (Crepis acuminata Nutt.). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 3 p.
Tilley, D.; Spencer, J.; Cracroft, T.; Brazee, B.; Vaughan, M.; Cruz, J.K. 2019. Creation and management of Utah butterfly habitat. Tech. Note Plant Materials 73. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 20 p.
Tilley, D.; Taliga, C.; Burns, C.; St. John, L. 2013. Plant materials for pollinators and other beneficial insects in eastern Utah and western Colorado. Tech. Note 2C. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 54 p.
Tisdale, E.W.; Hironaka, M.; Fosberg, M.A. 1965. An area of pristine vegetation in Craters of the Moon National Monument, Idaho. Ecology. 46(3): 349-352.
Tueller, P.T.; Eckert, R.E. 1987. Big sagebrush (Artemisia tridentata vaseyana) and longleaf snowberry (Symphoricarpos oreophilus) plant associations in northeastern Nevada. The Great Basin Naturalist. 47(1): 117-131.
U.S. Department of Agriculture, Forest Service [USFS]. 1937. Range Plant Handbook. Washington, DC: U.S. Department of Agriculture, Forest Service. 816 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2021. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Great Basin Native Plant Project [USFS GBNPP]. 2014. Seed weight table calculations made in-house. Report on file. Boise, ID: U.S. Department of Agriculture, Forest Service, Boise Aquatic Sciences Laboratory. Available: https://www.fs.fed.us/rm/boise/research/shrub/Links/Seedweights.pdf
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2017. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2023. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 45 p.
USDI Environmental Protection Agency [USDI EPA]. 2018. Ecoregions. Washington, DC: U.S. Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2023. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Vernon, J.; Meyer, T.; Walker, S.C. 2005. Native plant material development and seed and seeding technology for native Great Basin forbs and grasses. In: Shaw, N.L.; Pellant, M., eds. Great Basin Native Plant Project: 2004 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 14-17.
Vernon, J.; Meyer, T.; Whittaker, A. 2007. Native plant material development and seed and seeding technology for native Great Basin forbs and grasses. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Selection and Increase Project: 2006 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 19-28.
Walker, S.C.; Shaw, N.L. 2005. Current and potential use of broadleaf herbs for reestablishing native communities. In: Shaw, N.L.; Pellant, M.; Monsen, S.B., comps. Sage-grouse habitat restoration symposium proceedings; 2001 June 4-7; Boise, ID. RMRS-P-38. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 56-61.
Wehmeyer, L.E. 1947. Studies on some fungi from northwestern Wyoming. IV. Miscellaneous. Mycologia. 39(4): 463-478.
Weiner, N.I.; Strand, E.K.; Bunting, S.C.; Smith, A.M.S. 2016. Duff distribution influences fire severity and post-fire vegetation recovery in sagebrush steppe. Ecosystems. 19(7): 1196-1209.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Whitcomb, H.L. 2011. Temperature increase effects on sagebrush ecosystem forbs: Experimental evidence and range manager perspectives. Logan, UT: Utah State University. Thesis. 125 p.
Wik, P.A. 2002. Ecology of greater sage-grouse in south-central Owyhee County, Idaho. Moscow, ID: University of Idaho. Thesis. 141 p.
Yake, S.; Brotherson, J.D. 1979. Differentiation of serviceberry habitats in the Wasatch Mountains of Utah. Journal of Range Management. 32(5): 379-383.
Yensen, E.; Quinney, D.L. 1992. Can Townsend’s ground squirrels survive on a diet of exotic annuals? The Great Basin Naturalist. 52(3): 269-277.
Young, J.A.; Evans, R.A. 1978. Population dynamics after wildfires in sagebrush grasslands. Journal of Range Management. 31(4): 283-289.
Young, J.A.; Young, C.G. 1986. Collecting, processing and germinating seeds of wildland plants. Portland, OR: Timber Press. 236 p.
Young, S.A.; Schrumpf, B.; Bouck, M.; Moore, M. 2020. How the Association of Official Seed Certifying Agencies (AOSCA) tracks wildland sourced seed and other plant propagating materials. Logan, UT: Utah State University, Utah Agricultural Experiment Station. 36 p.
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2025. Tapertip hawksbeard (Crepis acuminata). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org