Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
September 2019
Nomenclature
Parsnipflower buckwheat (Eriogonum heracleoides Nutt.) belongs to the Polygonaceae family and the Oligogonum subgenus (Reveal 2005). Nomenclature for subtaxa follows Reveal (2005). Nomenclature for synonyms follows Reveal (2005) and Tropicos (2019).
Family
Polygonaceae – Buckwheat family
Genus
Eriogonum
Species
heracleoides
NRCS Plant Code
ERHE2 (USDA NRCS 2017).
Subtaxa
The Flora of North America (Reveal 2005) recognizes two varieties: heracleoides and leucophaeum.
Synonyms
Eriogonum heracleoides var. micranthum Gand., E. h. var. multiceps Gand., E. h. var. rydbergii Gand., E. h. var. simplex S. Watson ex. Piper, E. h. var. utahensis Gand., E. h. var. viride Gand.
Variety heracleoides: E. angustifolium Nutt., E. heracleoides subsp. angustifolium Piper, E. h. var. angustifolium Torrey & A. Gray, E. h. var. minus Bentham.
Variety leucophaeum: E. caespitosum subsp. ramosum (Piper) S. Stokes.
Common Names
Parsnipflower buckwheat, Wyeth buckwheat, creamy buckwheat, Hercules buckwheat, Indian tobacco, umbrella plant, wild buckwheat, whorled buckwheat (Dayton 1960; Craighead et al. 1963; Camp and Sanderson 2007; James and Nunnallee 2011; Pavek et al. 2012; Luna et al. 2018; UW Ext. 2018).
Chromosome Number
Chromosome numbers were not reported in the literature.
Hybridization
Parsnipflower buckwheat hybridizes with some sulphur-flower buckwheat (E. umbellatum) varieties where distributions overlap. Yellow-flowered parsnipflower buckwheat plants are an indication of this hybridization (Mee et al. 2003; Welsh et al. 2016).
Distribution
Parsnipflower buckwheat is an Inland West species, occurring from British Columbia and northern and central Montana south to eastern California, northern Nevada and Utah, and the northwestern corner of Colorado (Reveal 2005; Welsh et al. 2016). Currently parsnipflower buckwheat occurs as fairly small populations along the northern fringe of the central Great Basin and is more dominant in the Columbia Basin, Snake River Plain, and the Basin and Range of southeast Oregon (J. Johnson, NV Natural Heritage Program, personal communication, August 2019). Variety heracleoides is the most widely distributed and occurs throughout the species range. Variety leucophaeum is restricted to eastern Washington and northern Idaho. It occurs as scattered populations in Adams, Chelan, Douglas, Kittitas, Lincoln, Okanogan, Spokane, Stevens, and Whitman counties in Washington and adjacent Gooding and Kootenai counties in Idaho (Reveal 2005).
Habitat And Plant Associations
Parsnipflower buckwheat is common in big sagebrush (Artemisia tridentata) and antelope bitterbrush (Purshia tridentata)-dominated vegetation in mountain foothills (Fig. 1) at the upper edge of the Wyoming big sagebrush (A. t. subsp. wyomingensis) zone (Ogle et al. 2012; Tilley and Fund 2017). It also occurs in grasslands, mountain brush ecosystems, woodlands, and conifer forests throughout its range (Munz and Keck 1973; Monsen et al. 2004; Reveal 2005; Welsh et al. 2016; Hitchcock and Cronquist 2018). Parsnipflower buckwheat occurs in areas receiving 12 to 25 in (305–635 mm) of annual precipitation (Ogle et al. 2012; Tilley and Fund 2017). It dominates gravelly soils and rocky ridges (Taylor 1992).

Figure 1. Parsnipflower buckwheat growing in mixed grassland/shrubland habitats in Oregon. Photo: USDI BLM OR030 SOS.
Grasslands. Parsnipflower buckwheat is characteristic or dominant in fescue (Festuca spp.) and bluebunch wheatgrass (Pseudoroegneria spicata) grasslands from southern British Columbia to northeastern Oregon and central Idaho (Mueggler and Harris 1969; McLean 1970; McLean and Tisdale 1972). A rough fescue (F. scabrella)-parsnipflower buckwheat habitat type is recognized in southern British Columbia (McLean and Tisdale 1972). An Idaho fescue (F. idahoensis)-parsnipflower buckwheat habitat type occurs between the ponderosa pine (Pinus ponderosa) and Douglas-fir (Pseudotsuga menziesii) zones in the Similkameen Valley, British Columbia, where annual temperature averages less than 42 °F (5.5 °C) (McLean 1970). This habitat type occurs as a man-made climax community beneath powerlines kept free of trees and as a topoedaphic climax on steep south-facing slopes (≤ 6,070 ft [1,850 m]) where precipitation is sufficient for tree growth (McLean 1970). An Idaho fescue-parsnipflower buckwheat habitat type also occurs in the Okanogan Mountains near Spokane, Washington. It occurs within the ponderosa pine and Douglas-fir zones on sites too dry to support tree growth (Daubenmire 1970). In the northern Blue Mountains of Oregon, a bluebunch wheatgrass-Sandberg bluegrass (Poa secunda)-parsnipflower buckwheat plant community occurs between 2,960 and 6,280 ft (900–1,910 m) on south slopes (≤ 32%) with shallow soils (Johnson and Swanson 2005). In surveys of central Idaho mountain grasslands, parsnipflower buckwheat was conspicuous in Idaho fescue-bluebunch wheatgrass-rosy pussytoes (Antennaria rosea) communities and common in bluebunch wheatgrass communities (Mueggler and Harris 1969). In Hells Canyon National Recreation Area in the Wallowa-Whitman National Forest, the bluebunch wheatgrass-parsnipflower buckwheat plant association is the highest elevation bluebunch wheatgrass type (4,300–5,600 ft [1,300–1,700 m]) (Johnson and Simon 1987).
Shrublands. In descriptions of rangeland cover types in the US, parsnipflower buckwheat is common in antelope bitterbrush-Idaho fescue cover types in the Pacific Northwest and mountain big sagebrush cover types in the northern Great Basin (Fig. 2) (Shiflet 1994). On the eastern slope of the Cascade Range, parsnipflower buckwheat is common in antelope bitterbrush/Idaho fescue habitats (Franklin and Dyrness 1973). At Craters of the Moon National Monument in south-central Idaho, parsnipflower buckwheat is common in mountain big sagebrush (Artemisia tridentata subsp. vaseyana)/needle and thread (Hesperostipa comata) and Thurber’s needlegrass (Achnatherum thurberianum) vegetation on sandy, shallow soils (Day and Wright 1985). In a study of dwarf sagebrush vegetation in northern Nevada, parsnipflower buckwheat occurred in habitat types dominated by low sagebrush (A. arbuscula) but not those dominated by black sagebrush (A. nova) (Zamora and Tueller 1973). Parsnipflower buckwheat occurred (1–3% cover) in more than half of 15 Saskatoon serviceberry (Amelanchier alnifolia)-dominated stands on steep, south- to southwest-facing slopes near Victor, Idaho, and adjacent western Wyoming (Major and Rejmanek 1992). In Utah, parsnipflower buckwheat is most abundant in mixed-mountain brush communities in the Wasatch Range (R. Johnson, Brigham Young University, personal communication, September 2019).

Figure 2. Parsnipflower buckwheat growing in big sagebrush habitat in Oregon. Photo: USDI BLM SOS OR010.
Woodlands, forests. Parsnipflower buckwheat occurs in the understory of Rocky Mountain juniper (Juniperus scopulorum) and western juniper (J. occidentalis) woodlands and ponderosa pine and Douglas fir forests. It is common in western juniper woodlands in the Pacific Northwest (Eddleman et al. 1994) and was one of many forbs in the Rocky Mountain juniper/mountain big sagebrush-mountain snowberry (Symphoricarpos oreophilus)/basin wildrye (Leymus cinereus) community type in southeastern Idaho (Rust 1999). In the interior Columbia Basin, the bluebunch wheatgrass-parsnipflower buckwheat habitat type occurs with and without a coniferous overstory (Hessberg et al. 1999). On steep, south-facing slopes above the Spokane River in eastern Washington, parsnipflower buckwheat occurs in open Douglas-fir forests. These forests generally have scant, scattered, xerophytic understory vegetation due to intense insolation and exceedingly dry winds (Turesson 1914).
Elevation
Parsnipflower buckwheat occurs at elevations from 660 to 11,500 ft (200–3,500 m) (Reveal 2005; Welsh et al. 2016). Variety heracleoides occurs from 980 to 11,500 ft (300–3,500 m) and variety leucophaeum from 660 to 6,200 ft (200–1,900 m) (Reveal 2005).
Soils
In parsnipflower buckwheat habitats, soils are often described as dry, shallow, and gravelly to rocky (Fig. 3), but it does occur on soils with textures ranging from clay to loam to sand (Reid et al. 1980; Skinner 2008; Hitchcock and Cronquist 2018).

Figure 3. Parsnipflower buckwheat growing in rocky soils in Oregon. Photo: USDI BLM SOS OR130.
Texture, parent material. In the Similkameen Valley, British Columbia, the Idaho fescue-parsnipflower buckwheat habitat type occurs on black chernozem, sandy loam to loam soils (McLean 1970). The Idaho fescue-parsnipflower buckwheat habitat type in the Okanogan Mountains near Spokane, Washington, occurs on dry, shallow, gravelly, fine sandy loam to loam soils. In eastern Washington, parsnipflower buckwheat was commonly associated with the bluebunch wheatgrass-Idaho fescue habitat type occupying stony soils (Daubenmire 1970). In the Hells Canyon National Recreation Area in Oregon and Idaho, the bluebunch wheatgrass-parsnipflower buckwheat association occurs on sites with silt loam to clay soils, 16 to 40 in (41–102 cm) deep on steep slopes (30–65%) (Johnson and Simon 1987). In southeastern Idaho, parsnipflower buckwheat was one of many forbs in the Rocky Mountain juniper/mountain big sagebrush-mountain snowberry/basin wildrye community type that occurred in mixed carbonate substrates at an average elevation of 6,359 ft (1,938 m) (Rust 1999).
In the Bear River Range of northeastern Utah and southeastern Idaho, parsnipflower buckwheat was characteristic of and almost entirely restricted to quartzite soils when dolomite and quartzite soils were compared (Neely and Barkworth 1984). Quartzite soils averaged 73% sand, 20% silt, and 8% clay. Soil pH, silt content, and percent moisture were significantly lower (P < 0.01) and sand content was significantly higher in quartzite than dolomite soils. Parsnipflower buckwheat was most common on quartzite soils with pH levels of 4.6 to 5.7 (Neely and Barkworth 1984).
When crest, mid-slope, and base locations were compared in Utah’s shrub-dominated foothills, parsnipflower buckwheat cover was significantly greater at the crest (10%) than at the mid- (4.6%) or base (2.2%) slope locations. Crest locations were the most xeric and had soils that were significantly more shallow with the highest percentages of exposed rock and sand, and lowest percentage of clay (Brotherson 1999).
Depth, composition. Although often associated with shallow soils, parsnipflower buckwheat did not occur in the shallowest soils near Davenport, Washington (McColley and Hodgkinson 1970). Vegetation composition and production were compared at sites occurring within 600 ft (180 m) of each other on silt loam soils that varied only in their depth to bedrock. At the shallowest soil site (5 in [13 cm]), dominated by scabland sagebrush (Artemisia rigida) and Sandberg bluegrass, parsnipflower buckwheat did not occur. At the mid-depth soil site (12 in [30 cm]), dominated by bluebunch wheatgrass, parsnipflower buckwheat production was 120 lbs/ac (134 kg/ha). At the deepest soil (25 in [63 cm]) site, dominated by Idaho fescue and threadleaf sedge (Carex filifolia), parsnipflower buckwheat production was 3.3 lbs/ac (3.7 kg/ha) (McColley and Hodgkinson 1970).
In central Utah’s Strawberry Valley, parsnipflower buckwheat was fairly common in sagebrush–grassland vegetation (2.8% cover) but did not occur in basin wildrye communities (Walker and Brotherson 1982). Soils were much shallower in sagebrush (9 in [23 cm]) than in basin wildrye (16 in [41 cm]) (P ≤ 0.1), and badger (Taxidea taxus) activity was 13 times greater in basin wildrye than in sagebrush. However, many soil characteristics were similar in the two communities. Composition of both was 48% sand, 32% silt, 19% clay, 18% gravel and 4 to 5% organic matter. Soil pH was about 6 in both communities and contents of most nutrient were similar, but potassium levels were much lower in sagebrush (281 ppm) than in basin wildrye (585 ppm) (P ≤ 0.05) (Walker and Brotherson 1982).
Temperature. Soil temperatures were monitored for a year (October 1962–August 1963) in eight eastern Washington grassland and shrubland habitats. The Idaho fescue-parsnipflower buckwheat habitat type, common on dry low-elevation sites in Okanogan Mountains, had the coldest soils, which dried deeply in drought conditions (Table 1; Daubenmire 1972).
Table 1. Soil temperatures (°F) for one year (October 6, 1972 to August 17, 1963) in the Idaho fescue-parsnipflower buckwheat habitat type in eastern Washington (Daubenmire 1972).
|
Soil |
Nov |
Dec |
Mar |
Apr |
May |
June |
July |
Aug |
|
––––––––––––––––––––––––––– (°F)––––––––––––––––––––––––––––––––– |
||||||||
|
20 |
45.5 |
39.6 |
39.9 |
43.2 |
52.7 |
59.2 |
59.5 |
64.0 |
|
40 |
49.1 |
42.1 |
37.6 |
40.8 |
48.2 |
53.8 |
55.8 |
59.0 |
Description
Parsnipflower buckwheat is a long-lived, herbaceous or suffruticose perennial with a spreading woody caudex (Taylor 1992; Reveal 2005; Ogle et al. 2012). Plants grow as low spreading mats with erect flowering stems 2 to 30 in (5–75 cm) tall (Fig. 4) (Ogle et al. 2012; Welsh et al. 2016). Flowering stems often have a whorl of 2 to 10 leaflike bracts at their midpoint (Figs. 4 and 5) (Reveal 2005; Welsh et al. 2016). Stems and leaves are typically covered in dense short hairs giving them a gray- to blue-green color (Fig. 4) (Munz and Keck 1973; Ogle et al. 2012; Welsh et al. 2016).
 Figure 4. Parsnipflower buckwheat plant growing in Utah. Photo: USDA FS Rocky Mountain Research Station, Shrub Sciences Laboratory, Provo, UT.
Figure 4. Parsnipflower buckwheat plant growing in Utah. Photo: USDA FS Rocky Mountain Research Station, Shrub Sciences Laboratory, Provo, UT.
Belowground description. Parsnipflower buckwheat plants have extensive root systems. A single plant excavated from a dry prairie in southeastern Washington produced a strong woody taproot (1 in [2.5 cm] diameter), which split into seven equal–sized lateral roots at the 6-in (15 cm) depth. One of the laterals extended 13.5 ft (4 m) from the plant and had a total length of 14.3 ft (4.4 m). All laterals tapered gradually and produced additional lateral roots, which branched up to five times. Plants also had prostrate branches with adventitious roots, which anchored them and extracted moisture from superficial soil layers (Weaver 1915). A single plant excavated from the Boise River Watershed in Idaho produced six primary roots (0.8–1.2 in [2–3 cm] thick), which remained thick down to about 3 ft (1 m) deep. Roots were highly branched and occupied a large portion of an area 4.6 ft (1.4 m) long and 3.3 ft (1 m) wide. Roots reached a maximum depth of 15 ft (4.6 m) and spread a little more than 3.3 ft (1 m) (Woolley 1936).
Aboveground description. Plants often form large spreading mats up to 3.2 ft (0.6 m) across (Weaver 1915; Reveal 2005; Pavek et al. 2012; Welsh et al. 2016). Vegetative stems are persistent (Welsh et al. 2016). Flowering stems are not persistent but are long lasting and give plants their height (<30 in [75 cm]) (Ogle et al. 2012). Leaves are linear to broadly oblanceolate with short petioles (0.1–1.2 in [3–30 mm] long) and at least three and often four times long as wide (Fig. 5) (Munz and Keck 1973; Reveal 2005; Pavek et al. 2012; Welsh et al. 2016; Hitchcock and Cronquist 2018). They occur in loose basal rosettes and are tomentose throughout or at least on the undersides (Munz and Keck 1973; Pavek et al. 2012; Welsh et al. 2016).

Figure 5. Parsnipflower buckwheat (E. h. var. heracleoides) in bloom growing in Utah. Photo: USDI BLM SOS UT931.
Small, cup-shaped flowers 4 to 9 mm wide are clustered in simple or compound umbels (Figs. 5 and 6) (USDA FS 1937; Reveal 2005). Individual flowers have six creamy petal-like structures (calyx), which become reddish with age (Fig. 7) (USDA FS 1937; Taylor 1992). Flowers are typically perfect with nine stamens (Laubengayer 1937; Reveal 2005). Six stamens occur in pairs in an outer whorl and three make up an inner whorl (Laubengayer 1937). Parsnipflower buckwheat produces achenes, one-seeded fruits loosely referred to as seeds (Meyer and Paulsen 2000; Tilley and Fund 2017). Achenes are light to dark brown, 2 to 5.5 mm long, somewhat three-angled with points at both ends, and glabrous except for pubescence at the beak or apex (Munz and Keck 1973; Reveal 2005). Throughout this review, the term seed will be used to refer to the one-seeded achene.
Distinguishing varieties. Variety heracleoides can reliably be distinguished from variety leucophaeum by the whorl of leaflike bracts occurring midway up its flowering stems (Fig. 6) (Reveal 2005). Flowering stems of variety leucophaeum lack bracts entirely or have just one leaflike bract at their mid-length. Variety heracleoides is highly variable and may produce more narrow leaves when precipitation is limited. Plants in the eastern portion of its range have broader leaves (Reveal 2005).

Figure 6. Parsnipflower buckwheat (var. heracleoides) inflorescences on a plant growing in Utah. White arrow is pointing out whorled bracts at midstem. Photo: USDI BLM SOS UT931.

Figure 7. Parsnipflower buckwheat inflorescences and individual flowers on a plant growing in Oregon. Photo: USDI BLM SOS OR030.
Reproduction
Parsnipflower buckwheat reproduces from seed. Flowers (Fig. 7) are usually perfect, but plants are infrequently polygamodioecious, meaning they produce perfect and male flowers or perfect and female flowers (Reveal 2005). Parsnipflower buckwheat typically flowers in the summer, but flowers can appear from May to October (Munz and Keck 1973; Pitt and Wikeem 1990; Reveal 2005). Flowers are insect pollinated and attract a variety of pollinators (see Insects section of Wildlife and Livestock Use).
Ecology
Buckwheat species are early colonizers of disturbed sites. They tolerate harsh growing conditions and may facilitate recruitment of later seral species (Meyer 2008). Parsnipflower buckwheat is drought tolerant, long–lived (Shock et al. 2017), and generally recovers within several years following a top-killing disturbance (Mueggler and Blaisdell 1958; Monsen et al. 2004).
Parsnipflower buckwheat is common in early to mid–seral communities (Eggler 1941; Bunting et al. 1999). At Craters on the Moon National Monument in south-central Idaho, density of parsnipflower buckwheat increased with increasing age of lava flows. Lava flows ranged from young, very xeric surfaces representing primary succession conditions to surfaces colonized by grass and shrub communities. Once crevice plants established on the lava flows, they trapped wind-blown soil and improved growing conditions for other forbs, grasses and then shrubs. Parsnipflower buckwheat was a dominant species in the grass and shrub communities (Eggler 1941).
In western juniper woodlands, parsnipflower buckwheat was most common in early- to mid-seral vegetation dominated by grasses and shrubs. The composition of 40 plots on sites ranging from recently burned to mature woodlands was compared in southwestern Idaho. Parsnipflower buckwheat was primarily associated with grassland and sagebrush communities and was rare or absent in mature woodlands (Bunting et al. 1999). In Owyhee County, Idaho, parsnipflower buckwheat did not occur in climax western juniper woodlands but did occur in seral stands (3–8% frequency). In seral stands, cover of western juniper averaged 22% and mountain big sagebrush averaged 9%. In climax stands, cover of western juniper averaged 47% and mountain big sagebrush averaged 1% (Burkhardt and Tisdale 1969).
Seed And Seedling Ecology
Parsnipflower buckwheat seed generally matures in late summer. Seedlings appear early in the following spring. Although seedling vigor is considered low, survivors can become long-lived plants (Ogle et al. 2012).
Disturbance Ecology
Most studies suggest that although parsnipflower buckwheat is damaged by fire or other disturbances (Seefeldt and Leytem 2011), it often recovers to pre-disturbance levels within a few years (Mueggler and Blaisdell 1958). Recovery can take longer, however, if disturbances are severe or recurring (Blaisdell 1953).
At the US Sheep Experiment Station (USSES) near Dubois, Idaho, parsnipflower buckwheat cover was 4.3% outside and 0% inside sheep bedding use areas (0.2–0.8 ac [0.08–0.32 ha]). Bedding areas were used two to three times in 3 years by 800 to 900 ewes and 1,200 to 1,300 lambs. The vegetation was evaluated in use years 2 and 3. Perennial forb cover, total perennial cover, and total vegetative biomass were all greater outside than within the bedding use areas (Seefeldt and Leytem 2011).
In big sagebrush–grassland vegetation at the USSES, parsnipflower buckwheat production exceeded pre–treatment levels within 3 years of big sagebrush removal treatments (Table 2). There were chemical and mechanical treatments, and sites were rested for 1 year before moderate sheep grazing was resumed on untreated and treated sites. Production of big sagebrush was reduced most by rotobeating and least by the herbicide treatment.
Parsnipflower buckwheat has been described as fire intolerant and experiencing moderate to severe damage when burned (Miller et al. 2014). It has also been described as persistent and increasing after fire (Monsen et al. 2004). In northeastern Oregon, parsnipflower buckwheat had not recovered within 1 year of fire but was much more common within 5 years of burning (Johnson 1998). Recovery took even longer if fires were severe. In an Idaho fescue-bluebunch wheatgrass-silky lupine (Lupinus sericeus) community, cover of parsnipflower buckwheat was 5% before a moderate fire, 2% in the first post-fire year, and 7% in the fifth post-fire year. In mountain big sagebrush/Idaho fescue, cover of parsnipflower buckwheat was 24% before a severe fire and 3% in the first post-fire years. On moderately burned sites in the same vegetation type, cover of parsnipflower buckwheat was 9% before the fire, 5% in the first post-fire year, and 15% in the fifth post-fire year. In Idaho fescue-prairie Junegrass (Koeleria macrantha), cover of parsnipflower buckwheat was 5% before a light fire, 3% in the first post-fire year, and 2% in the fifth post-fire year. For these burned and unburned comparisons, sample sizes were too small for statistical analysis (Johnson 1998).
Production of parsnipflower buckwheat was markedly reduced in the first post-fire year but equaled or exceeded unburned production 12 to 15 years after fire in eastern Idaho (Table 3; Blaisdell 1953). Prescribed fires to remove big sagebrush were conducted in September in Fremont County and August in Clark County. Fire effects were evaluated in lightly to heavily burned areas, where light burns consumed only big sagebrush leaves, moderate burns consumed the smaller big sagebrush branches, and heavy burns consumed the main trunk and stems of big sagebrush (Blaisdell 1953).
At the USSES, changes in parsnipflower buckwheat abundance were similar on burned and unburned plots by the third post-fire year following a prescribed fire in mountain big sagebrush that removed 95% of the biomass and produced temperatures less than 104 °F (40 °C) at 2-in (5 cm) depths (Seefeldt et al. 2007). Four years after a fall prescribed fire in mountain big sagebrush communities in Strawberry Valley, Wasatch County, Utah, parsnipflower buckwheat produced greater cover on burned (0.7%) than unburned (0.1%) plots. The fire consumed all mountain big sagebrush. Cover differences were not compared statistically (Goodrich 2008).
Table 2. Production of parsnipflower buckwheat (lbs/ac) before and 3 years after sagebrush removal treatments at the US Sheep Experiment Station near Dubois, Idaho (Mueggler and Blaisdell 1958).
|
Treatment |
Untreated |
Burned |
Railed |
Rotobeaten |
Herbicide |
|
–––––––––––––––––– Production (lbs/ac) ––––––––––––– |
|||||
|
Pretreatment |
3 |
8 |
2 |
4 |
8 |
|
3 yrs after treatment |
7 |
13 |
10 |
14 |
17 |
Table 3. Comparison of parsnipflower buckwheat production on unburned, lightly, moderately, and heavily burned plots 12 and 15 years after late summer prescribed fire in big sagebrush in eastern Idaho. Differences were not compared statistically (Blaisdell 1953).
|
Location and time since fire |
Unburned |
Lightly burned |
Moderately burned |
Heavily burned |
|
–––––––––––––––Production (lbs/ac) –––––––––––– |
||||
|
Clark Co., 12 yrs after fire |
19.4 |
33.8 |
18.3 |
21.8 |
|
Fremont Co., 15 yrs after fire |
21 |
17 |
24 |
15.1 |
Wildlife And Livestock Use
Parsnipflower buckwheat is an important food and cover source for many wildlife species and is eaten by livestock, especially domestic sheep (Craighead et al. 1963; Ralphs et al. 1991; Ogle et al. 2011). Foliage and flowers are eaten by various large mammals (Holmgren 1954). Seeds are eaten by small mammals and birds (Taylor 1992; Monsen et al. 2004), and pollen and developing seeds are important to many insects (James and Nunnallee 2011; James et al. 2014).
Large mammals. Mountain goats (Oreamnos americanus), mule deer (Odocoileus hemionus), and elk (Cervus canadensis) feed on parsnipflower buckwheat. Field observations made over a single winter in Washington revealed parsnipflower buckwheat made up 5% of mountain goat diets (Anderson 1940). It averaged 10% of spring and fall mule deer diets in grazed and ungrazed grasslands in Asotin, Washington (Wagoner et al. 2013). When elk diets were analyzed from fecal samples collected in the Blue Mountains near La Grande, Oregon, parsnipflower buckwheat made up a high of 3% of the diets in July of 1972 and 4% in June of 1973. (Korfhage et al. 1980). In a subalpine big sagebrush/grassland (6,800–8,000 ft [2,070–2,440 m]) in Oregon’s Whitman National Forest, parsnipflower buckwheat was 9% of early summer elk use (Pickford and Reid 1943).
Bighorn sheep (Ovis canadensis) in British Columbia preferred parsnipflower buckwheat in all but the summer months. In Similkameen Valley, British Columbia, parsnipflower buckwheat made up 4% of winter and spring bighorn sheep diets (Blood 1967). In the transition zone between ponderosa pine and big sagebrush south of Penticton, British Columbia, bighorn sheep diets and grazing effects were evaluated by enclosing 20 bighorn sheep within a 104-ac (42 ha) area (Wikeem and Pitt 1979, 1991, 1992). Frequency of parsnipflower buckwheat was significantly reduced in the enclosure over time (1976–83, P < 0.05) (Wikeem and Pitt 1991). It made up a low percentage of bighorn sheep diets but an even lower percentage of the vegetation composition (Wikeem and Pitt 1979). At the same location in 1977 and 1978, parsnipflower buckwheat was grazed consistently each year, and selection indices were high in all seasons except summer (Table 4; Wikeem and Pitt 1992).
Table 4. Selection of parsnipflower buckwheat by bighorn sheep in an enclosure in transitional vegetation between big sagebrush and ponderosa pine near Penticton, British Columbia.
|
Year |
Summer |
Fall |
Winter |
Spring |
|
Selection index* (frequency of diet/frequency in vegetation) |
||||
|
1977 |
0.9 |
3.3 |
5.9 |
2.6 |
|
1978 |
1.0 |
3.2 |
6.3 |
4.7 |
*Selection index (SI) values greater than 1 suggest selection for and SI values less than 1 suggest selection against parsnipflower buckwheat.
Livestock. Generalizations vary about the palatability and grazing response of parsnipflower buckwheat to livestock (McLean 1970; Hall 1973), but depending on the location and grazing animal, use of and short-term damage to parsnipflower buckwheat can be heavy. At a site south of Sheridan, Montana, 5% of cattle bites were on parsnipflower buckwheat from June 26 to July 16 in big sagebrush-Idaho fescue vegetation (Ralphs and Pfister 1992). From late spring to fall, parsnipflower buckwheat averaged 2% by weight for cattle diets in grasslands at the Starkey Experimental Range, Oregon. In this area, cover of parsnipflower buckwheat averaged 6% and it made up a high of 4% of cattle diets in early summer (Holechek et al. 1982). In a 6-year study of livestock grazing on summer range in Utah, parsnipflower buckwheat use by cattle averaged 23% and by domestic sheep averaged 18% based on herbage removal (Cook 1983). In a subalpine big sagebrush grassland in Oregon’s Whitman National Forest, the estimated utilization of parsnipflower buckwheat was 8% by domestic sheep in early summer (Pickford and Reid 1943). In threetip sagebrush (Artemisia tripartita)-grassland vegetation at the USSES, parsnipflower buckwheat and cushion buckwheat (Eriogonum ovalifolium) decreased more than 85% when sheep grazing was changed from heavy fall use (1924–1949) to heavy spring use (1950–1964) (Laycock 1967).
Small mammals. Various small mammals feed on the foliage and seeds of parsnipflower buckwheat.
At the USSES, parsnipflower buckwheat made up to 2% of the mean density of fecal samples from pygmy rabbits (Brachylagus idahoensis) collected from September to October and March to April (Green and Flinders 1980). Foliage and seeds were gathered by least chipmunks (Neotamias minimus) and white-footed mice (Peromyscus leucopus) in the Rocky Mountains (Craighead et al. 1963). Shoots were found in 2 of 15 stomachs and made up an average of 12.5% of the composition of northern pocket gopher (Thomomys talpoides) stomachs collected in early June from Lawrence Memorial Grassland Preserve, north-central Oregon (Cox 1989).
Parsnipflower buckwheat is a valuable food for the threatened endemic northern Idaho ground squirrel (Urocitellus brunneus). It was abundant in five xeric meadows in Adams County, Idaho, where northern Idaho ground squirrel diets were evaluated. Although diets were quiet diverse, five fecal samples contained up to 72% parsnipflower buckwheat flowers. From all five sites and 151 fecal samples evaluated from April 23 to July 25 for 3 years, parsnipflower buckwheat foliage made up 0.3% of the diet composition and flowers made up 5.6% of diets. Flowers became abundant in northern Idaho ground squirrel diets late in the study period (July 1–25) (Yensen et al. 2018).
Birds. Buckwheat seeds provide food for a variety of game birds and song birds (Martin et al. 1951). Parsnipflower buckwheat attracts a variety and abundance of insects, which are important food sources for birds, especially greater sage–grouse (Centrocercus urophasianus) (Ogle et al. 2012; Luna et al. 2018). In big sagebrush communities in the Okanagan and Similkameen valleys of British Columbia, Brewer’s sparrow (Spizella breweri) presence was positively associated with parsnipflower buckwheat, and it was more abundant where abundance of parsnipflower buckwheat was greater (P < 0.05). These relationships were determined from a broad distribution of occupied and unoccupied areas. Sites used by Brewer’s sparrows had three times the abundance and two times the species richness of arthropods (Krannitz and Paczek 2005).
Insects. Parsnipflower buckwheat is important to a variety of pollinators, beetles, harvester ants, and other beneficial insects. Its flowers are a good late–season nectar source for bees (Ogle et al. 2012). It serves as a larval host for the following butterflies: blue copper (Lycaena heteronea), western green hairstreak (Callophrys affinis), Sheridan’s hairstreak (C. sheridanii), Cascadia blue (Euphilotes, a potentially undescribed species), and lupine blue (Plebejus lupini) (James and Nunnallee 2011).
Long–horned beetles (Cortodera subpilosa) were collected from parsnipflower buckwheat flowers at Craters of the Moon National Monument, Butte County, Idaho (Rice et al. 2017). In open ponderosa pine forests in Larimer County, Colorado, 55 adult beetles (Listrus senilis) were collected from parsnipflower buckwheat flowers. The beetles consumed both nectar and pollen (Mawdsley 1999) and pollinated flowers during their normal course of feeding (Mawdsley 2003).
In central Washington, more than 50 beneficial predators, parasitoids, and pollinators were collected in traps on parsnipflower buckwheat plants. Predators and parasitoids attracted to native buckwheats could prove useful in the biological control of wine grape pests. Parsnipflower buckwheat and other buckwheats could be cultivated within or around crops to attract natural enemies in organic or low-pesticide cropping (James et al. 2014).
In a field experiment conducted in high-elevation, shrub-steppe vegetation located near Kemmerer, Wyoming, parsnipflower buckwheat was the second most preferred seed of those provided to harvester ants (Pogonomyrmex occidentalis) (Broome 1988). In an observational study in the same area, parsnipflower buckwheat made up 2.7% of the relative abundance of seeds brought back to nests. Seed size averaged 2.7 mg and energy averaged 53 J/seed. In preference experiments, more parsnipflower buckwheat was selected from groups of seeds placed 33 ft (10 m) from nests than from those placed 16 ft (5 m) from nests. Ants selected larger seeds with higher energy content (like parsnipflower buckwheat) from the locations farther from the nest (Crist and MacMahon 1992).
Nutritional Value
In south–central British Columbia, parsnipflower buckwheat is important in bighorn sheep diets. Its nutritional content was compared for collections made from late August to mid-March (Table 5; Demarchi 1968). Of the eight commonly used forages evaluated, crude protein content of parsnipflower buckwheat decreased less from August to March and rated higher than all other species for nitrogen-free extract. Calcium content of parsnipflower buckwheat exceeded the minimum recommended by the National Research Council for growth and maintenance of cattle (0.24–0.34%) but phosphorus content was lower than that recommended for gestating and lactating domestic sheep and cattle (0.16–0.20%) (Demarchi 1968).
Table 5. Nutritional content of parsnipflower buckwheat collected periodically from summer through winter in south-central British Columbia (Demarchi 1968).
|
Sampling month |
Crude fat |
Crude protein |
Crude fiber |
Ash |
Nitrogen-free extract (NFE) |
Ca |
P |
|
Percent composition based on oven-dried forage |
|||||||
|
Aug |
2.54 |
6.64 |
21.29 |
4.75 |
64.78 |
0.72 |
0.08 |
|
Nov |
4.16 |
5.71 |
20.18 |
5.44 |
64.51 |
0.86 |
0.05 |
|
Mar |
2.57 |
6.01 |
34.16 |
3.28 |
53.98 |
0.40 |
0.04 |
Ethnobotany
Parsnipflower buckwheat was used medicinally and ceremonially by western Indian tribes. Thompson Indians of British Columbia toasted stalks and leaves until brittle, then crushed them into a powder that was mixed with grease and rubbed on the skin to reduce swellings (Steedman 1928). Mild to strong decoctions of the whole plant, including the roots, were drunk to relieve stiff, sore muscles and bones and to treat internal pains, rheumatism, and syphilis by Thompson Indians. It was also used to purify the body during ceremonial sweats (Steedman 1928). The Okanagan-Colville boiled roots and stems in water and ingested the liquid to treat colds or applied it to clean infected cuts. They also used a poultice of mashed leaves on cuts and sores (Turner et al. 1980). The Salishan people of northeastern Washington boiled roots in water and drank the liquid to relieve diarrhea. They mashed leaves and immersed them in water to encourage rains to come (Ray 1933).
Horticulture
Parsnipflower buckwheat is an attractive, drought tolerant, low-maintenance species that is available from specialty nurseries (Tilley and Fund 2017; UW Ext. 2018). Plants transplant well, grow in poor and rocky soils, and provide a good ground cover on harsh, dry sites (UW Ext. 2018). Parsnipflower buckwheat plants are semi–evergreen, have attractive brown to red fall color, persistent dry flowers, and are ideal for xeriscaping (Sutton and Johnson 1974; Mee et al. 2003; Parkinson 2003; Ogle et al. 2012).
Revegetation Use
Tolerance of early seral conditions, natural spread, and importance to pollinators are just a few reasons parsnipflower buckwheat is useful in revegetation (Monsen et al. 2004; Ogle et al. 2012). Buckwheat species are often the pioneers of disturbed sites. Their early establishment and spread on disturbed sites may help facilitate recruitment of later seral species (Monsen et al. 2004; Meyer 2008). Parsnipflower buckwheat can be used to diversify the seed mixes used to revegetate mine sites, road cuts, and weed-dominated burned areas within its range (Monsen et al. 2004; Meyer 2008; Tilley and Fund 2017).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for parsnipflower buckwheat, generalized provisional seed zones developed by Bower et al. (2014) may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat:moisture index). In Figure 8, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Seedlot Selection Tool (Howe et al. 2017) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
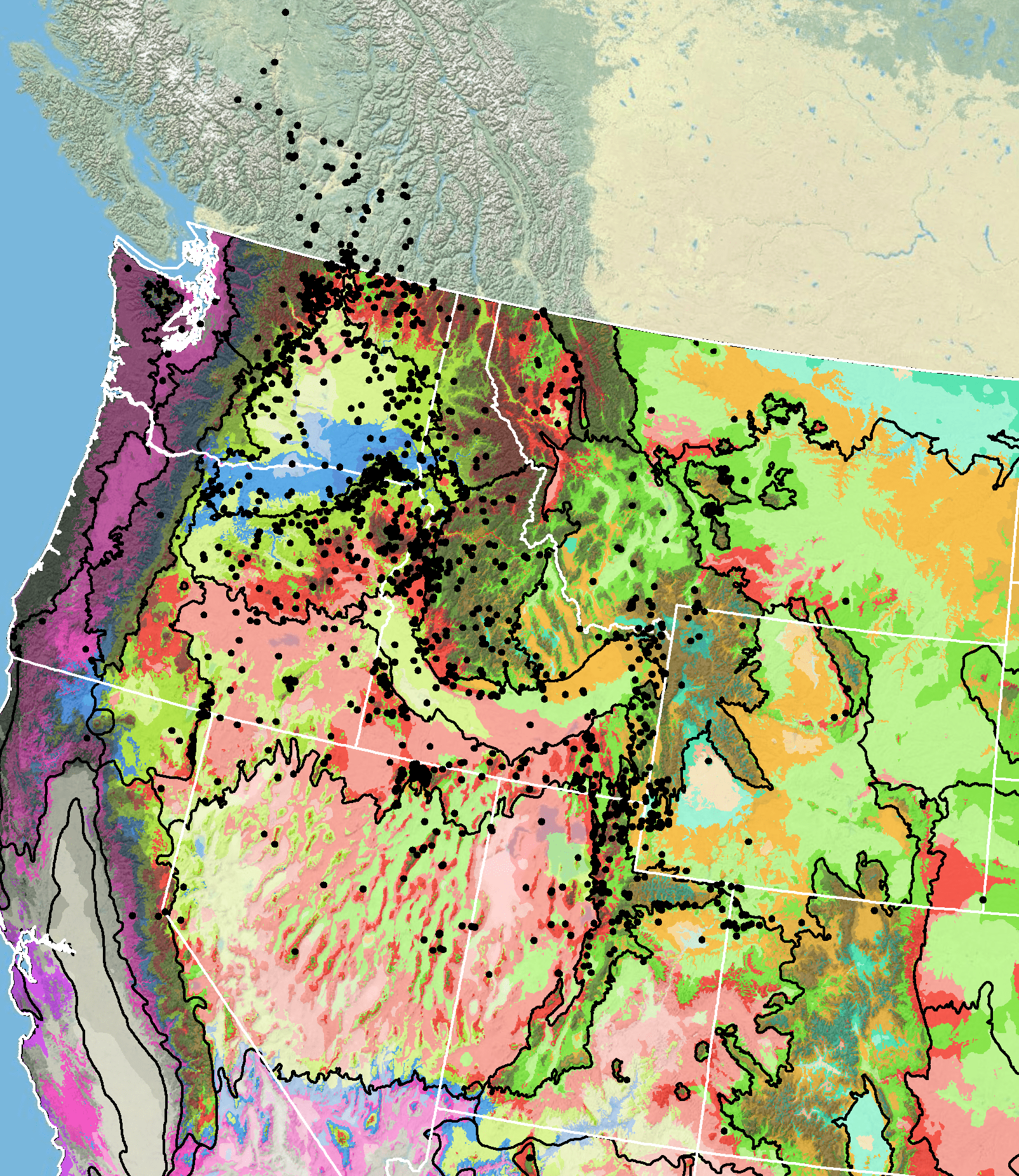
Figure 8. Distribution of parsnipflower buckwheat (black circles) based on geo-referenced herbarium specimens and observational data from 1880-2016 (CPNWH 2017; SEINet 2017; USDI USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USDI USGS.
Releases
Soda Springs Germplasm (E. heracleoides var. heracleoides) was released in 2017 by the Natural Resources Conservation Service’s (NRCS) Aberdeen, Idaho, Plant Materials Center (PMC) (Tilley and Fund 2017). It was one of several parsnipflower buckwheat accessions grown by the Aberdeen PMC and showed better establishment, seed production, and longevity than the other accessions. Soda Springs Germplasm was grown from seed collected from plants growing in mountain big sagebrush vegetation in Caribou County, Idaho, where annual precipitation is 18 to 20 in (457–508 mm) (St. John and Tilley 2014). It is suited for planting in the Snake River Plains (B11) and the eastern Idaho Plateaus (B13) Major Land Resource Areas (MLRA) and ecologically similar locations across the Intermountain West (Tilley and Fund 2017).
Wildland Seed Collection
Parsnipflower buckwheat seed is generally ripe in the summer (USDI BLM SOS 2017) and is ready for harvest when flowers are dry and rust colored (Fig. 9) (Camp and Sanderson 2007). Holmgren (1954) reports that ripe parsnipflower buckwheat seed remains on the plant for a considerable amount of time. Stevens et al. (1996) suggests seed is retained for up to 3 weeks once mature. Good seed crops are reported in most years and even in some dry years (Monsen et al. 2004), but a portion of seeds will likely have insect damage (Tilley and Fund 2017). Camp and Sanderson (2007) report that seed yield is often low and advise collecting large amounts of seed when possible. See cleanout ratios in Table 7.

Figure 9. Parsnipflower buckwheat seed heads on a plant growing in Utah. Photo: USDI BLM SOS UT931.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (Young et al. 2020; UCIA 2015). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source–Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Parsnipflower buckwheat seed is commonly ready to harvest in the summer (Fig. 10), and seed at low elevations generally matures before seed at high elevations. Ripe seed is typically available when flowers are dry, rust–brown, and easily released when plants are shaken (Camp and Sanderson 2007). Seed collection crews for the BLM SOS project have records for seed harvests made over 11 years in Washington, Oregon, Idaho, Nevada, and Utah (USDI BLM SOS 2017). Of the 47 collections, 14 were made in July and 18 in August. The earliest collection was made on June 24, 2013 in Malheur County, Oregon, at 4,600 ft (1,400 m). The latest collection was made on October 6, 2010 in Davis County, Utah, at 9,005 ft (2,745 m). In the single year with the greatest number of collections (14 harvests in 2010), the earliest occurred in early July in Douglas County, Washington, at 1,830 ft (560 m) and the latest in early October in Davis County, Utah, at 9,005 ft (2,745 m) (USDI BLM SOS 2017). In southern British Columbia, parsnipflower buckwheat produced most flowers in June, but in a year when October precipitation was 6 mm more than normal, some plants flowered a second time (Pitt and Wikeem 1990).

Figure 10. Wildland collected parsnipflower buckwheat seed. Photo: N. Shaw, USFS RMRS.
Collection Methods
Wildland seed can be collected by shaking, hand stripping, or beating flowering stems over a container (Holmgren 1954; Meyer and Paulsen 2000; Jorgensen and Stevens 2004; Camp and Sanderson 2007).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of parsnipflower buckwheat.
Post-Collection Management
Insect larvae are common in parsnipflower buckwheat seed. Freezing seeds (0–10 °F [-18 to -12 °C] for 7–10 d) before long–term storage is an effective method for killing insects (Tilley and Fund 2017).
Seed Cleaning
Wild-collected parsnipflower buckwheat seed is easy to clean. Seed collections commonly include perianths, involucres, and inflorescence branches. Seed is first dried, then processed through a barley debearder and then through an air-screen separator (Shaw 1984; Jorgensen and Stevens 2004; Monsen et al. 2004). Germination is possible with the perianth still attached (Meyer 2008). Camp and Sanderson (2007) caution that any cleaning procedure needs to be gentle enough to not damage the radicle.
There are additional detailed cleaning procedures for wildland seed collections. At the Pullman PMC, small seed lots were first rubbed, then processed through an air column separator. Larger seed lots were threshed using a hammermill, then cleaned using air-screen equipment (Skinner 2008). Camp and Sanderson (2007) dried parsnipflower buckwheat at room temperature for 2 to 4 weeks before cleaning. Seed was then processed through a standard testing sieve #6 to remove large debris, then rubbed on screens, and finally processed through a fanning mill (Camp and Sanderson 2007). A small collection of hand-stripped seeds cleaned for use in a germination study was first hand rubbed through a 2-mm round-hole screen. This released seeds from the perianths. A tabletop-model blower was then used to separate seeds from the floral debris (Meyer and Paulsen 2000).


Figure 11. Clean and enlarged parsnipflower buckwheat seed and a representation of seed size (mm). Photo (top): USDA USFS Bend Seed Extractory. Photo (bottom): USDI BLM OR030 SOS.
Several procedures were reported for large collections of field–grown seed. The general procedure was to dry the seed, then screen, chop, or rub the seed to remove it from the perianth, and then re-screen. Seed was then processed using a gravity table, air screen, or air blower to remove unfilled seed and debris (Stevens et al. 1996). Seed grown at the Oregon State University Malheur Experiment Station in Ontario, Oregon, was collected using a small-plot combine. Unthreshed seed was manually processed through a meat grinder and then through a small clipper (Shock et al. 2017).
The Bend Seed Extractory cleaned 18 lbs (8 kg) of seed grown at Lucky Peak Nursery near Boise, Idaho, using the following procedure: 1) processed using Westrup Model LA-H laboratory brush machine (Hoffman Manufacturing, Corvallis, OR) with #40 mantel, speed of 3 to remove seed from flower heads; 2) finished by air-screening to remove non-viable seed and inert material using an office clipper with top screen: 7 round and bottom screen 1/22 round, medium speed and medium to high air. This cleaning process resulted in 158,000 seeds/lb (348,000 seeds/kg) at 97% purity, 86% fill, 96% viability, and 7.2% moisture content (Barner 2009).
Seed Storage
Seed should be stored in a dry location protected from rodents (Stevens et al. 1996). Insect larvae are common in parsnipflower buckwheat seed, and seed lots should be frozen or treated to kill any insects before long-term storage (Stevens et al. 1996; Tilley and Fund 2017). Longevity of stored seed ranges from 4 to 15 years ( Stevens et al. 1996; Jorgensen and Stevens 2004; RBG Kew 2017). Storage conditions ranged from -4 to 40 °F (-20 to 4 °C), and relative humidity of 15 to 40% (Skinner 2008; Barner 2009). Tests conducted by the Royal Botanical Gardens Kew showed that parsnipflower buckwheat seed is orthodox, and 100% viability was retained after seed was dried to 15% relative humidity and kept for 7.2 years at -4 °F (-20 °C) (RBG Kew 2017).
Parsnipflower buckwheat seed retained consistent germination for up to 7 years of storage in an open warehouse in Sanpete County, Utah. Seed was cleaned to at least 85% purity and stored in cotton bags in a metal cabinet in a warehouse where the low temperature reached -22 °F (-30 °C) and the high, 100 °F (38 °C). After 2 years of storage, 51% of seeds germinated. After 3 years, germination was 87%, which was significantly greater than that of 2-year-old seed (P < 0.05). Germination percentage decreased significantly from 5 years (90%) to 7 years (64%) of storage and from 7 years to 10 years (16%) of storage (P < 0.05). After 15 years, just 5% of seed germinated, and no seed germinated after 25 years of storage (Stevens and Jorgensen 1994).
Seed Testing
The Association of Official Seed Analysts (AOSA) developed a tetrazolium chloride (TZ) viability test for buckwheat species (AOSA 2010). Seed is kept moist overnight at 68 to 77 °F (20–25 °C). Imbibed seeds are cut laterally and the distal end of the cotyledons is removed. Seeds are soaked overnight in a 1% TZ solution at 86 to 95 °F (30–35 °C). Seeds are non–viable if any portion of the embryo does not stain (AOSA 2010).
Germination Biology
Seed germination is improved with an afterrippening period of at least 30 days (Stevens and Jorgensen 1994; Stevens et al. 1996). Cold temperature exposure is the primary regulator of seed dormancy, thus the rate and total germination percentage can be increased with cold-moist stratification even though seed will germinate without pretreatment (Meyer and Paulsen 2000; Skinner 2008). The duration of cold exposure necessary to achieve maximum germination varies with the conditions of the seed collection site. Longer cold temperature exposure is required for seed collected from high-elevation habitats with long winters than for collections from warm, low-elevation habitats (Meyer and Paulsen 2000).
Cold-stratification increased the emergence rate and total emergence of wildland seed collected at Paradise Creek near Pullman, Washington. In germination trials conducted at the Pullman PMC, emergence in the greenhouse was 20% without stratification, 45% with 45 days of cold-moist stratification, and 72% with 90 days of cold-moist stratification. Seed sown in containers outdoors reached 95% emergence, which was likely due to a longer stratification period. Stratified seed also emerged more rapidly than untreated seed (Skinner 2008).
Germination was low (<20%) without chilling, but 16 or more weeks of chilling removed dormancy in seeds collected from parsnipflower buckwheat populations in Utah (Table 6; Meyer and Paulsen 2000). Seed was collected from at least 20 plants at each of three Utah locations. Seed was stored in paper envelopes in a laboratory (68–72 °F [20–22 °C] and 30–40% relative humidity) for 1 to 2 months before testing. Low percentages of germination occurred without chilling (≤ 15%), and long chilling periods were needed for complete dormancy removal (≥ 16 wks). Germination was slow, taking 17 to 19 weeks to reach 50% germination at 36 °F [2 °C]). Across all seed collected for the Eriogonum genus, collections from high-elevation habitats with long winters required longer chilling periods to germinate than collections from warm, low-elevation habitats (Meyer and Paulsen 2000).
Experiments suggest that smoke exposure does not encourage parsnipflower buckwheat germination. When seeds were soaked in distilled water or smoke water concentrations of 1:10, 1:100, and 1:1000, germination was significantly lower for seeds treated to high smoke exposure (1:10) than the controls and lesser smoke concentrations. Germination was not different between 1:100, 1:1000, and distilled water treatments (Cox 2016).
Wildland Seed Yield And Quality
Post–cleaning seed yield and quality of seed lots collected in the Intermountain region are provided in Table 7 (USFS BSE 2017). The results indicate that parsnipflower buckwheat seed can generally be cleaned to high levels of purity but fill and viability of fresh seed are highly variable. Other sources reported a range of 119,974 to 298,421 seeds/lb (264,493–657,895 seeds/kg) for wildland-collected seed, close to the range reported in Table 7 (Stevens et al. 1996; Jorgensen and Stevens 2004; Skinner 2008; Ogle et al. 2011; USFS GBNPP 2014; RBG Kew 2017). For farm-grown seed, Tilley and Fund (2017) reported 374,000 seeds/lb (825,000 seeds/kg).
Table 6. Variability in seed size, germination, and viability for parsnipflower buckwheat growing at increasing elevations in Utah (Meyer and Paulsen 2000).
|
Collection |
Mass of 100 achenes (g) |
Mean viability (%) |
Chilling (36 °F) Duration (wks) |
|||||
|
0 |
4 |
8 |
12 |
16 |
24 |
|||
|
Mean germination (%, as percentage of total viable seeds) |
||||||||
|
Utah County 7,500 ft |
2.9 |
85 |
1 |
11 |
43 |
69 |
99 |
100 |
|
Summit County 7,550 ft |
3.0 |
85 |
3 |
5 |
19 |
52 |
80 |
99 |
|
Wasatch County 8,300 ft |
2.8 |
69 |
15 |
24 |
55 |
68 |
98 |
97 |
Table 7. Seed yield and quality of parsnipflower buckwheat seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Seed lot characteristic |
Mean |
Range |
Samples (no.) |
|
Bulk weight (lbs) |
9.7 |
0.08–193 |
63 |
|
Clean weight (lbs) |
3.0 |
0.02–55 |
63 |
|
Clean–out ratio |
0.2 |
0.03–0.9 |
63 |
|
Purity (%) |
96 |
85–99 |
63 |
|
Fill (%)¹ |
90 |
30–99 |
63 |
|
Viability (%)² |
78 |
30–97 |
53 |
|
Seeds/lb |
174,379 |
77,000–265,000 |
63 |
|
Pure live seeds/lb |
130,691 |
42,336–196,419 |
53 |
¹100 seed X–ray test
²Tetrazolium chloride test
Marketing Standards
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed quite well. The following standards are suggested for marketed seed: 95 to 98% purity, >90% viability, and 75% germination (Jorgensen and Stevens et al. 1996; Stevens 2004).
Agricultural Seed Production
Seed production plots were evaluated by researchers at Oregon State University’s Malheur Experiment Station (OSU MES) and the Aberdeen PMC (Fig. 12, Shock et al. 2017; Tilley and Fund 2017). Crops were harvestable by the second growing season, and seed yield averaged about 200 to 300 lbs PLS/ac (220–340 kg/ha) at OSU MES, where stands were harvested for 6 years (Shock et al. 2018).
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Site Preparation
Seedbeds should be weed free, moist, and firmly packed (Tilley and Fund 2017).
Weed Management
Mechanical and hand weeding is recommended until plants reach at least 2 years old and are considered competitive with weeds (Stevens et al. 1996). Weed barrier fabric is also an effective control method (Tilley and Fund 2017).

Figure 12. Parsnipflower buckwheat seed production plots growing at Oregon State University’s Malheur Experiment Station (OSU MES) in Ontario, OR. Photo: N. Shaw, USFS RMRS.
Seeding
Fall or early winter seeding of parsnipflower buckwheat is recommended (Stevens et al. 1996; Tilley and Fund 2017). Seed should be planted no more than 3 to 5 mm deep (Meyer 2008; Tilley and Fund 2017). Seeds can be hand planted or by using a single-row or drill seeder (Stevens et al. 1996; Tilley and Fund 2017).
Seeding rates vary with the seeding method. Stevens et al. (1996) suggest 15 to 20 PLS per linear ft of row (50–65 PLS/m), with rows spaced 30 to 36 in (76–91 cm) apart or 4 to 5 PLS in hills 2 to 3 ft (0.6–0.9 m) apart. Tilley and Fund (2017) recommended planting seeds 9 to 18 in (23–46 cm) apart in weed barrier fabric and using a rate of 20 to 30 PLS/linear ft (65–85 PLS/m) when drill seeding (Tilley and Fund 2017). If transplants are used, a 30- to 36-in (76–91 cm) spacing within and between rows is recommended (Stevens et al. 1996).
At OSU MES, parsnipflower buckwheat seed was planted in November on the soil surface at a rate of 20 to 30 seeds/ft of row (65–100 seeds/m). Seeds were planted in four rows spaced 30 in (76 cm) apart using a custom, small–plot, grain drill. Seeds were covered with a narrow band of sawdust. Plots were covered with row cover, which was removed when seedlings emerged the following April. Any gaps in the rows were reseeded by hand the following November (Shock et al. 2017).
Parsnipflower buckwheat was fall planted into weed barrier fabric at the Aberdeen PMC. Germination was poor because of drier and warmer than normal winter and spring conditions. The following summer, green-house grown transplants were used to establish seed increase plots (St. John and Tilley 2014).
Establishment And Growth
Fertilization of seed production stands can be done once every 2 years, and over fertilizing should be avoided (Stevens et al. 1996).
Irrigation
Typically, parsnipflower buckwheat grows well where annual precipitation and supplemental irrigation total 14 to 20 in (356–508 mm). The suggested timing of irrigation is from mid-spring to early summer if conditions are dry, but too much water can weaken stands (Stevens et al. 1996).
In seed production plots at OSU MES in Ontario, Oregon, parsnipflower buckwheat seed yield increased with irrigation in years with low spring precipitation. The study evaluated additions of 0, 4, and 8 in (0–200 mm) of irrigation for 6 years (2011–2016) after stands had reached 2 years old. Drip irrigation was delivered 12 in (30 cm) deep in four bi-weekly increments starting when flower buds were forming (Table 8). At this location annual precipitation averaged 10 in (254 mm). Over 6 years, the seed yield averaged 315 lbs/ac (353 kg/ha) and ranged from 72 to 525 lbs/ac (81–588 kg/ha) (Table 9). Seed yield responded significantly to irrigation only in 2013 and 2014, years with the lowest spring precipitation. Averaged over the 6-year study, seed yield of parsnipflower buckwheat was maximized by 5 in (126 mm)/year of irrigation (Table 9; Shock et al. 2017).
Table 8. Flowering timing as related to timing of irrigation of parsnipflower buckwheat seed production plots growing at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2017)
|
Year |
Flowering |
Irrigation |
||||
|
Start |
Peak |
End |
Start |
End |
Harvest |
|
|
2011 |
26 May |
10 June |
8 July |
27 May |
6 July |
1 Aug |
|
2012 |
23 May |
30 May |
25 June |
11 May |
21 June |
16 July |
|
2013 |
29 Apr |
13 May |
10 June |
24 Apr |
5 June |
1 July |
|
2014 |
20 May |
20 May |
12 June |
29 Apr |
10 June |
3 July |
|
2015 |
13 May |
–––––– |
17 June |
15 April |
27 May |
24 June |
|
2016 |
16 May |
–––––– |
16 June |
18 April |
31 May |
23 June |
Table 9. Maximum seed yield of parsnipflower buckwheat seed production plots by irrigation amount applied based on quadratic response. Plots were grown at Oregon State University’s Malheur Experiment Station in Ontario, OR. Seed was harvested using a small-plot combine in all years, except for 2013 and 2016 when seed was harvested by hand (Shock et al. 2017).
|
Year |
Highest yield (lbs/ac) |
Spring ppt. (in) |
Growing degree hrs (50–86 °F) Jan–June |
Additional water |
|
2011 |
71.6 |
4.8 |
476 |
3.6 |
|
2012 |
316.8 |
2.6 |
682 |
4.2 |
|
2013 |
524.7 |
0.8 |
733 |
4.8 |
|
2014 |
370.2 |
1.7 |
741 |
7.4 |
|
2015 |
150.2 |
3.2 |
895 |
4.8 |
|
2016 |
486.9 |
2.2 |
810 |
4.2 |
|
Average |
315.2 |
2.8* |
665** |
5.0 |
*72-year average; **23-year average
Pollinator Management
Insects are needed for pollination of parsnipflower buckwheat flowers and thus maximum seed yield (Stevens et al. 1996). See Insects section of Wildlife and Livestock Use for more information on pollinators.
Pest Management
Insects infesting flowers and developing seeds can result in 10 to 35% crop loss (Stevens et al. 1996). The following fungi use parsnipflower buckwheat as a host: Gleosporium eriogoni, Ramularia eriogoni, Sclerotinia spp., and Uromyces intricatus (Farr and Rossman 2017) and scale (Acanthococcus euphorbiae) uses parsnipflower buckwheat as a host (Miller and Miller 1992) but their impact on seed production fields was not evaluated.
Seed Harvesting
Parsnipflower buckwheat seed ripens over a wide window, making multiple hand harvests necessary to maximize yield (Tilley and Fund 2017). Mechanical harvests using modified combines can be used for less labor intensive harvesting (Shock et al. 2017; Tilley and Fund 2017). Seed is usually harvested between mid-July and mid-August, but it can be earlier at lower elevations and later at higher elevations. Seed often remains on the plant for 2 to 3 weeks after maturing (Stevens et al. 1996).
Seed Yields And Stand Life
Flowers and seeds are produced by parsnipflower buckwheat in the second year after planting when about 30 to 50% of the full crop is produced (Stevens et al. 1999; Shock et al. 2017). By the third or fourth year, seed yields average up to 300 lbs/ac (336 kg/ha) at 95% purity (Stevens et al. 1996). At OSU MES, seed yield ranged from 72 to 525 PLS lbs/ac (81–588 kg/ha), and stands were harvested for 6 years after plants had 2 years to establish. Stand life is at least 6 years according to research conducted at OSU MES (Shock et al. 2017).
Nursery Practice
Production of parsnipflower buckwheat container stock takes 4 to 6 months plus hardening time (Shaw 2004). Based on bareroot seedling production at the USFS Lucky Peak Nursery near Boise, Idaho, seed can be sown in fall without pretreatment or in spring with artificial stratification (Fig. 13). Roots can be pruned to improve plant uniformity. Plants are lifted after 1 year in the seedbed and then stored cold until wildland use (Shaw 1984).

Figure 13. Parsnipflower buckwheat bareroot nursery production plots growing at USFS Lucky Peak Nursery in Boise, ID. Photo: M. Fisk, USFS RMRS.
Camp and Sanderson (2007) used the following protocol to propagate parsnipflower buckwheat in Peshastin, Washington. Seed was sown in a soil mix of 45% peat, 30% vermiculite, and 35% sand in 3-in³ cones in early November. Cones were kept outside through March to allow for natural stratification. Germination began in late February and continued through mid-March when 85% of cones contained seedlings. Seedlings were transplanted to 10-in³ cones, which were fertilized and then irrigated as needed until outplanting in the fall (Camp and Sanderson 2007).
Skinner (2008) describes the following procedure used by the Pullman PMC to produce parsnipflower buckwheat plugs with well-developed roots. Seeds were sown in late October and early November in 10-in³ Ray Leach Super cell cone-tainers filled with Sunshine #4 soil mix (Sun Gro Horticulture, Agawam, MA). Seeds were covered lightly with soil and a thin layer of coarse grit to prevent floating when watered. Cone-tainers were watered deeply and kept outdoors until February when they were moved into the greenhouse. Alternately, seed can be moist stratified in a refrigerator at 35 to 40 °F (2–4 °C) for 90 to 120 days before sowing to skip the outdoor stratification step. Germination occurred over 4 to 12 days in the greenhouse. Seedlings were watered deeply every third day and fertilized once a week using a complete, water-soluble fertilizer containing micronutrients. Plants were moved to a cold frame in mid-April and watered every other day in cool weather and every day in hot, dry weather. The active growth period lasted 3 months, and the hardening phase, 2 to 3 weeks. It took 4 months to produce plugs with tight roots throughout the cone-tainer. A few plants flowered the year following outplanting, but most required 2 to 3 years to produce seed (Skinner 2008).
Wildland Seeding And Planting
Parsnipflower buckwheat has been established from seed and transplanted successfully in wildland settings. It grows in well-drained soils with slightly basic to neutral pH and is recommended for pollinator habitat at sites receiving 9 to 18 in (230–460 mm) of annual precipitation (Stevens et al. 1996; Ogle et al. 2011). Because parsnipflower buckwheat has low seedling vigor but plants once established are long–lived, transplants are often used in restoration (Ogle et al. 2012).
Methods. Fall or early winter seeding is recommended to allow for natural stratification. Seed can be drilled or broadcast but should be covered by less than 0.2 in (0.5 cm) of soil (Monsen et al. 2004; Camp and Sanderson 2007). The full seeding rate for parsnipflower buckwheat is 4 PLS lbs/ac (3.4 kg/ha), however, it is typically seeded as part of a seed mix at a rate of 0.25 to 0.5 PLS lb/ac (0.3–0.6 kg/ha) (Tilley and Fund 2017). Parsnipflower buckwheat generally grows well when seeded with shrubs, grasses, and other forbs, but its growth can be limited when seeded with aggressive perennial grasses (Monsen et al. 2004). Transplants should be spaced 4 ft (1.2 m) apart (Ogle et al. 2011). Because parsnipflower buckwheat takes 2 years to produce seed and 3 years to produce full seed crops, it should be protected from grazing for at least 2 years and even longer on dry sites (Stevens 2004).
Field examples. After a study comparing the establishment and growth of parsnipflower buckwheat on weedy and weed-free sites along the South Fork of the Payette River in Boise County, Idaho, Holmgren (1954) rated the species high for: dependability of seed crop, ease of seed collection, ease of seed handling and storage, seedling survival, and growth rate. Based on initial field experiments with parsnipflower buckwheat in Ephraim Canyon, Utah, Plummer et al. (1968) rated the species very good for ease of planting and persistence. Germination, final establishment, natural spread, soil stability, grazing tolerance, disease and insect resistance, compatibility with other plants, and summer palatability were rated good. Initial establishment, ease of transplanting, growth rate, and herbage yield were rated fair to poor (Plummer et al. 1968).
Parsnipflower buckwheat was established successfully as part of a revegetation project in California’s Tahoe Basin. Roadside revegetation sites had 50 to 100 parsnipflower buckwheat plants growing in full sun and partial shade. No other details about the project were reported, except that the restoration area was likely south of the known natural range for the species (Hoyer 2013).
Field studies evaluated parsnipflower buckwheat establishment at three sites (Spanish Fork, UT; Clarkston, UT, and Virginia, ID), where annual precipitation averaged 13 to 19 in (330–480 mm). Studies used snow fences that increased soil water availability, N-sulate fabric treatments that protected seeds from freezing and rapidly drying, and those treatments together with seed pretreatments to evaluate them as ways to improve seedling establishment. For all 10 native forb species evaluated, germination was not significantly different between snow fences, N-sulate fabric, or controls (P–value not reported). Parsnipflower buckwheat had high germination (30–40%) and 40 to 50% of seedlings survived the first growing season. Germination and seedling emergence were highest for seeds coated with a broad-spectrum fungicide before planting (Johnson et al. 2017; Tilley and Fund 2017).
Parsnipflower buckwheat showed better competitive ability with broadleaf weeds than with cheatgrass (Bromus tectorum) when seedings were compared on deer winter range along the South Fork of the Payette River in Boise County, Idaho (Table 10; Holmgren 1954). The site had coarse, loose, granite soils with low moisture retaining capacity. In November 1949, seed of parsnipflower buckwheat and 26 other browse species were hand planted into furrows in a weed-free site (all weeds were controlled before seeding and each subsequent spring), broadleaf-weed site (cheatgrass removed but replaced by a stand of annual broadleaf weeds), and cheatgrass site (relatively undisturbed and cheatgrass dominated). Parsnipflower buckwheat seed was not treated prior to planting and germination was good. No insect or pathogen damage was noted. Seedling mortality was primarily due to insufficient moisture, and the wilting and dying of otherwise undamaged plants (Holmgren 1954).
Table 10. Survival and height of parsnipflower buckwheat seeded into sites with various types of weed pressure in Boise County, ID (Holmgren 1954).
|
Conditions |
June 1950 |
Sept 1950 |
May 1951 |
May 1952 |
May 1951 |
|
––––––––––––––Survival %–––––––––––– |
Average height (in) |
||||
|
Weed free |
55 |
32 |
24 |
24 |
3.75 |
|
Broadleaf weeds |
49 |
34 |
30 |
30 |
2.25 |
|
Cheatgrass |
31 |
5 |
4 |
3 |
1.5 |
Survival and growth of seedlings and transplants were monitored by the Bureau of Land Management in Wenatchee, Washington. Seeding and planting were done for 2 consecutive years (2004 and 2005). In direct-seeded plots, 10 to 20 seeds were planted per plot. For plots seeded in fall 2004, 85% had at least one seedling in spring 2005 and 45% had at least one seedling in spring 2006. For plots seeded in fall 2005, 100% had at least one seedling in spring 2006. For transplants, survival was compared with and without irrigation. For 2004 transplants, survival to spring 2005 was 88% for irrigated and 90% for non-irrigated and survival to spring 2006 was 70% for irrigated and 40% for non-irrigated. For 2005 transplants, survival to spring 2006 was 98% for irrigated and 96% for non-irrigated (Camp and Sanderson 2007).
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Robert L. Johnson, Brigham Young University, and Janel D. Johnson, Nevada Natural Heritage Program.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Anderson, N.A. 1940. Mountain goat study: Progress report. Bio. Bull. No. 2. Seattle, WA: State of Washington Department of Game. 21 p.
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Barner, J. 2009. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Eriogonum heracleoides Nutt. seeds. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2019 July 31].
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Blaisdell, J.P. 1953. Ecological effects of planned burning of sagebrush-grass range on the upper Snake River Plains. Tech. Bull. 1975. Washington, DC: U.S. Department of Agriculture. 39 p.
Blood, D.A. 1967. Food habits of the Ashnola bighorn sheep herd. The Canadian-Field Naturalist. 81: 23-29.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Broome, L.S. 1988. Determinant of small rodent distribution and abundance in a shrub-steppe ecosystem: Influences of seeds, ants, and shrubs. Logan, UT: Utah State University. Thesis. 152 p.
Brotherson, J.D. 1999. Measured and inferred moisture gradient relationships across ecotone boundaries in shrub-dominated foothill communities. In: McArthur, E.D.; Ostler, W.K.; Wambolt, C.L., comps. Proceedings: Shrubland ecotones; 1998 August 12-14; Ephraim, UT. Proc. RMRS-P-11. Ogden, UT: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 40-48.
Bunting, S.C.; Kingery, J.L.; Strand, E. 1999. Effects of succession on species richness of the western juniper woodland/sagebrush steppe mosaic. In: Monsen, S.B.; Stevens, R., comps. Proceedings: Ecology and management of pinyon-juniper communities within the Interior West: Sustaining and restoring a diverse ecosystem. 1997 September 15-18; Provo, UT. Proc. RMRS-P-9. Ogden, UT: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 76-81.
Burkhardt, J.W.; Tisdale, E.W. 1969. Nature and successional status of western juniper vegetation in Idaho. Journal of Range Management. 22(4): 264-270.
Camp, P.; Sanderson, J. 2007. Seed collection, propagation and reintroduction of native wildflowers in the Columbia Basin. Wenatchee, WA: U.S. Department of the Interior, Bureau of Land Management, Wenatchee Field Office. 32 p.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Cook, C.W. 1983. “Forbs” need proper ecological recognition. Rangelands. 5(5): 217-220.
Cox, G.W. 1989. Early summer diet and food preferences of northern pocket gophers in north central Oregon. Northwest Science. 63(3): 77-82.
Cox, R. 2016. Smoke-induced germination of Great Basin native forbs. In: Kilkenny, F; Edwards, F.; Malcomb, A., eds. Great Basin Native Plant Project: 2015 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 78-82.
Craighead, J.J.; Craighead, F.C. Jr.; Davis, R.J. 1963. A field guide to Rocky Mountain wildflowers from northern Arizona and New Mexico to British Columbia. Boston, MA: Houghton Mifflin Company. 277 p.
Crist, T.O.; MacMahon, J.A. 1992. Harvester ant foraging and shrub-steppe seeds: Interactions of seed resources and seed use. Ecology. 73(5): 1768-1779.
Daubenmire, R. 1970. Steppe vegetation of Washington. Washington Agricultural Experiment Station Tech. Bull. 62. Pullman, WA: Washington State University. 131 p.
Daubenmire, R. 1972. Annual cycles of soil moisture and temperature as related to grass development in the steppe of eastern Washington. Ecology. 53(3): 419-424.
Day, T.; Wright, R.G. 1985. The vegetation types of Craters of the Moon National Monument. Bull. 38. Moscow, ID: University of Idaho, College of Forestry, Wildlife, and Range Sciences, Forest, Wildlife and Range Experiment Station. 6 p.
Dayton, W.A. 1960. Notes on western range forbs: Equisetaeae through Fumariaceae. Washington, DC: U.S. Department of Agriculture, Forest Service. 254 p.
Demarchi, R.A. 1968. Chemical composition of bighorn winter forages. Journal of Range Management. 21(6): 385-388.
Eddleman, L.E.; Miller, P.M.; Miller, R.F.; Dysart, P.L. 1994. Western juniper woodlands (of the Pacific Northwest): Science assessment. Walla Walla, WA: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station, Interior Columbia Basin Ecosystem Management Project. 132 p.
Eggler, W.A. 1941. Primary succession on volcanic deposits in southern Idaho. Ecological Monographs. 11(3): 277-298.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Farr, D.F.; Rossman, A.Y. 2017. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://nt.ars-grin.gov/fungaldatabases/.
Franklin, J.F.; Dyrness, C.T. 1973. Natural vegetation of Oregon and Washington. Gen. Tech. Rep. PNW-8. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station. 452 p.
Goodrich, S. 2008. Trout Creek 1999 burn. In: Kitchen, S.G.; Pendleton, R.L.; Monaco, T.A.; Vernon, J., comps. Proceedings – Shrublands under fire: Disturbance and recovery in a changing world. 2006 June 6-8; Cedar City, UT. Proc. RMRS-P-52. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 147-150.
Green, J.S.; Flinders, J.T. 1980. Habitat and dietary relationships of the pygmy rabbit. Journal of Range Management. 33(2): 136-142.
Hall, F.C. 1973. Plant communities of the Blue Mountains in eastern Oregon and southeastern Washington. R6 Area Guide 3-1. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Region. 62 p.
Hessberg, P.F.; Smith, B.G.; Kreiter, S.D.; Miller, C.A.; Salter, R.B.; McNicoll, C.H.; Hann, W.J. 1999. Historical and current forest and range landscapes in the interior Columbia River Basin and portions of the Klamath and Great Basins. Part 1: Linking vegetation patterns and landscape vulnerability to potential insect and pathogen disturbances. Gen. Tech, Rep. PNW-GTR-458. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station; Department of the Interior, Bureau of Land Management. 357 p.
Hitchcock, C.L.; Cronquist, A. 2018. Flora of the Pacific Northwest: An illustrated manual. Second Ed. Giblin, D.E.; Legler, B.S.; Zika, P.F.; Olmstead, R.G., eds. Seattle, WA: University of Washington Press. 882 p.
Holechek, J.L.; Vavra, M.; Skovlin, J.; Krueger, W.C. 1982. Cattle diets in the Blue Mountains of Oregon, I. Grasslands. Journal of Range Management. 35(1): 109-112.
Holmgren, R.C. 1954. A comparison of browse species for revegetation of big-game winter ranges in southwestern Idaho. Res. Pap. No. 3. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station. 12 p.
Howe, G.; St, Clair, B.; Bachelet, D. 2017. Seedlot Selection Tool. Corvallis, OR: Conservation Biology Institute. https://seedlotselectiontool.org/sst/
Hoyer, W.F. 2013. Noteworthy collection. Madrono. 60(3): 261.
James, D.G.; Nunnallee, D. 2011. Life histories of Cascadia butterflies. Corvallis, OR: Oregon State University Press. 447 p.
James, D.G.; Seymour, L.; Lauby, G.; Buckley, K. 2014. Beneficial insects attracted to native flowering buckwheats (Eriogonum Michx) in central Washington. Environmental Entomology. 43(4): 942-948.
Johnson, C.G. 1998. Vegetation response after wildfires in national forests of northeastern Oregon. R6-NR-ECOL-TP-06-98. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Region. 128 p.
Johnson, C.G.; Simon, S.A. 1987. Plant associations of the Wallowa-Snake Province: Wallowa-Whitman National Forest. R6-ECOL-TP-255A-86. Baker, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Region, Wallowa-Whitman National Forest. 399 p.
Johnson, C.G.; Swanson, D.K. 2005. Bunchgrass plant communities of the Blue and Ochoco Mountains: A guide for managers. R6-NR-ECOL-TP-06-98. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. 128 p.
Johnson, D.A.; Monaco, T.A.; Hulvey, K. 2017. Forb islands: Possible techniques to improve forb seedling establishment for diversifying sagebrush-steppe communities. In: Kilkenny, F.; Edwards, F.; Irwin, J.; Barga, S. Great Basin Native Plant Project: 2016 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 167-173.
Jorgensen, K.R.; Stevens, R. 2004. Seed collection, cleaning, and storage. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 3. Gen. Tech. Rep. RMRS-GTR-136-vol-3. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 699-716.
Korfhage, R.C.; Nelson, J.R.; Skovlin, J.M. 1980. Summer diets of Rocky Mountain elk in northeastern Oregon. The Journal of Wildlife Management. 44(3): 746-750.
Krannitz, P.; Paczek, S. 2005. The importance of floristics to sagebrush breeding birds of the south Okanagan and Similkameen valleys, British Columbia. In: Ralph, C.J.; Rich, T.D., eds. 2005. Bird conservation implementation and integration in the Americas: Proceedings of the Third International Partners in Flight Conference. 2002 March 20-24; Asilomar, CA, Vol. 1 Gen. Tech. Rep. PSW-GTR-191. Albany, CA: U.S. Dept. of Agriculture, Forest Service, Pacific Southwest Research Station: 621-629.
Laubengayer, R.A. 1937. Studies in the anatomy and morphology of the Polygonaceous flower. American Journal of Botany. 24(6): 329-343.
Laycock, W.A. 1967. How heavy grazing and protection affect sagebrush-grass ranges. Journal of Range Management. 20(4): 206-213.
Luna, T.; Mousseaux, M.R.; Dumroese, R.K. 2018. Common native forbs of the northern Great Basin important for greater sage-grouse. Gen. Tech. Rep. RMRS-GTR-387. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station; Portland, OR: U.S. Department of the Interior, Bureau of Land Management, Oregon-Washington Region. 76 p.
Major, J.; Rejmanek, M. 1992. Amelanchier alnifolia vegetation in eastern Idaho, USA and its environmental relationships. Vegetatio. 98(2): 141-156.
Martin, A.C.; Zim, H.S.; Nelson, A.L. 1951. American wildlife and plants: A guide to wildlife food habits. New York, NY: Dover Publications. 500 p.
Mawdsley, J.R. 1999. Redescription and notes on the biology of Amecocerus senilis (Leconte) (Coleoptera: Melyridae: Dasytinae). Journal of the New York Entomological Society. 107(1): 68-72.
Mawdsley, J.R. 2003. The importance of species of Dasytinae (Coleoptera: Melyridae) as pollinators in western North America. The Coleopterists Bulletin. 57(2): 154-160.
McColley, P.D.; Hodgkinson, H.S. 1970. Effect of soil depth on plant production. Journal of Range Management. 23(3): 189-192.
McLean, A. 1970. Plant communities of the Similkameen Valley, British Columbia, and their relationships to soils. Ecology. 40(4): 403-424.
McLean, A.; Tisdale, E.W. 1972. Recovery rate of depleted range sites under protection from grazing. Journal of Range Management. 25(3): 178-184.
Mee, W.; Barnes, J.; Kjelgren, R.; Sutton, R.; Cerny, T.; Johnson, C. 2003. Water wise: Native plants for Intermountain landscapes. Logan, UT: Utah State University Press. 220 p.
Meyer, S.E. 2008. Eriogonum Michx.; wild-buckwheat, buckwheatbrush. In: Bonner, F.T.; Karrfalt, R.P., eds. The woody plant seed manual. Agriculture Handbook 727. Washington, DC: U.S. Department of Agriculture, Forest Service: 499-503.
Meyer, S.E.; Paulsen, A. 2000. Chilling requirements for seed germination of 10 Utah species of perennial wild buckwheat (Eriogonum Michx. [Polygonaceae]). Native Plants Journal. 1(1): 18-24.
Miller, D.R.; Miller, G.L. 1992. Systematic analysis of Acanthococcus (Homoptera: Coccoidea: Eriococcidae) in the western United States. Transactions of the American Entomological Society. 118(1): 1-106.
Miller, R.F.; Chambers, J.C.; Pellant, M. 2014. A field guide for selecting the most appropriate treatment in sagebrush and pinon-juniper ecosystems in the Great Basin: Evaluating resilience to disturbance and resistance to invasive annual grasses, and predicting vegetation response. Gen. Tech. Rep. RMRS-GTR-322-rev. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. 66 p.
Monsen, S.B.; Stevens, R.; Shaw, N.L. 2004. Chapter 23. Shrubs of other families. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 2. Gen. Tech. Rep. RMRS-GTR-136-vol-2. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 597-698.
Mueggler, W.F.; Blaisdell, J.P. 1958. Effects on associated species of burning, rotobeating, spraying, and railing sagebrush. Journal of Range Management. 11(2): 61-66.
Mueggler, W.F.; Harris, C.A. 1969. Some vegetation and soil characteristics of mountain grasslands in central Idaho. Ecology. 50(4): 671-678.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Neely, E.E.; Barkworth, M.E. 1984. Vegetation on soils derived from dolomite and quartzite in the Bear River Range, Utah: A comparative study. Bulletin of the Torrey Botanical Club. 111(2): 179-192.
Ogle, D.; St. John, L.; Stannard, M.; Holzworth, L. 2012. Conservation plant species for the Intermountain West. Plant Materials Technical Note 24. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 57 p.
Ogle, D.; Tilley, D.; Cane, J.; St. John, L.; Fullen, K.; Stannard, M.; Pavek, P. 2011. Plants for pollinators in the Intermountain West. Plant Materials Tech. Note 2A. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 40 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Parkinson, H. 2003. Landscaping with native plants of the Intermountain Region. Tech. Ref. 1730-3. Boise, ID: U.S. Department of the Interior, Bureau of Land Management. 46 p.
Pavek, P.; Erhardt, B.; Heekin, T.; Old, R. 2012. Forb seedling identification guide for the Inland Northwest: Native, introduced, invasive, and noxious species. Pullman, WA: U.S. Department of Agriculture, Natural Resources Conservation Service, Pullman Plant Materials Center. 144 p.
Pickford, G.D.; Reid, E.H. 1943. Competition of elk and domestic livestock for summer range forage. The Journal of Wildlife Management. 7(3): 328-332.
Pitt, M.; Wikeem, B.M. 1990. Phenological patterns and adaptations in an Artemisia/Agropyron plant community. Journal of Range Management. 43(4): 350-358.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Plummer, A.P.; Christensen, D.R.; Monsen, S.B. 1968. Restoring big-game range in Utah. Pub. 68-3. Salt Lake City, UT: Utah Division of Fish and Game. 183 p.
Ralphs, M.H.; Panter, K.E.; James, L.F. 1991. Grazing behavior and forage preference of sheep with chronic locoweed toxicosis suggest no addiction. Journal of Range Management. 44(3): 208-209.
Ralphs, M.H.; Pfister, J.A. 1992. Cattle diets in tall forb communities on mountain rangelands. Journal of Range Management. 45(6): 534-537.
Ray, V.F. 1933. The Sanpoil and Nespelem: Salishan peoples of northeastern Washington. Seattle, WA: University of Washington Press. 237 p.
Reid, E.H.; Strickler, G.S.; Hall, W.B. 1980. Green fescue grassland: 40 years of secondary succession. Res. Pap. PNW-RP-274. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station. 49 p.
Reveal, J.L. 2005. Eriogonum Michaux. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 5 Magnoliophyta: Caryophyllidae, part 2. Flora of North America. http://www.efloras.org
Rice, M.E.; Merickel, F.; Macrae, T.C. 2017. The longhorned beetles (Coleoptera: Cerambycidae) of Idaho. The Coleopterists Bulletin. 71(4): 667-678.
Royal Botanic Gardens Kew [RBG Kew]. 2017. Seed Information Database (SID). Version 7.1. http://data.kew.org/sid/
Rust, S.K. 1999. Pinyon-juniper woodland classification and description in Research Natural Areas in southeastern Idaho. In: Monsen, S.B.; Stevens, R. comps. Proceedings – Ecology and management of pinyon-juniper communities within the Interior West: Sustaining and restoring a diverse ecosystem; 1997 September 15-18; Provo, UT. Proc. RMRS-P-9. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 82-93.
Seefeldt, S.S.; Germino, M.; DiCristina, K. 2007. Prescribed fires in Artemisia tridentata ssp. vaseyana steppe have minor and transient effects on vegetation cover and composition. Applied Vegetation Science. 10(2): 249-256.
Seefeldt, S.S; Leytem, A.B. 2011. Sheep bedding in the Centennial Mountains of Montana and Idaho: Effects on vegetation. Western North American Naturalist. 71(3): 361-373.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shaw, N. 1984. Producing bareroot seedlings of native shrubs. In: Murphy, P.M., comp. The challenge of producing native plants for the Intermountain area: Proceedings: Intermountain Nurseryman’s Association 1983 conference; 1983 August 8-11. Las Vegas, NV. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station: 6-15.
Shaw, N.L. 2004. Production and use of planting stock. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 3. Gen. Tech. Rep. RMRS-GTR-136-vol-3. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station. p. 745-768.
Shiflet, T.N. 1994. Rangeland cover types of the United States. Denver, CO: Society for Range Management. 152 p.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D. 2018. Direct surface seeding systems for the establishment of native plants in 2016 and 2017. In: Kilkenny, F.; Edwards, F.; Irwin, J.; Barga, S. 2018. Great Basin Native Plant Project: 2017 Progress Report. Boise, ID: U.S. Department of Agriculture, Rocky Mountain Research Station: 154-165.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D.; Shaw, N.; Kilkenny, F.F. 2017. Irrigation requirements for seed production of two Eriogonum species in a semiarid environment. Hortscience. 52(9): 1188-1194.
Skinner, D.M. 2008. Propagation protocol for production of container (plug) Eriogonum heracleoides Nutt. plants 10 cu. in. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2019 July 31].
St. John, L.; Tilley, D. 2014. Aberdeen Plant Materials Center report of activities. In: Kilkenny, Francis; Shaw, N.; Gucker, C. 2014. Great Basin Native Plant Project: 2013 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 99-102.
Steedman, E.V. 1928. Ethnobotany of the Thompson Indians of British Columbia: Based on field notes by James A. Teit. Washington, D.C.: United States Government Printing Office. 522 p.
Stevens, R. 2004. Management of restored and revegetated sites. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 1. Gen. Tech. Rep. RMRS-GTR-136-vol-1. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 193-198.
Stevens, R.; Jorgensen, K.R. 1994. Rangeland species germination through 25 and up to 40 years of warehouse storage. In: Monsen, S.B.; Kitchen, S.G., comps. Proceedings- Ecology and management of annual rangelands; 1992 May 18-22; Boise, ID. Gen. Tech. Rep INT-GTR-313. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Research Station: 257-265.
Stevens, R.J.; Jorgensen, K.R.; Young, A.S.; Monsen, S.B. 1996. Forb and shrub seed production guide for Utah. AG 501. Logan, UT: Utah State University Extension. 52 p.
Sutton, R.F.; Johnson, C.W. 1974. Landscape plants from Utah’s mountains. EC-368. Logan, UT: Utah State University, Cooperative Extension Service. 135 p.
Taylor, R.J. 1992. Sagebrush country: A wildflower sanctuary. Missoula, MT: Mountain Press Publishing Company. 211 p.
Tilley, D.; Fund, A. 2017. Plant guide: parsnipflower buckwheat (Eriogonum heracleoides). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 3 p.
Tropicos, Botanical Information System at the Missouri Botanical Garden [Tropicos]. 2019. Saint Louis, MO. Missouri Botanical Garden. http://www.tropicos.org/NameSearch.aspx [Accessed 2019 September 4].
Turesson, G. 1914. Slope exposure as a factor in the distribution of Pseudotsuga taxifolia in arid parts of Washington. Bulletin of the Torrey Botanical Club. 41(6): 337-345.
Turner, N.J.; Bouchard, R.; Kennedy, D.I. 1980. Ethnobotany of the Okanagan-Colville Indians of British Columbia and Washington. Victoria, BC: British Columbia Provincial Museum. 179 p.
U.S. Department of Agriculture, Forest Service, Great Basin Native Plant Project [USFS GBNPP]. 2014. Seed weight table calculations made in-house. Report on file. Boise, ID: U.S. Department of Agriculture, Forest Service, Boise Aquatic Sciences Laboratory.
University of Wyoming Extension [UW Ext.] 2018. Native plants for the Intermountain West. Laramie, WY: University of Wyoming. http://www.wyoextension.org/westernnativeplants
USDA Forest Service [USDA FS] 1937. Range plant handbook. Washington, DC: U.S. Department of Agriculture, Forest Service. 816 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2017. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2016. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 37 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2017. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Wagoner, S.J.; Shipley, L.A.; Cook, R.C.; Hardesty, L. 2013. Spring cattle grazing and mule deer nutrition in a bluebunch wheatgrass community. The Journal of Wildlife Management. 77(5): 897-907.
Walker, G.R.; Brotherson, J.D. 1982. Habitat relationships of basin wildrye in the high mountain valleys of central Utah. Journal of Range Management. 35(5): 628-633.
Weaver, J.E. 1915. A study of the root systems of prairie plants of southeastern Washington (Concluded). The Plant World. 18(10): 273-292.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Wikeem, B.M.; Pitt, M.D. 1979. Interpreting diet preference of California bighorn sheep on native rangeland in south-central British Columbia. Rangelands. 1(5): 200-202.
Wikeem, B.M.; Pitt, M.D. 1991. Grazing effects and range trend assessment on California bighorn sheep range. Journal of Range Management. 44(5): 466-470.
Wikeem, B.M.; Pitt, M.D. 1992. Diet of California bighorn sheep, Ovis canadensis californiana, in British Columbia: Assessing optimal foraging habitat. Canadian Field Naturalist. 106(3): 327-335.
Woolley, S.B., comp. 1936. Root systems of important range plants of the Boise River watershed: A catalogue of species excavated by Liter E. Spence [collaborator]. Unpublished paper on file at: U.S. Department of Agriculture, Forest Service, Intermountain Fire Sciences Lab, Missoula, MT. p. 59.
Yensen, E.; Shock, B.M.; Tarifa, T.; Mack, D.E. 2018. Forbs dominate diets of the threatened endemic northern Idaho ground squirrel (Urocitellus brunneus). Northwest Science. 92(4): 290-310.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
Zamora, B.; Tueller, P.T. 1973. Artemisia arbuscula, A. longiloba, and A. nova habitat types in northern Nevada. Great Basin Naturalist. 33(4): 225-242.
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2019. Parsnipflower buckwheat (Eriogonum heracleoides). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org/species/parsnipflower-buckwheat-eriogonum-heracleoides/