Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
February 2019
Nomenclature
Dalea searlsiae (A. Gray) Barneby is commonly called Searls’ prairie clover. This species belongs to the Fabaceae or pea or legume family, Amorpheae tribe, Kuhnistera section, and Compactae series (Barneby 1977). Searls’ prairie clover was named after Fanny Searls, a 19-year-old botanist who first collected the species in 1871 in southeastern Nevada (Tiehm cited in Belofsky et al. 201
Family
Fabaceae – Pea family
Genus
Dalea
Species
searlsiae
NRCS Plant Code
DASE3 (USDA NRCS 2017).
Subtaxa
Currently there are no varieties or subspecies recognized (Welsh et al. 2016; USDA NRCS 2017).
Synonyms
Petalostemon searlsiae A. Gray (Barneby 1989).
Common Names
Searls’ prairie clover.
Chromosome Number
Searls’ prairie clover is diploid. Chromosome numbers are 2n = 14, or rarely 16 (Barneby 1989; Welsh et al. 2016).
Hybridization
Natural hybrids of Searls’ prairie clover were not reported in the literature. Researchers in Utah successfully crossed Searls’ prairie clover with Blue Mountain prairie clover (D. ornata), but hybrid fertility has not been determined (S. Bushman, USDA ARS, personal communication, January 2019).
Distribution
Searls’ prairie clover occurs in southeastern California, throughout much of Nevada, western and southern Utah, and northern Arizona (Welsh et al. 2016). Populations occur in the Central Basin and Range, Colorado Plateau, Arizona Plateau, and Mojave Basin and Range Level III Ecoregions (Johnson et al. 2015). Somewhat isolated populations occur around the western edge of the Salt Lake Basin on the Nevada-Utah border (Barneby 1989).
Habitat And Plant Associations
Searls’ prairie clover occupies warm and cool desert shrublands, pinyon-juniper (Pinus–Juniperus spp.) woodlands, and less commonly, spruce-fir (Picea–Abies spp.) forests (Welsh et al. 2016). Populations are widely distributed but infrequent in sagebrush (Artemisia spp.) steppe and pinyon-juniper woodlands (Cane et al. 2012). dry bluffs, hillsides, barren valley knolls, alkaline, clay lake terraces, and sandy, rocky flats and slopes provide habitat for the species (Barneby 1977). Other common plant associates include shadscale saltbush (Atriplex confertifolia), cliffrose (Purshia spp.), and sunray (Enceliopsis spp.) (Barneby 1989).
Elevation
Searls’ prairie clover occurs from 3,300 to 9,200 ft (1,000–2,800 m) (Welsh et al. 2016), but plants are most common from 4,300 to 6,900 ft (1,300–2,100 m) in the Intermountain West.
Soils
Searls’ prairie clover is commonly associated with lime-rich soils, but it also occurs on sandstone and other bedrock substrates (Barneby 1977). Sandy or gravelly substrates in areas receiving 6 to 14 in (152–356 mm) of annual precipitation are common habitats (St. John et al. 2011).

Figure 1. Searls’ prairie clover growing in the Great Salt Lake Desert, Nevada. Photo: USFS.
Description
Searls’ prairie clover is a perennial with a woody taproot and mostly upright stems, 10 to 24 in (25–60 cm) tall that develop from a short, shallow caudex (Welsh et al. 2016). Stems are glabrous and clumped with alternate, deciduous, odd–pinnate compound leaves (0.8 to 2.2 in [2–5.5 cm] long) along the lower half (Fig. 1) (Barneby 1977; Welsh et al. 2016). Leaves are commonly comprised of five or seven leaflets, but number of leaflets can range from 3 to 11. Leaflets are thick, folded or arched, smooth and green above, gland dotted below, and 3 to 18 mm long and 0.8 to 6 mm wide (Fig. 2) (Barneby 1977, 1989; St. John et al. 2011; Welsh et al. 2016). Searls’ prairie clover foliage gives off a strong scent when bruised (Barneby 1989).
Flowers are tiny and perfect with five pink to purple petals (Cane et al. 2012; Johnson et al. 2015). They are clustered on moderately dense, cylindrical spikes (0.6–5 in [1.6–13 cm] long and 8–12 mm wide)] (Barneby 1989; K. Connors, USDA ARS, personal communication, January 2019). Calyces are ten-ribbed and mostly glabrous except for the lobes, which have long hairs. Flowers have five stamens. The style is 4 to 5 mm long (Welsh et al. 2016). Although the small, hairy fruits (pods) contain two ovules, just one seed matures (St. John et al. 2011; Johnson et al. 2015). Seeds are villous (Welsh et al. 2016).

Figure 2. Searls’ prairie clover plants growing in research plots in northern Utah. Photo: S. Bushman, USDA, ARS.
Reproduction
Searls’ prairie clover reproduces from seed. Flowers bloom upward from the base to the tip of the inflorescence over a 2- to 3-week period in late spring or early summer (Barneby 1989; Johnson et al. 2015). In an agricultural setting, plants flowered and produced seed in their second year (Shock et al. 2018). In a common garden study of 20 Searls’ prairie clover accessions, the number of inflorescences produced per plant ranged from 9 to 61 (Bhattarai et al. 2011). In a common garden in Logan, Utah, individual Searl’s prairie clover spikes averaged 131 flowers in 2 years of observations (Cane et al. 2012).
Pollination
Cross pollination is necessary for Searls’ prairie clover seed production. Cross–pollinated flowers produced 4 to 10 times as much seed as self-pollinated flowers during experiments at the ARS Pollinating Insect Research Unit (PIRU [now the Pollinating Insect-Biology, Management, Systematics Research]) in Logan, Utah (Cane et al. 2012). Just 3 to 4% of self-pollinated flowers on caged inflorescences produced seed, and 80 to 97% of this seed was small and likely non-viable. Half of flowers that were manually outcrossed set seed that was large and likely viable (Cane 2007). The finding of limited seed production without outcrossing is also supported by studies conducted by Bhattarai et al. (2011), which revealed that much of the species diversity occurs within populations (76%), suggesting substantial gene flow through pollen flow, seed dispersal, or both.
Bees are the primary pollinators. When researchers netted insects in natural Searls’ and Blue Mountain prairie clover populations, 114 bees representing 22 species were collected. Netting in Searls’ prairie clover populations was done at two sites in northwestern Utah, outside the natural range of the species. The most prevalent bees netted were carder or potter bees (Anthidium spp.), bumblebees (Bombus spp.), plasterer bees (Colletes spp.), and long-horned bees (Eucera and Melissodes spp.) (Cane et al. 2012). At plots in Utah, many insects including butterflies, wasps, flies, and moths were observed on Searls’ prairie clover flowers and may also contributed to pollination (K. Connors, USDA ARS, personal communication, January 2019).
Ecology
Searls’ prairie clover is a long-lived perennial. Stems sprout from the crowns in early or late spring. Plants flower and produce seed by late spring or early summer, often by early July. Stems die back in the fall, and plants are dormant through the winter (Shock et al. 2018; K. Connors, USDA ARS, personal communication, January 2019). Plants cut during the growing season typically regrow new stems that will flower (K. Connors, USDA ARS, personal communication, January 2019). This growth pattern suggests Searls’ prairie clover should be fire and grazing tolerant, especially by late summer. However, Searls’ prairie clover is sensitive to grazing in early summer and crowns can be damaged by trampling (S. Bushman, USDA ARS, personal communication, January 2019).
Searls’ prairie clover was present each year when burned sites were monitored for 5 years following an early November fire on the western edge of Arizona’s Colorado Plateau. Abundance before or after the fire was not reported for the basin big sagebrush (Artemisia tridentata subsp. tridentata), rubber rabbitbrush (Ericameria nauseosa), and cheatgrass (Bromus tectorum) community. Post-fire seeding of grasses with and without raking also occurred in the monitored burned area (Brooks et al. 2010).
Wildlife And Livestock Use
Overgrazing may have reduced the abundance and distribution of Searls’ prairie clover (St. John et al. 2011). The presence of Searls’ prairie clover in the diets of cattle may improve nutrient use and prevent bloating (Bushman cited in Belofsky et al. 2014). In creosote brush vegetation in the West Mojave Desert, domestic sheep showed a strong preference for Searls’ prairie clover late in the grazing season, a 60- to 90-day period in late February and early March (Phillips et al. 1996).
Nutritive value. Compounds that can be toxic to livestock and wildlife, including swainsonine, selenium, and nitrotoxins, which are present in other legumes, were barely detectable and well below toxic levels in Searls’ prairie clover (Bhattarai et al. 2011; Johnson et al. 2015).
Forage quality was assessed for plants of 20 different accessions from Utah and Nevada growing in a common garden in Hyde Park, Utah. Acid detergent fiber averaged 35% (range: 28–39%). Neutral-detergent fiber averaged 46% (range: 40 to 50%). Crude protein averaged 18% (range: 17–21%) (Bhattarai et al. 2011). At the conclusion of the common garden study, researchers and land managers selected three of the 20 accessions for release as selected germplasms for commercial seed production (see Releases section; Johnson et al. 2015). Their forage quality traits are provided below (Table 1).
Table 1. Forage quality (evaluated in June and October for 2 yrs) of Searls’ prairie clover selected germplasm releases when grown together in Hyde Park, Utah (Johnson et al. 2015).
|
Collection |
Acid-detergent fiber |
Neutral-detergent fiber |
Crude protein |
|
––––––––––––––––––––%–––––––––––––––––––– |
|||
|
Fanny |
36.5a |
49.5a |
17.9b |
|
Carmel |
36.2a |
46.5ab |
17.0b |
|
Bonneville |
32.6b |
43.9b |
20.7a |
Within a column, values followed by different letters are significantly different (P < 0.05).
Current Medicinal Use
Searls’ prairie clover has antimicrobial properties, with the highest concentration in the roots. These antimicrobial substances were active against several human illness causing bacteria (Streptococcus mutans, Bacillus cereus, and wild and antibiotic-resistant Staphylococcus aureus) (Belofsky et al. 2012, 2014).
Horticulture
Prolific flowering and attractiveness to pollinators make Searls’ prairie clover an option for home landscaping and roadside plantings (St. John et al. 2011).
Revegetation Use
Searls’ prairie clover is recommended for diversification of revegetation or restoration seed mixes for sites within its range receiving annual precipitation of 6 to 14 in (152–356 mm) (St. John et al. 2011). It is also recommended for enhancing pollinator habitat (Fig. 3). In a guide to pollinator plantings for the Intermountain West, prairie clovers (Dalea spp.) were described as important to bees and exhibiting a moderate growth rate (Ogle et al. 2011). As a legume that contributes to atmospheric nitrogen fixation, the presence of Searls’ prairie clover can increase a site’s nutrient availability, improve forage quality, and occupy niches otherwise colonized by weed species (Review in Johnson et al. 2015).

Figure 3. Honey bee (Apis spp.) visiting Searls’ prairie clover flowers. Photo: D. Tilley, USDA ARS.
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for Searls’ prairie clover, generalized provisional seed zones developed by Bower et al. (2014), may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat:moisture index). In Figure 5, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Seedlot Selection Tool (Howe et al. 2017) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.

Figure 4. Searls’ prairie clover growing in seed production plots at Oregon State University’s Malheur Experiment Station in Ontario, Oregon. Photo: USFS.
Common garden experiments using seed collected over a wide portion of the Searls’ prairie clover range coupled with genetic analyses using neutral DNA markers (amplified fragment length polymorphisms [AFLPs]) guided the development of these plant materials for use in restoration (Bhattarai et al. 2011).
Common garden studies of 20 wildland Searls’ prairie clover populations revealed strong relationships between growth, seed production, and collection site precipitation (Bhattarai et al. 2011). Searls’ prairie clover seed was collected from sites in Utah and Nevada ranging from latitudes of 37°13’N to 41°28’N and longitudes of 111°25’W to 117°53’W. Collection site elevations ranged from 4,350 to 6,680 ft (1,326–2,036 m), mean annual temperature from 45 to 52 °F (7.2–11.3 °C), and mean annual precipitation from 6 to 14.6 in (152–372 mm). Common gardens were established at two northern Utah sites separated by about 10 mi (16 km). Gardens had silt loam soils and received 17 in (430 mm) of average annual precipitation. Forty plants from seed collected at each of the wild populations were planted at each garden. Plant size and the number and weight of inflorescences were highly correlated with collection site precipitation (P = 0.0002), suggesting plants from high–precipitation sites may not grow as large or produce as much seed when transplanted to sites with low precipitation. Flowering date was negatively correlated with collection–site elevation (P = 0.0285) and positively correlated with mean annual temperature (P = 0.03), indicating that flowering occurred earlier for plants from high-elevation sites with low mean annual temperatures (Table 2; Bhattarai et al. 2011; Johnson et al. 2013).
Table 2. Characteristics of 20 accessions of Searls’ prairie clover growing in common gardens in northern Utah (Bhattarai et al. 2011). First value represents the average for all accessions. The range is provided in parentheses.
|
Common garden location |
Dry matter yield (g/plot) |
Plant height (in) |
Stems (no.) |
Inflorescences (no.) |
Foliage diameter (in) |
Inflorescence weight¹(g/plot) |
Flowering date² (gardens combined) |
|
Hyde Park, UT |
121 |
12.6 |
11 |
27 |
19 |
–––– |
June 13 |
|
Millville, UT |
95 |
–––– |
–––– |
39 |
–––– |
46 |
¹Plants checked daily, and mature seed harvested as available until the entire florescence was harvested. Inflorescences were bulked by replication, dried in a greenhouse, and weighed.
² Flowering date is number of days from January 1 (160=June 8 and 173=June 20).
Genetic analyses of the same 20 populations revealed that 76% of the genetic variation was present within collections (P < 0.0001) or populations for this outcrossing species (Bhattarai et al. 2011). Phenotypic traits and within-collection genetic similarities were not significantly correlated, suggesting that increased genetic diversity did not necessarily mean larger plant size or greater fecundity. Collections from northwestern Utah had low genetic diversity and also low dry-matter yield and fewer inflorescences (Bhattarai et al. 2011; Johnson et al. 2013).
Occurrence Map
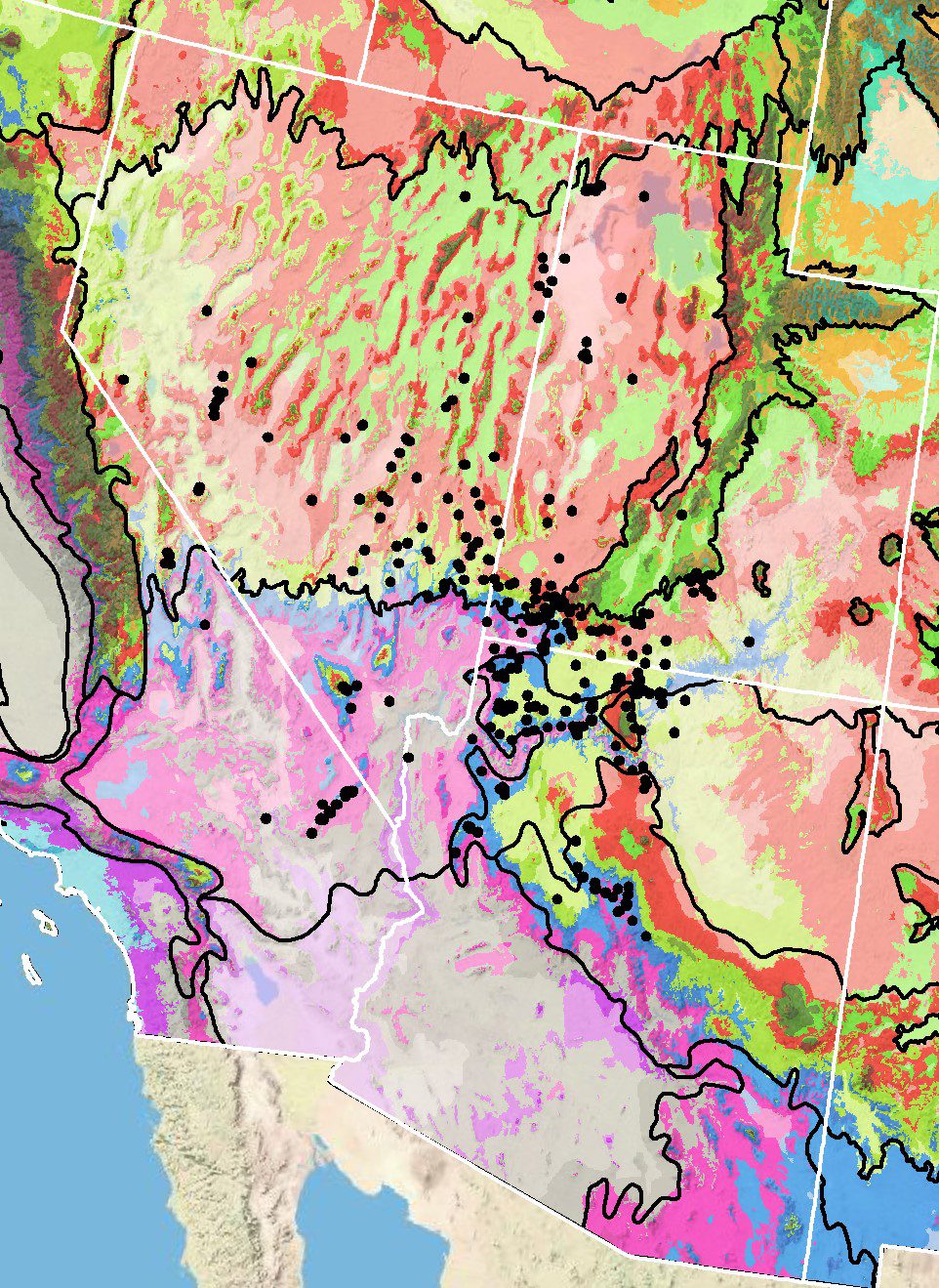
Figure 5. Distribution of Searls’ prairie clover (black circles) based on geo-referenced herbarium specimens and observational data from 1871-2011 (CPNWH 2017; SEINet 2017; USDI USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USDI USGS.
Releases
Three Searls’ prairie clover releases were recommended following the genetic and phenotypic analyses conducted by Bhattarai et al. (2011; study described above). These ARS releases represent natural track selected germplasm for use in revegetation and restoration of western rangelands (Table 3). Seed source selection should be guided by local climate, resistance to local pests, and intended use (St. John et al. 2011).
‘Fanny’ select germplasm came from seed collected from a 6,730-ft (2,050 m) site at Patterson Pass, east–central Nevada where annual precipitation averages 12.6 in (321 mm). It originated from an area within the Central Basin and Range ecoregion representing USDA plant hardiness zone 5b and heat zone 5. It was released for use in Nevada and southwestern Utah (Johnson et al. 2015).
‘Carmel’ select germplasm came from seed collected from a 5,560-ft (1,695 m) site in south-central Utah where annual precipitation averages 13.6 in (347 mm). The site was within the Colorado Plateau ecoregion in USDA plant hardiness zone 7a and heat zone 7. It was released for use in the Colorado Plateau (Johnson et al. 2015).
‘Bonneville’ select germplasm came from seed collected from 4,590 ft (1,400 m) near the Nevada border in northwestern Utah where annual precipitation averages 7 in (178 mm). It originated from an area within the Central Basin and Range ecoregion representing USDA as plant hardiness zone 6b and heat zone 7. It was identified as a genetically distinct metapopulation and released for use in northwestern Utah (Johnson et al. 2015).
Wildland Seed Collection
Wildland seed is easily collected by hand; the pods readily detach from the inflorescence (Ogle et al. 2012). When seeds were compared under x-ray, large plump seeds contained endosperm but small shrunken seeds did not, suggesting that rough estimates of seed viability can be assessed visually (Cane et al. 2012).
Wildland seed certification. Wildland seed collected for direct sale or for establishment of agricultural seed production fields should be Source Identified through the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program that verifies and tracks seed origin (Young et al. 2003; UCIA 2015). For seed that will be sold directly for utilization in revegetation, collectors must apply for certification prior to making collections. Applications and site inspections are handled by the state where collections will be made. For seed that will be used for planting agricultural seed fields or nursery propagation more details of the collection site and procedures are required. Seed collected by most public and private agencies following established protocols may enter the certification process directly without certification agency site inspections when protocols include collection of all data required for Source Identified certification (see Agricultural Seed Certification section). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect. Permits or permission from public or private land owners are required for all collections.
Table 3. Growth characteristics of Searls’ prairie clover natural track–selected germplasms from two common garden sites (Hyde Park [H] and Millville [M]) in northern Utah (Johnson et al. 2015).
|
Collection |
Dry matter yield |
Stems (no.) |
Plant height (in) |
Canopy diameter (in) |
Inflorescences (no.) |
Inflorescences (g/plot) |
Flowering date (H & M) |
|
Fanny |
161a (H) |
11a (H) |
13.8b (H) |
22.4a (H) |
35a (H) |
61a (M) |
June 8b |
|
Carmel |
178a (H) |
12a (H) |
15.4a (H) |
22.8a (H) |
26b (H) |
73a (M) |
June 15a |
|
Bonneville |
80b (H) |
9b (H) |
11.8c (H) |
14.2b (H) |
16c (H) |
30b (M) |
June 15a |
Values within the same column and common garden followed by a different lower case letter are significantly different (P < 0.05) .
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (Young et al. 2020; UCIA 2015). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Flowers bloom up from the base to the tip of the inflorescence, and often the tip of the inflorescence may be just producing flowers as the lower portion is shedding mature seed (Barneby 1989; Johnson et al. 2015; K. Connors, USDA ARS, personal communication, January 2019). Mature seed detaches quickly once ripe (Shock et al. 2018; K. Connors, USDA ARS, personal communication, January 2019), making monitoring for seed ripening critical for maximizing harvests. Bureau of Land Management’s Seeds of Success (BLM SOS) field crews made two wildland seed collections in 2016. Both harvests (June 20 and 22) came from an arid 5,282-ft (1,610 m) site in Garfield County, Utah (BLM SOS 2017).
Some suggest flowering occurs over a short 2- to 3-week period in late spring or early summer because seed is dropped quickly (Barneby 1989; Johnson et al. 2015). Connors and other’s experience collecting wildland seed in Utah and Nevada indicates that the timing of seed ripening is highly variable and related to unpredictable moisture availability within its range. If winter precipitation is normal to good, late spring/summer seed production is good. If winter precipitation is poor, seed production is poor or lacking. Searls’ prairie clover takes advantage of monsoons or other precipitation and may produce seed whenever moisture allows (K. Connors, USDA ARS, personal communication, January 2019).
Collection Methods
Because seed pods are easily detached from the inflorescence, small and clean collections of wildland seed can be made by shaking or gently stripping the inflorescence over a bag or tarp (Ogle et al. 2012). Longer inflorescences may disperse ripe seeds when unopened flowers still occur at the tip. Seed harvested when immature will not ripen (K. Connors USDA ARS, personal communication, January 2019).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of Searls’ prairie clover.
Post-Collection Management
To minimize potential seed damage from seed-feeding insects, researchers conducting studies in Utah stored seed with an insecticidal strip when seed was drying and before long-term storage (Bhattarai et al. 2011).
Seed Cleaning
Two seed cleaning processes were described by researchers conducting small plot research on Searls’ prairie clover. Researchers at FRRL air dried seed in the greenhouse, then processed it through a Wintersteiger thresher (Model LD180, Des Moines, IA) and did final cleaning using handheld sieves and a seed blower (Bhattarai et al. 2011). Staff at USDA ARS Plant Materials Center (IPMC) in Aberdeen, Idaho, hammermilled the seed pods using a 0.25-in (6 mm) screen. Inert material (unfilled or insect-damaged seed) was removed using an air-screen cleaner with a 2.75 mm top and 1.55 mm bottom screen and a relatively high air speed of 4. Seed was further cleaned to remove similar-sized weed seed (e.g., pigweed) using an indent cleaner and #2 drum (Ogle et al. 2012).
Seed Storage
Searls’ prairie clover seed is orthodox. Seed retained 100% viability after being dried and stored for 4 months at 68 °F (20 °C) and 15% relative humidity (RH) (RBG Kew 2017). Researchers in Utah, stored seed in the dark at 37 °F (3 °C) at an RH of 20 to 25% (Bhattarai et al. 2011).
Seed Testing
Association of Seed Analysts (AOSA) developed rules for tetrazolium (TZ) tests of prairie clover seed (AOSA 2010). Seed is imbibed at 68 to 77 °F (20–25 °C) and the seed coats of seed that fails to imbibe are nicked or clipped. Seed is then soaked for 2 to 72 hours in a 1% TZ solution at 86 to 95 °F (30–35 °C). Viability is assessed based on staining patterns. Immature seed may stain unevenly (AOSA 2010).
Germination Biology
A good proportion of Searls’ prairie clover seed is hard or physically dormant. Seed has a an extremely hard coating but with scarification it germinates rapidly, generally within 5 days with warm temperatures (Bushman et al. 2015; S. Bushman, USDA ARS, personal communication, January 2019). Seed scarification is necessary for reliable and uniform germination (Ogle et al. 2012). Tests conducted by RGB Kew (2017) resulted in 100% germination of scarified seeds (n = 10). Scarification partially removed or chipped the seed coat, and seeds were then incubated on 1% agar plates at 61 °F (16 °C) and 12-hour alternating dark and light conditions (RBG Kew 2017).
Seed dormancy can be removed by acid or mechanical scarification. Johnson et al. (2015) used sandpaper blocks in a cement mixer to scarify seed lots. At the IPMC, natural winter stratification of untreated seed planted in the fall failed to produce any seedlings (Ogle et al. 2012).
Bushman et al. (2015) found that a large percentage of Searls’ prairie clover seed (Fanny and Bonneville) was hard and viable. Scarification was required for good germination (Table 4). Neither planting depth nor soil composition exhibited effects as large as seed scarification on emergence. Seed viability was evaluated by TZ tests. Seeds that imbibed water and stained were considered viable, and seeds that imbibed water and stained only after nicking of the seed coat was considered viable and hard. Seeds were acid or mechanically scarified or left untreated. Acid scarification involved a 5-minute soak in 98% sulfuric acid, and a 3-minute tap water rinse followed by air drying. Mechanical scarification was done by rubbing the seed between two pieces of 120-grit sandpaper for 30 seconds. Treated and untreated seeds were then placed on top of a 1:4 peat-sand mix and subsequently covered by 6 or 19 mm of sifted sand. Scarification improved germination of both seed lots but improvements were much greater for Fanny than Bonneville. A more aggressive mechanical cleaning procedure for Fanny may have partially scarified this seed (St. John cited in Bushman et al. 2015).
Table 4. Viability and germination (%) of scarified (acid or mechanical) and untreated seed of Searls’ prairie clover germplasm releases (‘Bonneville’ and ‘Fanny’).
|
Source |
Treatment |
Viable |
Viable |
Total |
Germ- |
|
——————%—————— |
|||||
|
Bonneville |
Acid |
87 |
4 |
91 |
83 |
|
Mechanical |
69 |
20 |
89 |
74 |
|
|
Untreated |
3 |
84 |
87 |
5 |
|
|
Fanny |
Acid |
51 |
0 |
51 |
68 |
|
Mechanical |
37 |
15 |
52 |
65 |
|
|
Untreated |
31 |
30 |
61 |
49 |
|
Although seeding depth (6–19 mm) and soil composition (fine sandy loam or silty clay loam)had less impact on emergence than scarification, the findings provide insights to seedling emergence under natural conditions. While deeper planting did not have a significant effect on total emergence of Bonneville it did reduce total emergence of Fanny from 90 to 74% and deeper planting significantly reduced the rate of emergence for both seed lots (P < 0.05). Total emergence of scarified Fanny seed declined significantly as clay content in soils increased. Emergence rate for scarified and untreated Bonneville seed was faster in fine sandy loams than in soils with higher clay content. A slower germination rate with increased seeding depth under rangeland conditions could result in lower total emergence when rainfall events are missed. At the conclusion of the soil composition experiments, researchers lifted the soil crust that formed on high clay content soils and found germinated seedlings that were unable to penetrate the crust (Bushman et al. 2015).
The precise acid scarification method used by the FRRL to treat about 2.5 lbs (1.1 kg) of seed is as follows. Seeds were immersed in 98% sulfuric acid for 5 minutes. Seeds were then thoroughly rinsed and dried (Johnson et al. 2011, 2015).
Wildland Seed Yield And Quality
Seed lots from 17 wildland collections averaged 148,000 seeds/lb (326,000 seeds/kg) (St. John et al. 2011). Seed lots from 20 wildland collections from Utah and Nevada, ranged from 122,040 to 174,200 seeds/lb (269,000–384,000 seeds/kg). RGB Kew (2017) reported similar seed weights of 111,565 to 154,804 seeds/lb (245,955–341,279 seeds/kg). For the germplasm releases, Fanny averaged 136,560 seeds/lb (301,000 seeds/kg), Carmel averaged 113,500 seeds/lb (250,220 seeds/kg), and Bonneville averaged 157,430 seeds/lb (347,000 seeds/kg) (Johnson et al. 2015).
Post-cleaning seed yield and quality was reported for a single seed lot collected from Garfield County, Utah (USFS BSE 2017). A half pound of seed was received by the USFS Bend Seed Extractory. Clean weight was 0.108 lbs, for a clean out rate of 22%. There were 151,401 seeds/lb (333,776 seeds/kg). Purity was 99%, fill was 98%, and pure live seed (PLS) was 146,889 seeds/lb (66,628 seeds/kg). These results suggest seed can be cleaned to high levels of purity and fill, and viability of fresh seed was high (USFS BSE 2017). Wildland seed lots used for experiments at OSU MES were 81% and 84% viable based on TZ testing (Shock et al. 2017a).
Marketing Standards
Ogle et al. (2012) report that using an air screen in the seed cleaning process can yield purities close to 100%. Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment used in nurseries, while some rangeland seeding equipment handles less clean seed well.
Agricultural Seed Production
The mostly upright growth habit and production of numerous inflorescences near the top of the plant make it a potential candidate for commercial seed production and mechanical harvesting (Bhattarai et al. 2011; Bushman et al. 2015). Productive farming of seed will likely require bees for pollination and control of seed-feeding beetles (St. John et al. 2011).
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Site Preparation
Researchers at IPMC recommend seeding Searls’ prairie clover 0.25 to 0.5 in (0.6–1.3 cm) deep into a firm weed-free bed (St. John et al. 2011). At OSU MES, seed production plots were grown in silt loam soils with a pH of 8.3 and 1.1% organic matter (Fig. 6). Seeding into weed-free plots was done in the fall. Seed was deposited on the soil surface using a custom small-plot grain drill with disk openers. Seed was subsequently covered with sawdust at rate of 7 g/ft (24 g/m) of row and then by row cover, which was removed in April (Shock et al. 2018). In direct surface seeding trials at OSU MES, stand emergence for Searls’ prairie clover was low (0.1–3% stand) but better with row cover than without regardless of other seed coverings (none, sand, or sawdust) (Shock et al. 2017a).

Figure 6. Searls’ prairie clover seed production plots growing at the Oregon State University, Malheur Experiment Station in Ontario, Oregon. Photo: USFS.
Seed Pretreatments
Natural stratification of overwintering untreated seed planted in the fall failed to produce seedlings in Aberdeen, Idaho. Acid or mechanical scarification is necessary for reliable and uniform germination. A high percentage of seed is hard (≥ 90% for 1-year-old seed). Germination can be increased to more than 90% with a 5-minute 98% sulfuric acid treatment (Ogle et al. 2012). Johnson et al. (2015) used sandpaper blocks in a cement mixer to scarify seed lots.
Weed Management
In seed production research plots at IPMC, weed barrier fabric and hand rouging were used to control weeds (Ogle et al. 2012). At OSU MES, weeds were controlled with cultivation or hand rouging (Shock et al. 2018). In common garden experiments in Utah, hand weeding occurred as needed in all years of the study (Bhattarai et al. 2011). Landscape fabric, although a high initial cost, provides weed control and may allow for cleaner and more complete seed harvests (see Seed Harvesting section; K. Connors, USDA ARS, personal communication, January 2019). At the ARS Forage and Range Research Laboratory (FRRL) in Logan, Utah, Searls’ prairie clover emerged late in the spring. This allowed for glyphosate treatments to control winter annual weeds and injure emerging perennial weeds without affecting Searls’ prairie sprouting (K. Connors and S. Bushman, USDA ARS, personal communications, January 2019). Early spring glyphosate treatments provided control of all but field bindweed (Convolvulus arvensis) at FRRL research plots.
Herbicides to control weeds were evaluated on established (4-year-old) Searls’ prairie clover plants in study plots in Utah (Ransom and Edvarchuk 2010). Herbicides were applied (20 gal/ac [gpa] and 30 lbs/in² [psi]) on May 21, and plants were evaluated before and after treatments. Injury was severe initially but declined by 35 days after treatment (DAT) with oxyfluorfen and flumioxazin. Injury was moderately high but declined by 35 DAT with quinclorac and imazamox. Injury was severe and persistent with clopyralid treatments. Pendimethalin, bromoxynil, and butyric acid caused little injury. Plant and seed head biomass were greater, but not significantly so, for plants treated with oxyfluorfen, bromoxynil, and butyric acid than for untreated plants. Treated plants in 3 × 36 ft (1 × 11 m) plots had vegetative biomass of 489 to 518 g/plot, while the vegetative biomass in untreated plots was 423 g. Seedhead biomass of treated plots was 110 to 146 g/plot, while it was 99 g for untreated plots. Increased vegetative and seedhead biomass production with herbicide treatments may have been due to low injury levels by 35 DAT (5–16%) and slight suppression of field bindweed (Ransom and Edvarchuk 2010).
Seeding
Seed farming research conducted at IPMC led researchers to recommend seeding Searls’ prairie clover 0.25 to 0.5 in (0.6–1.3 cm) deep in firm, weed–free beds. The recommended full seeding rate is 7 PLS lbs/ac (7.8 kg/ha) for approximately 25 PLS/linear ft (82/m) (St. John et al. 2011). At OSU MES, Searls’ prairie clover stands were established by dropping seed on the soil surface at a rate of 20 to 30 seeds/linear ft (65–100 seeds/m) in the fall and subsequently covering it with sawdust and row cover that was removed in the following spring (Shock et al. 2018).
Establishment And Growth
Searls’ prairie clover may flower and produce seed in its first year, but harvestable amounts may not be available until the second year. Three Searls’ prairie ecotypes established successfully after spring seeding near Pleasant View, Colorado. These plants produced seed in their first year, but yields were not reported (Johnson and Bushman 2014). In seed production research plots at OSU MES, Searls’ prairie clover began flowering and produced seed the second year after fall planting (Shock et al. 2017b, 2018).
Irrigation
Seed yield and plant water requirements were evaluated at OSU MES for 6 years (2011–2016) (Shock et al. 2017b, 2018). Maximum seed yield occurred when irrigation and precipitation totaled 13 to 17 in (340–430 mm)/year. Irrigation studies added 0, 4, and 8 in (0–200 mm) of water through a subsurface drip irrigation system (12 in [30 cm] deep), with four bi-weekly increments starting at the time of flower bud formation (Table 5).
Over the course of the study, annual precipitation averaged 9.8 in (249 mm) (range: 7–11 in [179–292 mm]). Seed yield was maximized with 4 to 8 in (100–200 mm) of irrigation in warmer, drier years (2012–2016) and without irrigation in a cool, wet year (2011) (Table 6). Researchers concluded that water requirements for seed production were low and natural precipitation in Ontario, Oregon, provided a substantial amount of the water needed for seed production. Seed yield showed a quadratic response to irrigation from 2012 through 2015 with maximum seed yields at 6.5 in (164 mm) of added water in 2012, 8 in (203 mm) in 2013, 8.7 in (222 mm) in 2014, and 6.3 in (161 mm) in 2015, and 5.3 in (134 mm) in 2016. Averaged over 6 years, relative seed yield was highest with applied water plus fall, winter, and spring precipitation totaling 16 in (412 mm) (Table 7; Shock et al. 2018). Irrigation plots were seeded November 25, 2009, seed was covered with sawdust and row cover. Row cover was removed April 2010, irrigation started in 2011, and plots were hand weeded as necessary (Shock et al. 2017b).
Table 5. Flowering timing as related to timing of irrigation of Searls’ prairie clover seed production plots growing at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2018).
|
Year |
Flowering |
Irrigation |
||||
|
Start |
Peak |
End |
Start |
End |
Harvest |
|
|
2011 |
June 8 |
June 20 |
July 20 |
May 27 |
July 6 |
July 21 |
|
2012 |
May 23 |
June 10 |
June 30 |
May 11 |
June 21 |
July 10 |
|
2013 |
May 13 |
––––– |
June 15 |
May 8 |
June 19 |
June 29 |
|
2014 |
May 15 |
June 4 |
June 24 |
May 6 |
June 17 |
July 1 |
|
2015 |
May 13 |
May 26 |
June 16 |
May 5 |
June 17 |
June 22 |
|
2016 |
May 11 |
May 28 |
June 10 |
May 3 |
June 14 |
June 16 |
Table 6. Maximum seed yield of Searls’ prairie clover seed production plots by irrigation amount applied based on quadratic response. Plots grown at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2018).
|
Year |
Highest yield |
Fall + Winter + |
Growing degree days (50–86 °F) Jan–June |
Additional water |
|
2011 |
263.0 |
14.4 |
476 |
0 |
|
2012 |
309.1 |
8.4 |
682 |
6.5 |
|
2013* |
45.5 |
5.3 |
733 |
8.0 |
|
2014 |
233.8 |
8.1 |
741 |
8.7 |
|
2015 |
377.9 |
10.4 |
895 |
6.3 |
|
2016 |
245.0 |
10.0 |
810 |
5.3 |
|
Average |
231.7 |
9.1 |
665** |
6.7 |
* Seed pods had extensive damage from seed–feeding beetles in 2013 (Shock et al. 2018).
**22-year average.
Pollinator Management
Experiments conducted on Searls’ prairie clover at the ARS Pollinating Insect Research Unit, showed that seed production is limited without outcrossing, and outcrossed spikes produced 4 to 10 times the seed of selfed flowers (Cane et al. 2012). Providing manageable pollinators or encouraging and sustaining native bee populations in Searls’ prairie clover crops will benefit seed production operations (Cane 2007). For practical pollinator management in natural areas, see the Wildland Seeding and Planting section. Researchers in Utah, however, report excellent seed yields without adding pollinators even in areas with low natural pollinator levels (S. Bushman, USDA ARS, personal communication, January 2019).
Table 7. Seed yield (lbs/ac) for fall-seeded (November 2009) Searls’ prairie clover in response to no additional irrigation and supplemental irrigation of 4 and 8 in [100–200 mm) at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2018).
|
Year |
Supplemental irrigation (in/season*) |
||
|
0 |
4 |
8 |
|
|
––––––––––––––––Seed yield (lbs/ac)–––––––––––––––– |
|||
|
2011 |
262.5b |
231.0ab |
196.2a |
|
2012 |
175.4a |
288.6b |
302.8b |
|
2013 |
14.8a |
31.7b |
44.3c |
|
2014 |
60.0a |
181.3b |
232.1b |
|
2015 |
221.1a |
330.5b |
343.9b |
|
2016 |
148.5a |
238.7b |
222.2b |
|
Average |
147.0a |
216.9b |
223.6b |
* Irrigation was delivered through an underground drip irrigation system from bud formation to seed set. Numbers within a row followed by different letters are significantly different (P < 0.05).
Pest Management
Seed-feeding beetles (Acanthoscelides oregonensis and Apion amaurum) infest prairie clovers. These pests will likely require management to maximize seed yields (St. John et al. 2011). Powdery mildew (Leveillula papilionacearum) was also identified on plants growing at OSU MES but the extent of the damage was not reported (Mohan and Shock 2014).
In research plots at OSU MES, Searls’ prairie clover seed was damaged extensively by seed-feeding beetles in 2013. Seed was also damaged in 2014 and 2015 but to a much lesser degree (Table 6). In 2015, bifenthrin (0.04 lb/ac [0.05 kg/ha]) and novaluron insecticides (0.03 lb/ac [0.04 kg/ha]) were applied to plants in May. Insecticides were applied at night to minimize harm to pollinators. Effectiveness of the treatments was not determined (Shock et al. 2018), but seed beetles were not observed in flowers in 2016 (Shock et al. 2017b).
Seed Harvesting
Searls’ prairie clover seed is generally produced in harvestable quantities in the second year of growth. Because flowers bloom upward along the inflorescence over a 2- to 3-week period, plants are producing flowers near the top of the inflorescence while mature seed is being dropped from the bottom of the inflorescence. This seed development pattern means that maximum seed yields may require several harvests. A vacuum, flail vac, or soft-brush harvester can be used for multiple harvests during seed maturation (Ogle et al. 2012; Johnson et al. 2015). Adhesive pod sealants showed limited value in delaying seed shatter due to the loose arrangement of seeds along the inflorescence (S. Bushman, USDA ARS, personal communication, January 2019).
At OSU MES, Searls’ prairie clover seed was harvested in a single, once-over, manual harvest, which represented a compromise between immature, mature, and dehiscing seed. Seed ripening occurred over a 2-week period, and mature seeds detached readily (Shock et al. 2018). At the ARS Forage and Range Research Laboratory (FRRL) in Logan, Utah, harvests of seed production research plots were most successful when all seed had shattered and was then collected from the ground using a backpack leaf blower in vacuum mode (K. Connors, USDA ARS, personal communication, January 2019). This harvest process worked best for crops with landscape fabric but also worked on bare soil, although harvests from soils required more cleaning. Collecting shattered seed from landscape fabric allowed for a single harvest of likely fully ripe seed. When mechanical harvest of seed was tested at FRRL, just 20% of the maximum seed yield was collected. The harvester cut all plant stems to about 3 or 5 in (8–13 cm) (K. Connors, USDA ARS, personal communication, January 2019).
Seed Yields And Stand Life
At OSU MES, Searls’ prairie clover produced harvestable seed crops beginning when plants were 2 years old. Good seed production continued through year 6 (Shock et al. 2017b, 2018), but seed yield was not evaluated beyond the sixth harvest year. Highest annual seed yield (cleaned bulk seed) averaged 232 lbs/ac (260 kg/ha) and ranged from 45 to 378 lbs/ac (51–424 kg/ha) with irrigation. Seed yield was maximized when irrigation plus precipitation totaled 13 to 17 in (340 to 430 mm)/year (Shock et al. 2018). For more on the effects of irrigation on seed yield, see Irrigation section above.
Nursery Practice
Searls’ prairie clover transplants can be grown successfully under normal greenhouse conditions (Fig. 7), but seed scarification is generally necessary for reliable and uniform germination (St. John et al. 2011). At the FRRL, transplants were grown in the greenhouse from seed stored at 37 °F (3 °C) and RH of 20 to 25%. Seeds were germinated on moistened blotter paper in plastic boxes kept at room temperature. No scarification was described. Once seeds germinated, the seedlings were transplanted into plugs, which were later transferred to cone-tainers. The greenhouse was maintained at 86 °F (30 °C) day and 59 °F (15 °C) night temperatures. Fertilization and watering provided for 90 days of greenhouse growth before field transplanting (Bhattarai et al. 2011). Transplanting occurred in May 2006. Plants were spaced 1.6 ft (0.5 m) apart, routinely weeded and watered in the establishment year, and regularly weeded but unwatered in all subsequent years of the study (Bhattarai 2010).
In other investigations at FRRL, seedlings were produced by first acid scarifying seed for 5 minutes, then rinsing and drying them. Before planting, the seeds were treated with Rhizobium and just before planting they were soaked overnight in water at room temperature. Most soaked seeds had a 1 to 2 mm radical. These seeds were planted in Q plugs placed within cone-tainers. The tops of the Q plugs were covered with 5 to 10 mm soil, because emergence through soil was important to removal of the seed coat from the cotyledons. When seed coats were retained, seedlings were weak or died. After establishment, seedlings were treated periodically with a low concentration balanced fertilizer (K. Connors, USDA ARS, personal communication, January 2019).

Figure 7. Searls’ prairie clover container–grown seedling. Photo: J. Cane, USDA ARS.
Wildland Seeding And Planting
Searls’ prairie clover is recommended for use as a minor component of rangeland seeding mixes where adapted (St. John et al. 2011). It provides wildlife forage and pollinator habitat (Ogle et al. 2011). Sandy soils are preferred (St. John et al. 2011).
For drill seeding, the recommended full seeding rate is 7 PLS lbs/ac (7.8 kg/ha) (approximately 25 PLS/linear ft [82 PLS/m]) (St. John et al. 2011). This rate should be doubled for broadcast seeding. When seeding in a mix, the rate should be adjusted to reflect the desired mix percentage. Results are best when seed is planted 0.25 to 0.5 in (0.6–1.3 cm) deep in firm, weed-free beds, using press wheels to provide good soil contact. Searls’ prairie clover seedlings should be protected from grazing until well established (St. John et al. 2011).
Searls’ prairie clover requires cross pollination and bee visits for high seed production. When seeded as a minor component of a seed mix, Searls’ prairie clover plants will likely be spaced too widely for sequential visits during a given foraging trip (Cane et al. 2012). Seeding Searls’ prairie clover densely in clumps, ideally where soil is sandy, will provide for greater pollinator visitation, higher seed production, and longer-term persistence (St. John et al. 2011).
Searls’ prairie clover is also sought after by livestock and wildlife. Protection of newly planted areas may be necessary (K. Connors, USDA ARS, personal communication, January 2019).
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Kevin Connors (retired) and Shaun Bushman both with USDA ARS FRRL.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Association of Official Seed Analysts [AOSA]. 2010. AOSA/SCST Tetrazolium testing handbook. Contribution No. 29. Lincoln, NE: Association of Official Seed Analysts.
Barneby, R.C. 1977. Dalea imagines. Memoirs of The New York Botanical Garden, Volume 27. Bronx, NY: The New York Botanical Garden. 891 p.
Barneby, R.C. 1989. Intermountain Flora Volume 3, Part B: Fabales. In: Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K., eds. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Bronx, NY: The New York Botanical Garden. 279 p.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Belofsky, G.; Aronica, M.; Diamond, J.; Foss, E. 2012. Flavanoids of Dalea searlsiae: A plant under investigation for reintroduction to native habitats of the Great Basin. Planta Medica. 78(11): 1231-1231.
Belofsky, G.; Aronica, M.; Foss, E.; Diamond, J.; Santana, F.; Darley, J.; Dowd, P.F.; Coleman, C.M.; Ferreira, D. 2014. Antimicrobial and antiinsectan phenolic metabolites of Dalea searlsiae. Journal of Natural Products. 77(5): 1140-1149.
Bhattarai, K. 2010. Phenotypic and genetic characterization of wildland collections of western and Searls prairie clovers for rangeland revegetation in the western USA. Logan, UT: Utah State University. Dissertation. 154 p.
Bhattarai, K.; Bushman, B.S.; Johnson, D.A.; Carman, J.G. 2011. Searls prairie clover (Dalea searlsiae) for rangeland revegetation: Phenotypic and genetic evaluations. Crop Science. 51(2): 716-727.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Brooks, M.L.; Klinger, R.; Chase, J.; Deuser, C. 2010. Evaluating postfire seeding treatments designed to suppress cheatgrass (Bromus tectorum) in a ponderosa pine forest on the Colorado Plateau. 05-2-1-17 Joint Fire Science Program Final Report. 38 p.
Bushman, B.S.; Johnson, D.A.; Connors, K.J.; Jones, T.A. 2015. Germination and seedling emergence of three semiarid western North American legumes. Rangeland Ecology and Management. 68(6): 501-506.
Cane, J.H. 2007. Pollinator and seed predator studies. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2006 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 88-92.
Cane, J.H.; Weber, M.; Miller, S. 2012. Breeding biologies, pollinators, and seed beetles of two prairie-clovers, Dalea ornata and Dalea searlsiae (Fabaceae: Amorpheae), from the Intermountain West, USA. Western North American Naturalist. 72(1): 16-20.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Howe, G.; St, Clair, B.; Bachelet, D. 2017. Seedlot Selection Tool. Corvallis, OR: Conservation Biology Institute. https://seedlotselectiontool.org/sst/
Johnson, D.A.; Bushman, B.S. 2014. Developing protocols for maximizing establishment of Great Basin legume species. In: Kilkenny, F.; Shaw, N.; Gucker, C., eds. Great Basin Native Plant Project: 2013 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 71-73.
Johnson, D.A.; Bushman, B.S.; Bhattarai, K.; Connors, K.J. 2011. Notice of release of Majestic germplasm and Spectrum germplasm western prairie clover: Selected class of natural germplasm. Native Plants Journal. 12(3): 249-256.
Johnson, D.A.; Bushman, B.S.; Connors, K.J.; Bhattarai, K.; Jones, T.A.; Jensen, K.B.; Parr, S.D.; Eldredge, E.P. 2015. Notice of release of Fanny germplasm, Carmel germplasm, and Bonneville germplasm Searls’ prairie clover: Selected class of natural germplasm. Native Plants Journal. 16(3): 265-274.
Johnson, D.A.; Bushman, B.S.; Jones, T.A.; Bhattarai, K. 2013. Identifying geographically based metapopulations for development of plant materials indigenous to rangeland ecosystems of the western USA. In: Lüttge, U.; Beyschlag, W.; Francis, D.; Cushman, J., eds. Progress in Botany Vol 74: Springer, Berlin, Heidelberg: 265-291.
Mohan, S.K.; Shock, C.C. 2014. Etiology, epidemiology, and management of diseases of native wildflower seed production. In: Kilkenny, F.; Shaw, N.L.; Gucker, C.L., eds. Great Basin Native Plant Project: 2013 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 107-108.
Ogle, D.; Bushman, S.; Johnson, D.; St. John, L.; Tilley, D. 2012. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Dalea searlsiae (A. Gray) Barneby seeds. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://npn.rngr.net/propagation/protocols [Accessed 2018 June 13].
Ogle, D.; Tilley, D.; Cane, J.; St. John, L.; Fullen, K.; Stannard, M.; Pavek, P. 2011. Plants for pollinators in the Intermountain West. Plant Materials Tech. Note 2A. Boise, ID: U.S. Department of Agriculture, Natural Resources Conservation Service. 40 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Phillips, R.L.; McDougald, N.K.; Sullins, J. 1996. Plant preference of sheep grazing in the Mojave Desert. Rangelands. 18(4): 141-144.
Plant Conservation Alliance [PCA]. 2015. National seed strategy for rehabilitation and restoration 2015-2020. Washington, DC: U.S. Department of the Interior, Bureau of Land Management. 52 p.
Ransom, C.; Edvarchuk, K. 2010. Identification of herbicides for use in native forb seed production. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2009 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 98-102.
Royal Botanic Gardens Kew [RBG Kew]. 2017. Seed Information Database (SID). Version 7.1. http://data.kew.org/sid/
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Shock, C.C.; Feibert, E.B.; Saunders, L.D.; Parris, C. 2012. Seed production of Great Basin native forbs – Legume seed scarification. In: Shaw, N.; Pellant, M., eds. Great Basin Native Plant Project: 2011 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 127-132.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Sanders, L.D.; Kilkenny, F.; Shaw, N. 2017a. Direct surface seeding systems for the establishment of native plants in 2016. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2016. OSU AES Ext/CRS 157. Corvallis, OR: Oregon State University: 123-130.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D.; Johnson, D.A.; Bushman, B.S.; Shaw, N.; Kilkenny, F. 2017b. Prairie clover and basalt milkvetch seed production in response to irrigation. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2016. OSU AES Ext/CrS157. Corvallis, OR: Oregon State University: 153-159.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D.; Shaw, N.; Kilkenny, F. 2018. Irrigation requirements for native buckwheat seed production in a semi-arid environment. In: Kilkenny, F; Edwards, F.; Irwin, J.; Barga, S., eds. Great Basin Native Plant Project: 2017 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 110-118.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Shaw, N.; Kilkenny, F.F. 2018. Irrigation requirements for seed production of three leguminous wildflowers of the U.S. Intermountain West. HortScience. 53(5): 692-697.
St. John, L.; Tilley, D.; Ogle, D.; Johnson, D.; Bushman, S. 2011. Plant guide: Searls prairie clover (Dalea searlsiae). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 3 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2017. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2016. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 37 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2017. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 2015. A Utah Flora. Fifth Edition, revised. Provo, UT: Brigham Young University. 990 p.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2019. Searls’ prairie clover (Dalea searlsiae). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: http://greatbasinfirescience.org/western-forbs-restoration