Authorship
Gucker, Corey; Shaw, Nancy
Publication Date
August 2018
Nomenclature
Heliomeris multiflora Nutt., hereafter referred to as showy goldeneye, belongs to the Heliantheae tribe of the Asteraceae or Aster family (Schilling 2006).
Family
Asteraceae – Aster family
Genus
Heliomeris
Species
multiflora
NRCS Plant Code
HEMU3 (USDA NRCS 2017).
Subtaxa
The Flora of North America recognizes three varieties: brevifolia (Greene ex. Wooton & Standl.) Yates, multiflora Nutt., and nevadensis (A. Nelson) Yates (Schilling 2006)
Synonyms
Gymnolomia brevifolia Green ex. Wooton & Standl., Gymnolomia nevadensis A. Nelson, and Viguiera multiflora (Nutt.) S.F. Blake (Schilling 2006).
Common Names
Showy goldeneye, little sunflower, many-flowered sunflower, mountain sunflower, rosinweed (USDA FS 1937).
Chromosome Number
Chromosome numbers are 2n = 16, 18 for varieties brevifolia and multiflora (Keil and Pinkava 1976; Welsh et al. 1987) and 2n = 16, 18, or 32 for variety nevadensis (Solbrig et al. 1972; Hickman 1993; Schilling 2006).
Hybridization
Showy goldeneye hybrids with other species were not reported in the literature.
Distribution
Showy goldeneye is a western species. Variety multiflora is the most widely distributed, occurring in Montana, Idaho, Wyoming, Colorado, Utah, Nevada, Arizona, New Mexico, and Texas. Variety nevadensis is more southerly, occurring in California, Nevada, Utah, Arizona, New Mexico, and as far south as Jalisco, Mexico. Variety brevifolia occurs in Utah, Arizona, and New Mexico (Schilling 2006). Variety multiflora is found at higher elevations or in moister habitats than variety nevadensis where their distributions overlap (Cronquist 1994). In Montana, variety multiflora is primarily found in the southern part of the state (USDA FS 1937). In Utah, variety multiflora occurs in all counties, variety nevadensis occurs in Grand, Juab, Uintah, and Washington counties, and variety brevifolia occurs only in Beaver County (Welsh et al. 1987; USDA NRCS 2017). In California, variety nevadensis is found east of the Sierra Nevada range (Hickman 1993).
Habitat And Plant Associations
Showy goldeneye is considered common over its range and locally abundant across a broad altitudinal range from valley grasslands to spruce (Picea spp.) forests (USDA FS 1937). It is common along roadsides (Figs. 1 and 2) and on open, dry to moderately moist slopes receiving annual precipitation of 18 to 26 in (450 to 650 mm) (Shaw 1958; Andersen and Holmgren 1976; Cronquist 1994; Tilley 2012). Showy goldeneye is common in basin big sagebrush (Artemisia tridentata subsp. tridentata), mountain big sagebrush (A. t. subsp. vaseyana), pinyon-juniper (Pinus–Juniperus spp.), mountain brush, and quaking aspen (Populus tremuloides) communities (Shaw and Monsen 1983). It is also a common species in the tall forb rangeland type generally found at 6,300 to 9,000 ft (1,900-2,700 m) along streams, near springs, in forest openings, and in shrubland, woodland, and forest understories from the southern Wasatch range in Utah north to southern Montana (Shiflet 1994).

Figure 1. Showy goldeneye growing in a forest opening along a roadside in Utah. Photo: USDI BLM UT931 SOS.
Showy goldeneye is abundant or frequent in shrubland and woodland types in Colorado, Wyoming, and Idaho. In Colorado, it was common in a browse-shrub type occurring between grasslands and forests on the east side of the Rocky Mountains and along the border between sagebrush and pinyon-juniper zones in the western part of the state. Dominant overstory vegetation in the type was Gambel oak (Quercus gambelii), mountain snowberry (Symphoricarpos oreophilus), and Saskatoon serviceberry (Amelanchier alnifolia) (Costello 1944). Showy goldeneye occurred in 7 of 15 Saskatoon serviceberry mountain brush stands growing near Victor in eastern Idaho. In this vegetation type, showy goldeneye cover was 1 to 3% (Major and Rejmanek 1992). In Jackson Hole Wildlife Park in Wyoming, showy goldeneye was abundant in hillside quaking aspen woodlands and common in big sagebrush shrublands. Hillside quaking aspen stands were relatively open, xeric, and occurred on mostly south-facing slopes with rapidly draining soils. Showy goldeneye was not found in densely shaded, mesic quaking aspen stands occurring on level terrain (Reed 1952).
Variety associations. Variety nevadensis is generally found in drier and lower elevation habitats than varieties multiflora and brevifolia, although there is overlap (Welsh et al. 1987; Cronquist 1994; Stevens and Monsen 2004a). Variety multiflora often occurs in sagebrush, pinyon-juniper and other woodland types, forests, and riparian sites. Variety nevadensis is also found in sagebrush and pinyon-juniper vegetation, but it is sometimes found in saltbush (Atriplex spp.) and blackbrush (Coleogyne ramosissima) communities (Welsh et al. 1987; Cronquist 1994). In montane habitats, variety brevifolia is often found in shaded areas of canyons and woodlands; variety multiflora along roadsides, on rocky valley slopes, and in open woodlands; and variety nevadensis along roadsides and on dry, rocky slopes (Schilling 2006).

Figure 2. Variety nevadensis in sagebrush along a roadside in Nevada. Photo: USDI BLM NV040 SOS.
Elevation
Showy goldeneye occurs at elevations ranging from 3,000 to 11,800 ft (900-3,600 m) (Hickman 1993; Cronquist 1994; Stevens and Monsen 2004a). Variety nevadensis is found at the lowest elevations in this range (Hickman 1993) and multiflora at the highest (Cronquist 1994). Variety brevifolia is found from 7,900 to 9,800 ft (2,400–3,000 m) (Schilling 2006). In California, variety nevadensis occurs from 4,000 to 7,900 ft (1,200–2,400 m) (Munz and Keck 1973; Hickman 1993). In Utah, variety multiflora occurs from 4,400 to 9,400 ft (1,340–2,870 m) and variety nevadensis from 4,500 to 7,000 ft (1,370–2,135 m) (Welsh et al. 1987).
Soils
Showy goldeneye occurs on shallow to deep, basic to acidic soils ranging from moist to dry with heavy clay to sandy or gravelly textures (USDA FS 1937; Clary et al. 1968; Stevens and Monsen 2004a). In subalpine vegetation on the Wasatch Plateau in Utah, showy goldeneye was most common at sites between mesic and xeric (Ellison 1954). In the Coconino National Forest near Flagstaff, Arizona, showy goldeneye frequency was greatest (33%) in woodland openings where soil moisture was 7.3%, which was significantly lower than in woodland sites with greater canopy cover (P < 0.05) (Abella and Springer 2006).
Soil depths vary in showy goldeneye habitats. On the Mancos Shale barrens in southwestern Colorado, showy goldeneye occurred in plots with soil depths of 2 in (5 cm) or less (Silva and Ayers 2016). In twoneedle pinyon-Utah juniper (Pinus edulis–Juniperus osteosperma) vegetation on No Man’s Land Mesa near Kanab, Utah, showy goldeneye was more abundant on upland shallow breaks than upland sands. Shallow breaks had steeper slopes (5–30%) and occupied shallower, better-drained, very fine, sandy loam soils than upland sands with gentle slopes (1-10%) and deep, loamy, fine sandy soils. Both sites were primarily north-facing (Mason et al. 1967). Showy goldeneye was common in subalpine Thurber’s fescue (Festuca thurberi) grasslands in western Colorado’s White River National Forest where well-drained loam soils ranged from shallow to deep (Klemmedson 1956).
Although described as tolerant of acidic and basic soils (Stevens and Monsen 2004a), showy goldeneye occurred (0.05% cover, 35.3% frequency) on plots with granite but not limestone soil in the Mule Mountains of Cochise County, Arizona. The pH was 6.1 on granite and 7.8 on limestone (Wentworth 1981).
Description
Showy goldeneye is a multi-stemmed perennial that develops from a branched, somewhat woody caudex. The root system ranges from a short taproot to an almost fibrous network (USDA FS 1937; Cronquist 1994; Schilling 2006). Plants often produce adventitious buds in the first growing season (Schilling 2006). Stems are slender and wiry, branched, up to 51 in (130 cm) tall, and terminate in yellow flowers (Fig. 3) (USDA FS 1937; Andersen and Holmgren 1976; Schilling 2006). Stems range from hairless to having straight, stiff, appressed hairs (Schilling 2006). Leaves are opposite or alternate, linear to egg-shaped, and range from 0.4 to 3.5 in (1-9 cm) long and up to 1.1 in (2.8 cm) wide. Leaves are subsessile or with short petioles, entire to partly serrate and weakly three-nerved with glands dotting the undersides (USDA FS 1937, Craighead et al. 1963; Welsh et al. 1987; Cronquist 1994; Schilling 2006). Leaf characteristics are useful in distinguishing varieties (USDA FS 1937; Schilling 2006).

Figure 3. Heliomeris multiflora var. multiflora growing in Utah. Photo: USDI BLM UT933 SOS.
Plants produce numerous (6-30+), sunflower-like flower heads (Fig 4) measuring up to 1.5 in (3.8 cm) across with enlarged, rounded bases with two unequal rows of densely hairy bracts (USDA FS 1937; Craighead et al. 1963; Weber 1976; Shaw 1995; Schilling 2006). Flower heads can be solitary or occur in few-flowered inflorescences (Hickman 1993). Flower heads generally have 8 to 16 outer, infertile, yellow, petal-like ray flowers and 50 or more inner, perfect, fertile, golden disk flowers (USDA FS 1937; Hickman 1993; Cronquist 1994; Shaw 1995; Schilling 2006). Ray flowers are up to 1 in (2.5 cm) long, and the petal is notched at the tip (Fig. 5). The central, disk-flower portion of the flower head becomes cone shaped with age (Shaw 1995; Schilling 2006). Showy goldeneye fruits are cypselas or false achenes, often referred to as achenes in the literature. The cypsela is smooth, black or gray striate, 1.2 to 3 mm long, and compressed longitudinally making it four angled. Cypselas do not have scales or pappi (USDA FS 1937; Welsh et al. 1987; Schilling 2006).

Figure 4. Heliomeris multiflora var. nevadensis growing in Utah. Photo: SOS Provo Shrub Sciences Laboratory (PSSL).
Descriptions of varieties. Showy goldeneye varieties can often be distinguished by leaf arrangement and shape (Schilling 2006). Variety brevifolia produces mostly alternate leaves that are elliptic to oval, and 8 to 28 mm wide with flat to obscurely revolute margins. Variety nevadensis produces mostly opposite leaves that are linear to lance-shaped, 2 to 8 mm wide, and about 7 to 20 times as long with strongly revolute margins (Munz and Keck 1973; Hickman 1993; Cronquist 1994; Schilling 2006). Variety multiflora also produces mostly opposite leaves that are linear to lance-ovate shaped, 5 to 20 mm wide and about two to three times as long with flat margins and pointed tips (Cronquist 1994; Schilling 2006).
Reproduction
Showy goldeneye reproduces sexually from seed, which is produced by the perfect disk flowers (USDA FS 1937; Schilling 2006).

Figure 5. Flower of variety nevadensis growing in Nevada. Photo: USDI BLM NV040 SOS.
Phenology
Flowering occurs from May to October. Flowers are most common in late summer (USDA FS 1937; Craighead et al. 1963; Munz and Keck 1973; Andersen and Holmgren 1976; Schilling 2006). In a subalpine meadow in the Rocky Mountains near Gunnison, Colorado, the average flowering date for showy goldeneye was August 15; some plants were still flowering on September 2 in one year (Thomson 1980). In Utah, showy goldeneye seeds matured in August or September (Plummer et al. 1968). In subalpine vegetation on Utah’s Wasatch Plateau, showy goldeneye flowering began about 60 days after snowmelt and continued for about 30 days (Ellison 1954).
Pollination
Good seed production requires pollination, and bees are the common flower vistors. While some suggest flowers can be wind pollinated (Stevens et al. 1996), experiments conducted in mountain meadows in Colorado’s Gunnison Basin found that flowers protected from pollinators failed to set seed (Pleasants 1980).
Many bee species, including the cherry plum miner (Andrena cerasifolii) and Brooks’ andrena (A. brooksi), visit showy goldeneye flowers (LaBerge 1986; Larkin 2004). In observations made from June 22 to September 8 in meadows near Crested Butte, Colorado, there were 2 mountain bumble bee (Bombus appositus), 9 western bumble bee (B. occidentalis), 52 frigid bumble bee (B. frigidus), 68 forest bumble bee (B. sylvicola), 93 yellow-fronted bumble bee (B. flavifrons), and 139 two-form bumble bee (B. bifarius) visits to showy goldeneye flowers (Pyke 1982). In Peaceful Valley in Boulder County, Colorado, sweat bees (Halictus cressonii and H. oryx), mining bees (Panurginus porterage), and bumble bees (B. juxtus and B. bifarius) were observed on showy goldeneye flowers. At this site, showy goldeneye was considered a principal, late-August bee flower (Cockerell 1919).
In mountain meadows in Gunnison Basin, Colorado, two-form bumble bees were the primary visitors to showy goldeneye flowers, although there were also visits from yellow-fronted and western bumble bees. The attractiveness of showy goldeneye, determined by the number of bees on showy goldeneye flowers in a 30-minute census/number of open flowers per square meter, ranked fifth or sixth out of 13 or 14 flower species. In this area, showy goldeneye flowered from early August to early September with peak flowering from August 20 to 23 (Pleasants 1980).
Ecology
Showy goldeneye tolerates pioneer, early seral, and late-seral conditions (Stevens and Monsen 2004b). Showy goldeneye was an early colonizer of bare soil created by slope slides in subalpine vegetation on Utah’s Wasatch Plateau (Ellison 1954) and can also be found in forests and woodlands where it receives at least a few hours of sun each day (USDA FS 1937).
Although plant abundance may be greater with increasing light availability, many studies report showy goldeneye in late-seral, shaded forests. On the western slope of the Rocky Mountains near Crested Butte, Colorado, showy goldeneye was somewhat more frequent in sun (31.5%) than shade (20%) when aspen groves and open Thurber’s fescue meadows were compared. Sun sites were drier and experienced greater temperature and light variability than shade sites (Louda et al. 1987). In the Douglas-fir zone of northern Utah’s Cache National Forest, showy goldeneye cover averaged 3% on unshaded and 1% on shaded south-slope plots, but cover was equal (1%) on shaded and unshaded east-slope plots (Sharif and West 1968). Cover was not significantly different in cabled and untreated twoneedle pinyon-Utah juniper woodlands on the Fort Stanton Experimental Range in Lincoln County, New Mexico, where cabling significantly reduced total density and canopy cover of trees (P < 0.1). Cover of showy goldeneye was 3.2% on sites cabled about 20 years earlier and 4.3% on untreated sites. (Rippel et al. 1983).
Seed And Seedling Ecology
Showy goldeneye is quick to establish on disturbed sites, develops rapidly, and can rapidly dominate disturbed sites (Stevens and Monsen 2004a). In shortgrass prairie northwest of Flagstaff, Arizona, the proportion of showy goldeneye seedlings establishing on gopher mounds was significantly greater than the proportion of available disturbed habitat or Northern pocket gopher (Thomomys talpoides) mounds available (P < 0.006) (Martinsen et al. 1990). Showy goldeneye reestablished naturally on disturbed sites in Yellowstone and Glacier National Park (Majerus 1999).
Disturbance Ecology
The disturbance response for showy goldeneye is typically positive with respect to fire but more unpredictable with grazing and may depend on the timing and the grazer.
Fire response. Many studies report early appearance and increased abundance of showy goldeneye on burned sites and some suggest greater increases with severe fires. In southwestern Utah, showy goldeneye rapidly invaded burned Wyoming and basin big sagebrush vegetation (Stevens and Monsen 2004a). On the Coconino National Forest, Arizona, showy goldeneye was a dominant in ponderosa pine (Pinus ponderosa) stands burned severely 3 years earlier (Sabo et al. 2009). In the Apache-Sitgreaves National Forest in Arizona, showy goldeneye occurred on 10-year-old moderately and severely burned sites following a June fire (Neeley 2012).
Showy goldeneye is often more common on or restricted to burned sites when burned and unburned areas are compared. It occurred on a 1-year-old burned site (36% frequency, 2.5% cover), but it was absent from a nearby unburned site in the lower foothill zone of Utah’s central Wasatch Mountains. The August fire, which was considered intense, burned in Gambel oak-dominated vegetation (Poreda and Wullstein 1994). When 23 oak brush stands were compared in the Uinta National Forest in Utah, showy goldeneye was more frequent in stands burned within the last 30 years (16%) than in adjacent unburned stands (2.3%) (Kunzler et al. 1981). Showy goldeneye cover was also generally greater in burned than unburned Gambel oak vegetation assessed on 1-, 2-, 9-, and 18-year-old burns near Salt Lake City, Utah (McKell 1950). Showy goldeneye made up 21% of the vegetation composition on burned but 0% at unburned sites in a Mexican pinyon (Pinus cembroides) woodland in southeastern Arizona’s Whetstone Mountains. The June 1968 fire was “intense”, and effects were evaluated in 1974 and 1975 (Barsch 1977). Cover of showy goldeneye was considerably greater in logged and slash burned than untreated twoneedle pinyon-Utah juniper woodlands in Arizona’s Coconino National Forest. Five years after fuel wood harvesting and slash burning, cover of showy goldeneye was 5.9% on burned sites that were canopy covered before the fire (determined by the presence of stumps) and 8.6% on burned sites in the canopy interspaces. Cover was 0% beneath tree canopies and 0.3% in the interspaces on unburned sites (Haskins and Gehring 2004).
In a single study evaluating recovery as it related to fire severity, showy goldeneye was most common on severely burned sites. In the Apache-Sitgreaves National Forest, Arizona, showy goldeneye was an indicator species of more severely burned sites— those that were not cut prior to a June wildfire. At post-fire years 2, 3, and 10, showy goldeneye was more common on uncut burned than cut burned sites, which experienced lower severity fire (Shive et al. 2013).
Grazing response. Both increases and decreases in showy goldeneye abundance have been reported with grazing. Some suggest that plants are not particularly palatable. They increase on heavily grazed sites by aggressively colonizing spaces left once more palatable species are injured or killed. In northwestern Colorado, showy goldeneye was referred to as a pest on poor condition rangelands severely grazed by cattle (USDA FS 1937). On the Mogollon Rim in Arizona, abundance of showy goldeneye differed by site when grazed and ungrazed sites were compared (Rambo and Faeth 1999). At one open meadow site on the Rim, the relative abundance of showy goldeneye was not different within and outside 8- to 9-year-old exclosures protected from grazing by cattle, elk, and deer. At another open meadow site on the Rim, showy goldeneye was not found in grazed plots and made up 2.2% of the relative abundance in protected plots. When 1-year-old exclosures and grazed plots were compared, relative abundance of showy goldeneye was 1.1% of grazed and 0.5% of protected vegetation at one site and 0.4% of grazed and 2.5% of protected vegetation at another site (Rambo and Faeth 1999).
In a mountain brush vegetation enclosure in the central Wasatch Mountains of Utah, frequency of showy goldeneye fluctuated with increased duration of protection from livestock. The exclosure was constructed in 1949 when frequency of showy goldeneye was 34%. In 1958, frequency of showy goldeneye in the exclosure decreased to 29%, and in 1967 increased to 40% (Eastmond 1968).
In Ephraim Canyon, Sanpete County, Utah, cover of showy goldeneye was greater on sites protected from livestock, primarily sheep, and increased with increased duration of protection (Stevens et al. 1992). Grazing effects were evaluated by monitoring two subalpine meadow watersheds from 1921 to 1983. The watersheds were separated by only 900 ft (270 m). Watershed A was protected from 1921 through 1983. Watershed B was moderately to heavily grazed for all but 3 years from 1915 to 1952 and then protected. Cover over time on these watersheds is provided in Table 1 (Stevens et al. 1992).
Table 1. Cover (%) of showy goldeneye on two subalpine meadow watersheds in Sanpete County, Utah, from 1921–1983. Watershed A was protected from livestock (primarily sheep) grazing starting in 1921. Watershed B was protected starting in 1953 (Stevens et al. 1992).
|
Year |
1958 |
1961 |
1983 |
|
——Cover %—–– |
|||
|
Watershed A |
4.0 |
3.5 |
8.1 |
|
Watershed B |
0.2 |
0.4 |
3.0 |
Wildlife And Livestock Use
Showy goldeneye is consumed by big game, small mammals, birds, and livestock. Because it provides green forage from early spring until late in the growing season, it has been recommended for wildlife habitat improvement (Stevens 2004; Stevens and Monsen 2004a). Although utilized by a variety of wildlife and livestock, palatability ratings vary. USDA FS (1937) suggested it was worthless to poor cattle forage, fairly good sheep forage, and utilized in drought conditions when more palatable forage was lacking. Others indicated that big game will seek out showy goldeneye populations, and heavy grazing by big game and livestock has occurred in at least some seasons (Stevens 2004; Stevens and Monsen 2004a).
Wildlife. Elk (Cervus canadensis), deer (Odocoileus spp.), and pronghorn (Antilocapra americana) utilize showy goldeneye (Wallace 1991; Stevens 2004; Stevens and Monsen 2004a). In the White Mountains of Arizona, showy goldeneye made up 3.3% of the composition of female elk diets in the summer. Diets were monitored for 3 years (Wallace 1991). Stevens and Monsen (2004a) reported that mule deer (O. hemionus) seek out showy goldeneye and concentrate on sites where plant densities are high.
Northern pocket gophers (Thomomys talpoides) and deer mice (Peromyscus maniculatus) feed on showy goldeneye. In west-central Colorado, showy goldeneye was found in the stomachs of northern pocket gophers at a high of 0.5% volume even though it was not in vegetation plots in the Black Mesa study area (Ward and Keith 1962). On the Wasatch Plateau in central Utah, showy goldeneye was not a preferred northern pocket gopher food but did receive some use (Aldous 1951). In a feeding trial, deer mice consumed significantly more showy goldeneye than spotted knapweed (Centaurea stoebe) seeds when these two species were the provided food sources (P < 0.01; Pearson et al. 2011).
Showy goldeneye provides food and habitat for birds. Birds feed on showy goldeneye seeds (Stevens and Monsen 2004a), and the seeds are considered a preferred food of Columbian sharp-tailed grouse (Tympanuchus phasianellus columbianus) (Stevens 2004). In the Huachuca Mountains in Cochise County, Arizona, a Montezuma quail (Cyrtonyx montezumae mearnsi) nest was constructed with a grass floor and a roof of many showy goldeneye stems (Wallmo 1954).
Livestock. Descriptions of livestock use and palatability of showy goldeneye vary. On summer range in Utah, use of showy goldeneye was 10% by cattle and 5% by sheep based on observations from 1960 to 1966 (Cook 1983). Showy goldeneye was absent from summer sheep range in southwestern Utah grazed for about 90 years. In a protected area, grazed only minimally by livestock, production of showy goldeneye was 226 lbs/ac (257 kg/ha), and it made up 9% of the total vegetation biomass (Bowns and Bagley 1986). In subalpine Thurber’s fescue grasslands in Colorado’s White River National Forest, showy goldeneye increased as range condition deteriorated with grazing largely by sheep. The grazing regime was not described. Showy goldeneye was 0% of the total vegetation cover in good, 1.4% in fair, and 3.6% in poor condition range (Klemmedson 1956). In subalpine vegetation on the Wasatch Plateau, Utah, showy goldeneye decreased with increased grazing (Ellison 1954).
Showy goldeneye has been suggested for use in revegetation and restoration efforts as a highly palatable species to seed or plant strategically to attract animals away from depleted range. By attracting animals, highly palatable seedings would allow degraded areas time to rest and recover naturally (Monsen 2004).
Nutritional Value
Showy goldeneye can provide succulent forage from April to October (Monsen and Stevens 2004), but it is also thought to have been the cause of poisoning in grazing animals in the Southwest (Shaw 1995). In a greenhouse study, plants accumulated toxic levels of nitrate when ammonium nitrate was added to the soil. Leaves contained up to 4.3% nitrate when plants were fertilized at a rate of 194 lbs N/ac (220 kg/ha), and plants collected near a water tank and salt lick area contained 4.7% nitrate (Williams 1989). A nitrate concentration of 1.5% or greater can be lethal to livestock (Kingsbury cited in Williams 1989).
Showy goldeneye seeds average 34.3% oil and 24.1% protein (RBG Kew 2018).
Ethnobotany
The Goshute used showy goldeneye seeds as a food source. The Navajo used it to feed sheep and deer and also considered it a witchcraft plant (Moerman 2003).
Horticulture
Showy goldeneye has considerable potential for use as a horticulture specimen plant (Hickman 1993). It produces abundant flowers (Fig. 6), is easily managed in landscape settings, and works well in mixed borders and meadow gardens. It is recommended for use in USDA Plants Hardiness Zones 4b to 10a (Tilley 2012).

Figure 6. Showy goldeneye flowers on plants growing near Lowman, ID. Photo: N. Shaw, USFS.
Greenhouse experiments showed that showy goldeneye is an obligate long-day plant. Plants grew but were small and failed to flower with only 8 hours of light. Plants grew larger and produced flowers when given 18 hours of light. Experiments were conducted from October 15 to April 12 using plants grown from seed (Mattson and Erwin 2005).
Revegetation Use
A tolerance of early seral conditions, ability to colonize disturbed sites rapidly, and persistence in late-seral communities make show goldeneye a potentially valuable revegetation species (USDA FS 1937; Ellison 1954; Stevens and Monsen 2004b). Showy goldeneye is also important to pollinators, especially bees (Pyke 1982; LBJWC 2018), and provides late growing-season forage for wildlife and livestock (Stevens 2004; Stevens and Monsen 2004a). Production of an abundance of attractive flowers makes showy goldeneye a good choice for use in revegetation of campgrounds, visitor centers, or other high-visibility recreation areas (Stevens and Monsen 2004a).
Developing A Seed Supply
For restoration to be successful, the right seed needs to be planted in the right place at the right time. Coordinated planning and cooperation is required among partners to first select appropriate species and seed sources and then properly collect, grow, certify, clean, store, and distribute seed for restoration (PCA 2015).
Developing a seed supply begins with seed collection from native stands. Collection sites are determined by current or projected revegetation requirements and goals. Production of nursery stock requires less seed than large-scale seeding operations, which may require establishment of agricultural seed production fields. Regardless of the size and complexity of any revegetation effort, seed certification is essential for tracking seed origin from collection through use (UCIA 2015).
Seed Sourcing
Because empirical seed zones are not currently available for showy goldeneye, generalized provisional seed zones developed by Bower et al. (2014), may be used to select and deploy seed sources. These provisional seed zones identify areas of climatic similarity with comparable winter minimum temperature and aridity (annual heat:moisture index). In Figure 7, Omernik Level III Ecoregions (Omernik 1987) overlay the provisional seeds zones to identify climatically similar but ecologically different areas. For site-specific disturbance regimes and restoration objectives, seed collection locations within a seed zone and ecoregion may be further limited by elevation, soil type, or other factors.
The Western Wildland Environmental Threat Assessment Center’s (USFS WWETAC 2017) Threat and Resource Mapping (TRM) Seed Zone application provides links to interactive mapping features useful for seed collection and deployment planning. The Seedlot Selection Tool (Howe et al. 2017) can also guide restoration planning, seed collection, and seed deployment, particularly when addressing climate change considerations.
Occurrence Map
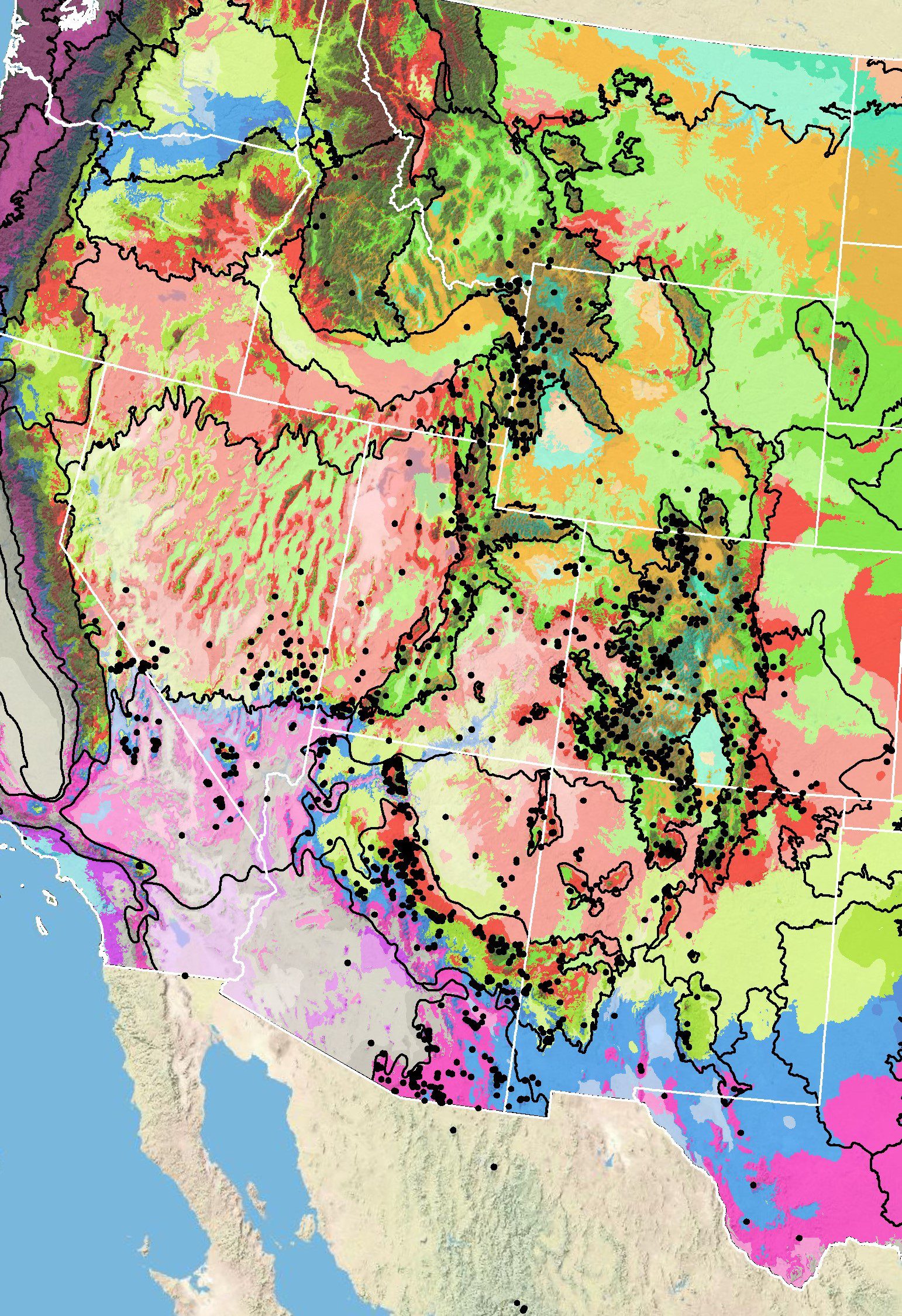
Figure 7. Distribution of showy goldeneye (black circles) based on geo-referenced herbarium specimens and observational data from 1895-2016 (CPNWH 2017; SEINet 2017; USGS 2017). Generalized provisional seed zones (colored regions) (Bower et al. 2014) are overlain by Omernik Level III Ecoregions (black outlines) (Omernik 1987; USDI EPA 2018). Interactive maps, legends, and a mobile app are available (USFS WWETAC 2017; research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap). Map prepared by M. Fisk, USGS.
Releases
As of 2018, there were no germplasm releases of showy goldeneye.
Wildland Seed Collection
Showy goldeneye seed is generally collected from wildland stands from July through September, but this can vary by year and site conditions (USDI BLM SOS 2017). Several collection guidelines or methods can be used as rules of thumb to maximize the genetic diversity of wildland collections: collect seed from a minimum of 50 randomly selected plants; collect from widely separated individuals throughout a population without avoiding habitat edges, microsites, or selecting the most robust individuals; make seed collections at peak seed maturity, and as possible, make collections at multiple dates to gather early to late-flowering plants (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g. ENSCONET 2009; USDI BLM SOS 2017). As is the case with wildland collection of many forbs, care must be taken to avoid inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of showy goldeneye.
Wildland Seed Certification
Verification of species and tracking of geographic source is necessary whether wildland seed is collected for immediate project use or as stock seed for cultivated increase. This official Source Identification process can be accomplished by following procedures established by the Association of Official Seed Certifying Agencies (AOSCA) Pre-Variety Germplasm Program (Young et al. 2020; UCIA 2015). Wildland seed collectors should become acquainted with state certification agency procedures, regulations, and deadlines in the states where they collect.
If wildland-collected seed is to be sold for direct use in ecological restoration projects, collectors must apply for Source-Identified certification prior to making collections. Pre-collection applications, site inspections, and species and seed amount verification are handled by the AOSCA member state agency where seed collections will be made (see listings at AOSCA.org).
If wildland seed collected by a grower or private collector is to be used as stock seed for planting cultivated seed fields or for nursery propagation (See Agricultural Seed Field Certification section), detailed information regarding collection site and collecting procedures must be provided when applying for certification. Photos and herbarium specimens may be required. Germplasm accessions acquired within established protocols of recognized public agencies, however, are normally eligible to enter the certification process as stock seed without routine certification agency site inspections. For contract grow-outs, however, this collection site information must be provided to the grower to enable certification.
Collection Timing
Most wildland seed collections of showy goldeneye made by BLM Seeds of Success (SOS) field crews (Fig. 8) were harvested in July and September but collection dates ranged from May to November throughout their five-state collection area (AZ, CO, NV, NM, UT) (USDI BLM SOS 2017). Other authors indicate that seeds are generally mature in August and September (Plummer et al. 1968; Jorgensen and Stevens 2004a).
Based on 51 SOS collections made over 9 years across an elevation range of 4,300 to 10,240 ft (1,310-3,120 m), collections were generally made later at higher elevations (USDI BLM SOS 2017). The earliest SOS collection date was May 19 from 4,440 ft (1,350 m) in Lincoln County, Nevada. The latest collection date was November 11 from 6,190 ft (1,890 m) in Doña Ana County, New Mexico. In the year with the greatest number of collections (18 in 2010), the earliest collection date was June 21 from 4,950 ft (1,510 m) in Lincoln County, Nevada, and the latest date was October 19 from 7,290 ft (2,220 m) in Coconino County, Arizona (USDI BLM SOS 2017).
Visual assessments of stands are useful to determine when seed is ripe. Harvests should occur when the majority of flowers have dry, yellow petals that have started to fall. At this time there will be a small number of fresh flowers, but most seed will be mature (Stevens et al. 1996).

Figure 8. Showy goldeneye seed collected by BLM SOS on the Colorado Plateau. Photo: USDI BLM CPI126 SOS.
Collection Methods
Wildland seed can be collected by hand stripping the seed heads (Plummer et al. cited in Redente et al. 1982), shaking or beating seed heads into a container, or clipping entire seed heads (Jorgensen and Stevens 2004a; Tilley 2016).
Several collection guidelines and methods should be followed to maximize the genetic diversity of wildland collections: 1) collect seed from a minimum of 50 randomly selected plants; 2) collect from widely separated individuals throughout a population without favoring the most robust or avoiding small stature plants; and 3) collect from all microsites including habitat edges (Basey et al. 2015). General collecting recommendations and guidelines are provided in online manuals (e.g., ENSCONET 2009; USDI BLM SOS 2021).
It is critical that wildland seed collection does not impact the sustainability of native plant populations. Collectors should take no more than 20% of the viable seed available at the time of harvest (USDI BLM SOS 2021). Additionally, care must be taken to avoid the inadvertent collection of weedy species, particularly those that produce seeds similar in shape and size to those of showy goldeneye.
Collection Rates
Based on eight collections made in Yellowstone or Glacier National Park, the average collection rate for showy goldeneye bulk (uncleaned) seed was 0.3 lb (126 g)/person hour (range: 0.01 to 0.7 lb [28-332 g]/person hr) (Majerus 1999).
Post-Collection Management
Seed collections made late in the growing season may have high moisture content. Properly and thoroughly drying this seed is critical for successful storage and cleaning. Harvested seed should be spread on racks in a protected area and thoroughly air dried prior to cleaning or transport. Little drying will likely be necessary for summer-collected seed. Drying importance increases with the amount of inert material collected with the seed and when collections are made during times of high humidity. At the Aberdeen, Idaho, Plant Materials Center (IDPMC), seed was dried on butcher paper on the shop floor before cleaning (Tilley 2016).
Seed Cleaning
Showy goldeneye seed is difficult to clean to high purity (Shaw and Monsen 1983). Cleaning procedures generally involve processing through a barley debearder, then an air-screen separator, followed by gravity separation (Stevens et al. 1996; Jorgensen and Stevens 2004a). At the IDPMC, seeds were separated from seed heads by hand or using a hammermill or brush machine. Seeds were then processed using an air-screen cleaner with a 6 × 20 top screen, 6 × 34 bottom screen, and light air (Tilley 2016).
Seed Storage
Showy goldeneye seed is orthodox (RBG Kew 2018), and no significant loss in viability should occur with 2 to 6 years of storage in a dry location (Plummer et al. 1968; Shaw and Monsen 1983; Stevens et al. 1996; Jorgensen and Stevens 2004a).
Seed can be stored in a dry location. No other special conditions are generally required (Stevens et al. 1996). At the IDPMC, clean dry seed was stored at about 50º F (10 ºC) and relative humidity of 20 to 30% (Tilley 2016). Germination of showy goldeneye seed was significantly (P < 0.05) reduced after 10 years of storage in an open warehouse in Utah. Germination was 13 to 18% after 2 to 7 years and 1% after 10 years of storage in the warehouse, which experienced extreme high and low temperature fluctuations. Germination was not tested prior to storage. Following 2 to 10 years of storage it was tested by placing seeds between moist newspaper in plastic and incubating at 34 to 38 ºF (1–3 ºC) in a refrigerator (Stevens et al. 1981a; Jorgensen and Stevens 2004b).
Seed Testing
The Association of Official Seed Analysts (AOSA) guidelines for germination testing recommend prechilling seed at 36 to 41 ºF (2–5 ºC) for 28 days, followed by incubation at 41 and 68 ºF (5–20 ºC). First seed counts should occur after 10 days and last counts after 28 days (AOSA 2016). Gibberellic acid is recommended for dormant seed (B.A. Atkins Seed Testing, personal communication, no date). There is no AOSA seed testing rule for showy goldeneye viability.
Germination Biology
Optimal germination conditions may differ by seed lot. Afterripening is necessary for maximum germination. Recommended afterripening periods vary from 1 to 12 months and may result in only moderate germination. Stevens et al. (1996) indicated that maximum germination was not reached until a month after harvesting, and seed then required a month of cool stratification (Jorgensen and Stevens 2004a). Germination was mostly even and rapid for mature seed, although germination of some lots was erratic (Stevens et al. 1996). The Lady Bird Johnson Wildflower Center (2018) suggested showy goldeneye required a long afterrippening period (10-12 months) and then germinated at moderate percentages with cool incubation temperatures. Karrfalt and Vankus (2011) indicated seed was very dormant and required 8 weeks of cool chilling and then germinated best at 50 °F (10 °C).
Warm temperatures following afterrippening and cool stratification appear to increase germination potential. Emergence was 30 to 38% for seeds stored at 23 ºF (-5 ºC) for 4 months, then sown into soil and kept moist in a 75 °F (24 °C) greenhouse with 14 hours of light. Emergence was not significantly different when seeds were treated with a 10% aqueous smoke solution (Abella 2009).
Germination of showy goldeneye varied by seed source in a study of relationships between source climate, species identity, and germination to improve seed sourcing decision making in restoration (Table 2; Seglias et al. 2018). There was dramatic variation between populations and close relationships to source site climate for showy goldeneye seed. Seed was collected from three locations: Ouray, Colorado, with the lowest average annual temperature (48 °F [9.1 °C]) and moderate annual precipitation (17 in [422 mm]); south of Flagstaff, Arizona, with a moderate average annual temperature (50.7 °F [10.4 °C]) and highest annual precipitation (22 in [556 mm]); and Organ, New Mexico with the highest average annual temperature (54.3 °F [12.4 °C]) and lowest annual precipitation (14 in [363 mm]). Viability of the seed lots from the three locations was 90 to 96%. Seed was afterrippened for 2 weeks at room temperature, then dried and stored at 59 °F (15 °C) and 15% relative humidity until stratification treatments designed to mimic long winter, spring germination; short winter, spring germination; and short summer monsoon and fall germination conditions were applied. Stratified seeds were then incubated at simulated spring, summer, or fall germination temperatures (Table 2; Seglias et al. 2018).
Table 2. Percentage germination of viable showy goldeneye seed collected from south of Flagstaff, AZ, central CO, and southern NM. exposed to various simulated winter, spring, and summer conditions. Each treatment included 4 replicates of 25 seeds each. No statistical analysis was conducted (Seglias et al. 2018).
|
Simulated conditions: |
Control |
Long winter, spring germ |
Short winter, spring germ |
Short summer monsoon, fall germ |
||||
|
Stratification (time, temp): |
none |
8 wks, 37 °F |
3 wks, 37 °F |
3 wks, 86 °F |
||||
|
Incubation*: |
cool |
warm |
cool |
warm |
cool |
warm |
cool |
warm |
|
Seed source |
————————————————–% germination————————————————- |
|||||||
|
Flagstaff, AZ |
11 |
42 |
16 |
48 |
9 |
65 |
22 |
61 |
|
Ouray, CO |
25 |
16 |
93 |
94 |
80 |
76 |
2 |
2 |
|
Organ, NM |
92 |
99 |
98 |
99 |
87 |
92 |
87 |
96 |
*Cool (spring/fall) incubation was 68/50 °F and warm (summer) incubation was 77/59 °F with 12 hr dark/12 hr light for both.
Wildland Seed Yield And Quality
Post-cleaning seed yield and quality of showy goldeneye seed lots collected in the Intermountain region are provided in Table 3 (USFS BSE 2017). The results indicate that showy goldeneye seed can be cleaned to high levels of purity and seed fill, but viability of fresh seed is variable. Seeds are small, averaging 0.56 mg (Abella 2009) but can vary widely from about 470,000 to more than 1,550,000 seeds/lb (1,040,000–3,400,000 seeds/kg) (Table 3; Plummer et al. 1968; Jorgensen and Stevens 2004a; Lambert 2005; Tilley 2016; RBG Kew 2018).
Average viability of seed lots received and tested from 2009 to 2016 by Oregon State University’s Malheur Experiment Station ranged from 76 to 94% (Shock et al. 2011, 2012, 2014a, 2015a, 2016, 2017a), and were nearly within the range reported in Table 3.
Table 3. Seed yield and quality of showy goldeneye seed lots collected in the Intermountain region, cleaned by the Bend Seed Extractory, and tested by the Oregon State Seed Laboratory or the USFS National Seed Laboratory (USFS BSE 2017).
|
Seed lot characteristic |
Mean |
Range |
Samples (no.) |
|
Bulk weight (lbs) |
2.37 |
0.17–42.5 |
54 |
|
Clean weight (lbs) |
0.24 |
0.012–1.77 |
54 |
|
Clean-out ratio |
0.18 |
0.02–0.85 |
54 |
|
Purity (%) |
96 |
68–99 |
55 |
|
Fill (%)¹ |
93 |
82–99 |
55 |
|
Viability (%)² |
95 |
78–98 |
47 |
|
Seeds/lb |
873,610 |
477,473–1,553,424 |
55 |
|
Pure live seeds/lb |
776,093 |
458,565-1,300,713 |
47 |
¹100 seed X-ray test
²Tetrazolium chloride test
Marketing Standards
Recommendations for acceptable purity of seed collected from wildlands and grown in seed fields varies from 50 to 95% (Plummer et al. 1968; Shaw and Monsen 1983; Stevens et al. 1996; Jorgensen and Stevens 2004a).
Acceptable seed purity, viability, and germination specifications vary with revegetation plans. Purity needs are highest for precision seeding equipment like that used in nurseries, while some rangeland seeding equipment handles less clean seed well.
Agricultural Seed Production
Showy goldeneye has been grown successfully by the Bridger, Montana, Plant Materials Center (Majerus 1999), the OSU MES, and IDPMC. At OSU MES, plants produced harvestable crops in the first year (Shock et al. 2015b, 2016). At IDPMC, 35 to 60% of plants produced seed in the establishment year, and in years 2 to 7, plants produced 150 to 200 lbs of seed/ac (169–225 kg/ha). Seeds were cleaned to 40 to 50% (Stevens et al. 1996).
Agricultural Seed Certification
In order to minimize genetic changes in specific accessions of native species when increased in cultivated fields, it is essential to track the geographic source and prevent inadvertent hybridization or selection pressure. This is accomplished by following third party seed certification protocols for Pre-Variety Germplasm (PVG) as established by the Association of Official Seed Certification Agencies (AOSCA). AOSCA members in the U.S., Canada, and other countries administer PVG requirements and standards that track the source and generation of planting stock. Field and cleaning facility inspections then monitor stand establishment, proper isolation distances, control of prohibited weeds, seed harvesting, cleaning, sampling, testing, and labeling for commercial sales (Young et al. 2020; UCIA 2015).
Seed growers apply for certification of their production fields prior to planting and plant only certified stock seed of an allowed generation (usually less than four). The systematic and sequential tracking through the certification process requires preplanning, knowing state regulations and deadlines, and is most smoothly navigated by working closely with state certification agency personnel. See the Wildland Seed Certification section for more information on stock seed sourcing.
Site Preparation
Seeding in weed-free fields is recommended (Stevens et al. 1996). At IDPMC, seed production stands were established in disked, packed, and weed barrier covered beds. Three-in (8 cm) diameter holes were made in the fabric at 18-in (46 cm) row spacings for seed sowing (Tilley 2016).
Seed Pretreatments
No pretreatment is needed for fall seedings. At IDPMC, dormant fall seeding was done in late October through mid-November (Tilley 2016). At OSU MES, seed pretreated with a liquid mix of fungicides produced poorer stands than untreated seed (Shock 2016).
Weed Management
Cultivation and pre-emergent or selective herbicides can be used to control weeds in seed production fields (Stevens et al. 1996). At IDPMC, broadleaf weeds were removed by hand, and annual grasses were controlled with selective herbicides (Tilley 2016).
Post-emergent herbicides can be harmful to seed production. When ammonium salt of imazapic was applied (5 oz/ac) to dormant showy goldeneye plants at the USFS Shrub Sciences Laboratory in Provo, Utah, seed production was eliminated for the following year (Jensen 2012). When post-emergent herbicides were tested on young plants at OSU MES, injury ranged from 1 to 11% and was not significant. Herbicides applications occurred in April or May 2014 on fields seeded in fall 2013. Injury was least following applications of a combination of an ammonium salt of imazamox, sodium salt of bentazon, and pendimethalin (Shock 2015b). In other post-emergent herbicide treatments, though stand losses were 33 to 41% after treatments of octanoic acid ester of bromoxynil or carfentrazone-ethyl. Treatments occurred on April 26 on plots seeded the previous fall (Shock 2014b).
Seeding
For establishment of seed production stands, seeding 20 to 40 pure live seeds (PLS)/linear ft of row (70-130 PLS/m) at 0.13 to 0.25 in (0.3–0.6 cm) depths in 28- to 36-in row spacings is recommended. Production is best with fall seeding on sandy loam to clay loam soils. Stevens et al. (1996) found a single-row seeder (Planet Junior ™) was best to maintain seeding depth, which is difficult to meter for showy goldeneye. At IDPMC about 20 showy goldeneye seeds were placed in 3-in diameter (7.6 cm) holes in weed barrier fabric. Seeds were scratched into the soil and packed for good seed-soil contact (Tilley 2016).
At OSU MES, duration of flowering increased with increased irrigation, and wetter plots flowered much longer than drier plots, making multiple harvests necessary in 2 of 4 years on the wetter plots. Seed production plots were seeded in the fall of 2012 in silt loam soils with an average pH of 8.2. Seeding was done in 30-in (76 cm) rows using a custom grain drill. Seed was deposited on the soil surface, then covered with sawdust and row cover. In March, the row cover was removed and bird netting was used to protect seedlings until early May when it was removed (Shock et al. 2017b).
Use of row cover to protect seedings increased stand establishment at OSU MES. Stand emergence on April 9, 2010 for seeds planted on November 12, 2009 ranged from 10.9% to 39.2%. Emergence was lowest for seeds covered with sawdust but not protected by row cover and was highest for seeds covered by sawdust and protected by row cover. Seed coverings did not significantly affect seed yield which ranged from 769 to 966 lbs/ac (865-1,087 kg/ha) but was greatest for seeds protected by row cover and least for seeds not protected by row cover. Seed was hand harvested in August and again in November (Shock et al. 2011). When stand establishment was evaluated for seedings done in 2013, 2015 and 2016, percent stand establishment was much greater for plots with row cover (3-year average of 23-34%) than for plots without row cover (2.4–5.4%). Row cover protected the soil from desiccation and seeds from bird predation. Although other seed coverings such as sawdust and sand were also tested, row cover with or without these additional treatments resulted in the best stand establishment (Shock et al. 2014a; 2016; 2017a).
Establishment And Growth
For showy goldeneye seed production stands at the IDPMC, the establishment phase was 1 month, active growing phase was 5 months, and seed was produced 11 months after late fall seeding (Tilley 2016). Although stands can support large insect populations, control measures are not usually necessary (Stevens et al. 1996). Annual between-row cultivation was suggested to maintain stand density (Stevens et al. 1996).
Irrigation
Supplemental irrigation increased seed production of showy goldeneye fields at OSU MES (Table 4; Shock et al. 2017b). At IDPMC, fields were watered using drip tape for about 4 hours/week during the establishment phase of about 1 month. Irrigation after that was for about 8 to 12 hours/month or as needed (Tilley 2016). Other researchers suggest as needed irrigation for establishment and delivery of 15 to 20 in (381–508 mm) of annual precipitation and irrigation, which may require up to four irrigations in the spring and at the pre-flower, mid-flower, and late-flower stages (Stevens et al. 1996).
Table 4. Seed yield (lbs/ac) for showy goldeneye stands with and without growing season irrigation applications totaling 0, 4, or 8 in at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2017b).
| Year | 0 inch | 4 inches | 8 inches |
| Seed yield (lbs/ac) | |||
| 2013 | 28.7 | 57.6 | 96.6 |
| 2014 | 154.6 | 200.9 | 271.7 |
| 2015 | 81.7 | 115.6 | 188.2 |
| 2016 | 92.3 | 89.2 | 98.0 |
| Average | 89.3 | 114.1 | 163.7 |
* Treatments significantly different (P < 0.1).
Seed yield increased with increased irrigation rates up to the highest level tested (8 in [20 cm]) in 3 of 4 years of testing at OSU MES (Shock et al. 2017b). Duration of flowering increased with increased irrigation, and wetter plots flowered much longer than drier plots, making multiple harvests necessary in 2 of 4 years. Stands were irrigated periodically in May and June through a buried drip-tape system. Accumulation of growing degree days (50–86 ºF [10–30 ºC) from January to June was above average for all years 2013 through 2016 and January to June precipitation was below the 5-year average (5.6 in [142 mm) for all years (Shock et al. 2017b).
Pollinator Management
Good seed production requires flower pollination, and bees are the common flower visitors. It is possible that seed production may benefit from providing nests or hives of manageable bees like the cavity-nesting Asteraceae specialist (Osmia californica) or the European honey bee (Apis mellifera) (Cane et al. 2012). If introducing new bee populations is not feasible, management practices to encourage and sustain native bee populations may benefit showy goldeneye seed production (Cane 2008).
Pest Management
Showy goldeneye is a host for many fungi species including Coleosporium viguierae, Comoclathris compressa, Leveillula picridis, Puccinia abrupta, P. aemulans, P. enceliae, and Ramularia helianthi (Mohan and Shock 2014; Farr and Rossmann 2017). It is also a host for aphids (Uroleucon ambrosiae) (Funk and Bernays 2001), plant bugs (Hadronema pictum) (Forero 2008), and Gorgone checkerspot (Chlosyne gorgone) butterflies (Wahlberg 2001).
The degree to which the above pests are or could be troublesome in nursery production or seed fields was not reported.
Seed Harvesting
Although showy goldeneye seed is produced in the first growing season, yields are better in years 2, 3, and beyond (Tilley 2016). Indeterminate flowering and seed maturation makes multiple harvests necessary for maximum yields (Tilley 2016; Shock et al. 2017b), which were shown to increase with irrigation at OSU MES (See Irrigation section above; Shock et al. 2017b). Both hand and mechanical harvesting methods have been used for seed production fields (Stevens et al. 1996; Tilley 2016; Shock et al. 2017b).
Harvesting of seed production stands in Utah occurred between August 20 and September 10, when a small amount of flowering was still occurring but the majority of yellow petals were dry and starting to fall. Seed retention was considered fair, and seeds were harvested using a combine or reel-type harvester (Stevens et al. 1996). Harvest dates at OSU MES ranged from early July to late August (Table 5; Shock et al. 2017b).
Table 5. Timing of flowering and seed harvest dates for seed production fields growing for 4 years at Oregon State University’s Malheur Experiment Station in Ontario, OR (Shock et al. 2017b). Seed yields areprovided in Table 4 above.
|
Year |
Start flowering |
Peak flowering |
End flowering |
Harvest |
|
2013 |
July 15 |
—- |
Aug 30 |
8/8, 8/15, 8/28 |
|
2014 |
May 20 |
June 20 |
Aug 30 |
Weekly 7/15-8/15 |
|
2015 |
May 5 |
May 26 |
July 10 |
7/13 |
|
2016 |
May 5 |
June 15 |
Sept 30 |
7/8 |
Seed Yields And Stand Life
In the establishment year, 35 to 60% percent of the mature seed crop is produced (Stevens et al. 1996), but after this and for up to 7 years, full seed yields of 50 to 250 lbs/ac (60–280 kg/ha) can be produced (Stevens et al. 1996; Shock et al. 2015). Yields reported by Stevens et al. (1996) were for purity of 40 to 50%.
At IDPMC, showy goldeneye produced some seed in the first growing season, but yields were best in years 2, 3, and beyond. Seed from 2- or 3-year-old plants was harvested three times in a year. Seed was swatted from the flower heads into canvas harvest hoops with tennis racquets. The yield at the first harvest date of September 17 was 0.5 lb (240 g), on September 26 was 1.8 lbs (800 g), and on October 6 was 1.8 lbs (800 g) for a total yield of 4 lbs (1,800 g) or about 57 lbs of clean seed/ac (64 kg/ha) (Tilley 2016).
Nursery Practice
Showy goldeneye is described as easily transplanted (Plummer et al. 1968) but protocols for growing nursery stock are not available. At the IDPMC, transplants in Jiffy Pellets (Jiffy Products, Lorain, OH) were used effectively to establish seed production fields (Tilley 2016).
Wildland Seeding And Planting
Showy goldeneye is attractive to pollinators (LBJWC 2018) and wildlife (Stevens 2004) and functions as a pioneer, early seral, and late seral species (Stevens and Monsen 2004b) making it desirable for restoration planting (See earlier Pollination, Ecology, and Wildlife and Livestock Use Sections). Showy goldeneye has been rated excellent for natural spread and very good or good for germination, initial and final establishment, growth rate, seed production, compatibility with other species, ease of transplanting, and early spring and summer palatability (Plummer et al. 1968; Stevens and Monsen 2004a). Soil stabilization potential is considered moderate (Shaw and Monsen 1983). Showy goldeneye has potential for use in subalpine, quaking aspen, ponderosa pine, mountain brush, pinyon-juniper, mountain big sagebrush, and basin big sagebrush restoration. Variety nevadensis may be used at dry Wyoming big sagebrush or salt desert shrub sites ( Stevens and Monsen 2004a). Showy goldeneye is part of a seed mix used to revegetate disturbed areas in lodgepole pine forests in Yellowstone National Park (Majerus 1999).
Stevens and Monsen (2004b) report that showy goldeneye establishes well and is capable of competing with other species when seeded at appropriate rates. In wildland settings, it typically takes 2 to 3 years to establish, mature, and flower (Stevens and Monsen 2004b).
Wildland seeding. Fall or winter seeding is recommended, and seed should not be planted more than 0.25 in (0.6 cm) deep (Table 6; Monsen and Stevens 2004; Stevens and Monsen 2004a; Lambert 2005). Showy goldeneye can be broadcast aerially or seeded with a cultipacker or drill as part of a mix (Shaw and Monsen 1983; Monsen and Stevens 2004; Stevens 2004).
Table 6. Showy goldeneye seeding rates required to meet desired target densities for drill and broadcast seeding (Monsen and Stevens 2004).
|
Requires |
0.04 lb PLS/ac |
0.2 lb PLS/ac |
0.4 lb PLS/ac |
|
To drill seed |
1 seed/linear ft |
5 seeds/linear ft |
10 seeds/linear ft |
|
To broadcast seed |
9 seeds/ft² |
45 seeds/ft² |
90 seeds/ft² |
Studies indicate variable success with interseeding showy goldeneye into existing grass stands. As a seedling, showy goldeneye is considered moderately competitive when grass competition is at a maximum level and extremely competitive when grass competition is minimal. Showy goldeneye establishment success is impacted by the degree and duration of competition removal and composition of interseeding species mixes (Stevens and Monsen 2004b). Stands of showy goldeneye were rated 3.8 on a 0-to-10 scale where 10 indicated a full stand about 3 years after it was seeded as part of a mix on degraded terraced rangelands in Davis County, Utah. Seed was broadcast and then covered on the site. In visits to this site about 30 years after seeding, showy goldeneye stands were rated as zero (Hull 1973).
Showy goldeneye was successfully interseeded into stands of intermediate wheatgrass (Agropyron intermedium) in Holden, Utah. Scalping equipment was used to scarify 30-in (76 cm) wide and 9-in (23 cm) deep rows, where seed was deposited and then covered by a dragging chain in October or November. After 2 years, sites had more than 5 showy goldeneye plants/linear ft (Stevens et al. 1981b).
Showy goldeneye established on one of two weedy openings within the spruce-fir zone in northern Utah (Hull 1974). Seed was drill seeded into firm weed-free seed beds in spring or fall at an average depth of 0.5 in (1.3 cm), which is deeper than currently recommended. Showy goldeneye established at the seeding site in northern Utah but not at a similar site in western Wyoming. In the establishment area, stands were rated very poor 18 years after seeding (Hull 1974).
Establishment of showy goldeneye was good with or without the addition of arbuscular mycorrhiza (AM) propagules on slash burns in ponderosa pine forests in Arizona’s Coconino National Forest. Seed was planted in July 2000, five months after slash was burned. At the end of the 2000 growing season, showy goldeneye averaged 0.01% cover and 2 mm plant height on seeded only plots and 0.04% cover and 3 mm plant height on seeded plots where AM propagules were added. In 2001, showy goldeneye was the second most abundant flowering species. Cover averaged 0.5% and plant height averaged 1.9 in (4.7 cm) on seed only, and cover averaged 0.4% and plant height averaged 2 in (4.9 cm) on seed with AM plots (Korb 2001).
Wildland planting. Use of container or bareroot stock and its success in wildland restoration was not reported in the literature, but Stevens (1994) predicted better establishment success from container-grown stock than from bareroot or wilding stock (Stevens 1994).
Stand management. Restoration sites should be protected from grazing for at least 2 years after seeding showy goldeneye. Once resumed, grazing should be managed to allow established stands to produce seed every 3 to 4 years (Stevens and Monsen 2004a).
Acknowledgements
Funding for Western Forbs: Biology, Ecology, and Use in Restoration was provided by the USDI BLM Great Basin Native Plant Materials Ecoregional Program through the Great Basin Fire Science Exchange. Great thanks to the chapter reviewers: Stan Young, Utah State University and Tara de Queiroz, University of Nevada, Reno.
This research was supported in part by the USDA Forest Service, Rocky Mountain Research Station. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy.
Literature Cited
Abella, S.R. 2009. Smoke-cued emergence in plant species of ponderosa pine forests. Fire Ecology. 5(1): 22-37.
Abella, S.R.; Springer, J.D. 2006. Canopy-tree influences along a soil parent material gradient in Pinus ponderosa–Quercus gambelii forests, northern Arizona. Journal of the Torrey Botanical Society. 135(1): 26-36.
Aldous, C.M. 1951. The feeding habits of pocket gophers (Thomomys talpoides Moorei) in the high mountain ranges of central Utah. Journal of Mammalogy. 32(1): 84-87.
Andersen, B.A.; Holmgren, A.H. 1976. Mountain plants of northeastern Utah. Circular No. 319. Logan, UT: Utah State University Extension Services. 148 p.
Association of Official Seed Analysts [AOSA]. 2016. AOSA rules for testing seeds. Vol. 1. Principles and procedures. Washington, DC: Association of Official Seed Analysts.
Barsch, B.K. 1977. Distribution of the Coues deer in pinyon stands after a wildfire. Tucson, AZ: University of Arizona. Thesis. 52 p.
Basey, A.C.; Fant, J.B.; Kramer, A.T. 2015. Producing native plant materials for restoration: 10 rules to collect and maintain genetic diversity. Native Plants Journal. 16(1): 37-53.
Bower, A.D.; St. Clair, J.B.; Erickson, V. 2014. Generalized provisional seed zones for native plants. Ecological Applications. 24(5): 913-919.
Bowns, J.E.; Bagley, C.F. 1986. Vegetation responses to long-term sheep grazing on mountain ranges. Journal of Range Management. 39(5): 431-434.
Cane, J.H. 2008. Pollinating bees crucial to farming wildflower seed for U.S. habitat restoration. In: James, R.; Pitts-Singer, T., eds. Bees in agricultural ecosystems. Oxford, UK: Oxford University Press: 48-64.
Cane, J.H.; Love, B.; Swoboda, K. 2012. Breeding biology and bee guild of Douglas’ dustymaiden, Chaenactis douglasii (Asteraceae, Helenieae). Western North American Naturalist. 72(4): 563-568.
Clary, W.P.; Ffolliott, P.F.; Jameson, D.A. 1968. Relationship of different forest floor layers to herbage production. Res. Note RM-123. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Forest and Range Experiment Station. 3 p.
Cockerell, T.D.A. 1919. The bees of Peaceful Valley, Colorado. Journal of the New York Entomological Society. 27(4): 298-300.
Consortium of Pacific Northwest Herbaria [CPNWH]. 2017. Seattle, WA: University of Washington Herbarium, Burke Museum of Natural History and Culture. pnwherbaria.org/
Cook, C.W. 1983. “Forbs” need proper ecological recognition. Rangelands. 5(5): 217-220.
Costello, D.F. 1944. Important species of the major forage types in Colorado and Wyoming. Ecological Monographs. 14(1): 107-134.
Craighead, J.J.; Craighead, F.C. Jr.; Davis, R.J. 1963. A field guide to Rocky Mountain wildflowers from northern Arizona and New Mexico to British Columbia. Boston, MA: Houghton Mifflin Company. 277 p.
Cronquist, A. 1994. Volume five: Asterales. In: Cronquist, A.; Holmgren, A.H.; Holmgren, N.H.; Reveal, J.L.; Holmgren, P.K., eds. Intermountain flora: Vascular plants of the Intermountain West, U.S.A. Bronx, NY: The New York Botanic Garden. 496 p.
Eastmond, R.J. 1968. Vegetational changes in a mountain brush community of Utah during eighteen years. Provo, UT: Brigham Young University. Thesis. 261 p.
Ellison, L. 1954. Subalpine vegetation of the Wasatch Plateau, Utah. Ecological Monographs. 24(2): 89-184.
European Native Seed Conservation Network [ENSCONET]. 2009. ENSCONET seed collecting manual for wild species. Edition 1: 32 p.
Farr, D.F.; Rossman, A.Y. 2017. Fungal databases, U.S. National Fungus Collections. U.S. Department of Agriculture, Agricultural Research Service. https://nt.ars-grin.gov/fungaldatabases/.
Forero, D. 2008. Revision and phylogenetic analysis of the Hadronema group (Miridae: Orthotylinae: Orthotylini), with descriptions of new genera and new species, and comments on the neotropical genus Tupimiris. Bulletin of the American Museum of Natural History. 312: 1-172.
Funk, D.J.; Bernays, E.A. 2001. Geographic variation in host specificity reveals host range evolution in Uroleucon ambrosiae aphids. Ecology. 82(3): 726-739.
Haskins, K.E.; Gehring, C.A. 2004. Long-term effects of burning slash on plant communities and arbuscular mycorrhizae in a semi-arid woodland. Journal of Applied Ecology. 41(2): 379-388.
Hickman, J.C., ed. 1993. The Jepson manual: Higher plants of California. Berkeley, CA: University of California Press. 1400 p.
Howe, G.; St, Clair, B.; Bachelet, D. 2017. Seedlot Selection Tool. Corvallis, OR: Conservation Biology Institute. https://seedlotselectiontool.org/sst/
Hull, A.C. 1973. Duration of seeded stands on terraced mountain lands, Davis County, Utah. Journal of Range Management. 26(2): 133-136.
Hull, A.C. 1974. Species for seeding mountain rangelands in southeastern Idaho, northeastern Utah, and western Wyoming. Journal of Range Management. 27(2): 150-153.
Jensen, S. 2012. Provisional seed zone-based seed increase. In: Shaw, N.; Pellant, M., eds. 2013. Great Basin Native Plant Selection and Increase Project: 2012 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station and U.S. Department of the Interior, Bureau of Land Management: 95-102.
Jorgensen, K.R.; Stevens, R. 2004a. Seed collection, cleaning, and storage. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 3. Gen. Tech. Rep. RMRS-GTR-136-vol-3. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 699-716.
Jorgensen, K.R.; Stevens, R. 2004b. Seed germination. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 3. Gen. Tech. Rep. RMRS-GTR-136-vol-3. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 723-732.
Karrfalt, R.; Vankus, V. 2011. Development of germination protocols, seed weight, purity, and seed conditioning/cleaning protocols for Great Basin grasses and forbs. In: Shaw, N.; Pellant, M., eds. 2011. Great Basin Native Plant Selection and Increase Project: 2010 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station and U.S. Department of the Interior, Bureau of Land Management: 36-39.
Keil, D.J., Pinkava, D.J. 1976. Chromosome counts and taxonomic notes for Compositae from the United States and Mexico. American Journal of Botany. 63(10): 1393-1403.
Klemmedson, J.O. 1956. Interrelations of vegetation, soils and range conditions induced by grazing. Journal of Range Management. 9(3): 134-138.
Korb, J.E. 2001. Understory plant community dynamics in southwestern ponderosa pine forest restoration. Flagstaff, AZ: Northern Arizona University. Thesis. 120 p.
Kunzler, L.M.; Harper, K.T.; Kunzler, D.B. 1981. Compositional similarity within the oakbrush type in central and northern Utah. The Great Basin Naturalist. 41(1): 147-153.
LaBerge, W.E. 1986. A revision of the bees of the genus Andrena of the Western Hemisphere. Part XII. Subgenera Leucandrena, Ptilandrena, Scoliandrena and Melandrena. Transactions of the American Entomological Society. 112(3): 191-248.
Lady Bird Johnson Wildflower Center [LBJWC]. 2018. Heliomeris multiflora (Pursh) Nutt. Native Plant Database. Austin, TX: Lady Bird Johnson Wildflower Center. https://www.wildflower.org/plants-main [Accessed 2018 January 3].
Lambert, S. 2005. Guidebook to the seeds of native and non-native grasses, forbs and shrubs of the Great Basin. Boise, ID: U.S. Department of the Interior, Bureau of Land Management, Idaho State Office. 136 p.
Larkin, L.L. 2004. Four new fall species of Andrena from the southwestern United States (Hymenoptera: Andrenidae). Journal of the Kansas Entomological Society. 77(3): 254-268.
Louda, S.M.; Dixon, P.M.; Huntly, N. 1987. Herbivory in sun versus shade at a natural meadow-woodland ecotone in the Rocky Mountains USA. Vegetatio. 72(3): 141-149.
Majerus, M. 1999. Collection and production of indigenous plant material for national park restoration. In: Holzworth, L.K.; Brown, R.W., comps. Revegetation with native species: Proceedings, 1997 Society for Ecological Restoration annual meeting; 1997 November 12-15; Fort Lauderdale, FL. Proc. RMRS-P-8. Ogden, UT: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 17-21.
Major, J.; Rejmanek, M. 1992. Amelanchier alnifolia vegetation in eastern Idaho, USA and its environmental relationships. Vegetatio. 98(2): 141-156.
Martinsen, G.D.; Cushman, J.H.; Whitham, T.G. 1990. Impact of pocket gopher disturbance on plant species diversity in a shortgrass prairie community. Oecologia. 83(1): 132-138.
Mason, L.R.; Andrews, H.M.; Carley, J.A.; Haacke, E.D. 1967. Vegetation and soils of No Man’s Land Mesa relict area, Utah. Rangeland Ecology and Management. 20(1): 45-49.
Mattson, N.S.; Erwin, J.E. 2005. The impact of photoperiod and irradiance on flowering of several herbaceous ornamentals. Scientia Horticulturae. 104(3): 275-292.
McKell, C.M. 1950. A study of plant succession in the oak brush (Quercus gambelii) zone after fire. Salt Lake City, UT: University of Utah. Thesis. 79 p.
Moerman, D. 2003. Native American ethnobotany: A database of foods, drugs, dyes, and fibers of Native American peoples, derived from plants. Dearborn, MI: University of Michigan. http://naeb.brit.org
Mohan, S.K.; Shock, C.C. 2014. Etiology, epidemiology, and management of diseases of native wildflower seed production. In: Kilkenny, F.; Shaw, N.L.; Gucker, C.L., eds. Great Basin Native Plant Project: 2013 Progress Report. Boise, ID: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 107-108.
Monsen, S.B. 2004. Controlling plant competition. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands. Gen. Tech. Rep. RMRS-GTR-136-vol-1. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 57-64.
Monsen, S.B.; Stevens, R. 2004. Seedbed preparation and seeding practices. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 1. Gen. Tech. Rep. RMRS-GTR-136-vol-1. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 121-154.
Munz, P.A.; Keck, D.D. 1973. A California flora and supplement. Berkeley, CA: University of California Press. 1905 p.
Neeley, H.L. 2012. A comparison of fire severity effects on post fire vegetation recovery nine years following the Rodeo-Chediski fire: A long term monitoring study. Tempe, AZ: Arizona State University. Thesis. 63 p.
Omernik, J.M. 1987. Ecoregions of the conterminous United States. Map (scale 1:7,500,000). Annals of the Association of American Geographers. 77(1): 118-125.
Pearson, D.E.; Callaway, R.M.; Maron, J.L. 2011. Biotic resistance via granivory: Establishment by invasive, naturalized, and native asters reflects generalist preference. Ecology. 92(9): 1748-1757.
Pleasants, J.M. 1980. Competition for bumblebee pollinators in Rocky Mountain plant communities. Ecology. 61(6): 1446-1459.
Plummer, A.P.; Christensen, D.R.; Monsen, S.B. 1968. Restoring big-game range in Utah. Pub. 68-3. Salt Lake City, UT: Utah Division of Fish and Game. 183 p.
Poreda, S.F.; Wullstein, L.H. 1994. Vegetation recovery following fire in an oakbrush vegetation mosaic. Great Basin Naturalist. 54(4): 380-383.
Pyke, G.H. 1982. Local geographic distributions of bumblebees near Crested Butte, Colorado: Competition and community structure. Ecology. 63(2): 555-573.
Rambo, J.L.; Faeth, S.H. 1999. Effect of vertebrate grazing on plant and insect community structure. Conservation Biology. 13(5): 1047-1054.
Redente, E.F.; Ogle, P.R.; Hargis, N.E. 1982. Growing Colorado plants from seed: A state of the art. Vol. III, forbs. FWS/OBS-82/30. Washington, D.C.: U.S. Department of the Interior, Fish and Wildlife Service. 141 p.
Reed, J.F. 1952. The vegetation of the Jackson Hole Wildlife Park, Wyoming. The American Midland Naturalist. 48(3): 700-729.
Rippel, P.; Pieper, R.D.; Lymbery, G.A. 1983. Vegetational evaluation of pinyon-juniper cabling in south-central New Mexico. Journal of Range Management. 36(1): 13-15.
Royal Botanic Gardens Kew [RBG Kew]. 2017. Seed Information Database (SID). Version 7.1. http://data.kew.org/sid/
Sabo, K.E.; Sieg, C.H.; Hart, S.C.; Bailey, J.D. 2009. The role of disturbance severity and canopy closure on standing crop of understory plant species in ponderosa pine stands in northern Arizona, USA. Forest Ecology and Management. 257(8): 1656-1662.
Schilling, E.E. 2006. 378. Heliomeris. In: Flora of North America Editorial Committee, ed. Flora of North America North of Mexico. Volume 21 Magnoliophyta: Asteridae, part 8: Asteraceae, part 3 Asterales, part 3 (Aster order). New York, NY: Oxford University Press: 169-172.
SEINet – Regional Networks of North American Herbaria Steering Committee [SEINet]. 2017. SEINet Regional Networks of North American Herbaria. https://symbiota.org/seinet
Sharif, C.M.; West, N.E. 1968. Forage moisture variations on mountain summer range. Journal of Range Management. 21(4): 228-235.
Shaw, N.; Monsen, S.B. 1983. Nonleguminous forbs for rangeland sites. In: Monsen, S.B; Shaw, N. Managing Intermountain rangelands-Improvement of range and wildlife habitats; 1981 September 15-17; Twin Falls, ID; 1982 June 22-24; Elko, NV. Gen. Tech. Rep. INT-157. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station: 123-131.
Shaw, R.J. 1958. Vascular plants of Grand Teton National Park. The American Midland Naturalist. 59(1): 146-166.
Shaw, R.J. 1995. Utah wildflowers: A field guide to northern and central mountains and valleys. Logan, UT: Utah State University Press. 218 p.
Shiflet, T.N. 1994. Rangeland cover types of the United States. Denver, CO: Society for Range Management. 152 p.
Shive, K.L.; Kuenzi, A.M.; Sieg, C.H.; Fule, P.Z. 2013. Pre-fire fuel reduction treatments influence plant communities and exotic species 9 years after a large wildfire. Applied Vegetation Science. 16(3): 457-469.
Shock, C.; Feibert, E.; Parris, C.; Saunders, L. 2012. Direct surface seeding strategies for establishment of Intermountain West native plants for seed production. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2011. OSU AES Ext/CrS141. Corvallis, OR: Oregon State University: 130-135.
Shock, C.C.; Feibert, E.B.G.; Parris, C.; Saunders, L.D. 2011. Operational success of direct surface seeding strategies for establishment of Intermountain West native plants. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2010. OSU AES Ext/CrS132. Corvallis, OR: Oregon State University: 179-184.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Sanders, L.D.; Kilkenny, F.; Shaw, N. 2017a. Direct surface seeding systems for the establishment of native plants in 2016. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2016. OSU AES Ext/CRS 157. Corvallis, OR: Oregon State University: 123-130.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D. 2015a. Direct surface seeding strategies for emergence of native plants in 2014. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2014. OSU AES Ext/CrS152. Corvallis, OR: Oregon State University: 275-279.
Shock, C.C.; Feibert, E.B.G.; Rivera, A.; Saunders, L.D. 2017b. Irrigation requirements for seed production of several native wildflower species planted in the fall of 2012. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2016. OSU AES Ext/CrS157. Corvallis, OR: Oregon State University: 131-140.
Shock, C.C.; Feibert, E.B.G.; Saunders, L.D. 2014a. Direct surface seeding strategies for successful establishment of native wildflowers. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2013. OSU AES Ext/CrS149. Corvallis, OR: Oregon State University: 159-165.
Shock, C.C.; Feibert, E.B.G.; Saunders, L.D. 2014b. Tolerance of native wildflower seedlings to preemergence and postemergence herbicides. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2013. OSU AES Ext/CrS149. Corvallis, OR: Oregon State University: 198-206.
Shock, C.C.; Ishida, J.; Feibert, E.B.G.; Rivera, A. 2015b. Forb response to postemergence herbicides. In: Shock, C.C., ed. Malheur Experiment Station Annual Report 2014. OSU AES Ext/CrS152. Corvallis, OR: Oregon State University: 280-282.
Silva, S.; Ayers, T. 2016. Plant endemism on Mancos Shale barrens. Natural Areas Journal 36(2): 166-176.
Solbrig, O.T.; Kyhos, D.W.; Powell, M.; Raven, P.H. 1972. Chromosome numbers in Compositae VIII: Heliantheae. American Journal of Botany. 59(8): 869-878.
Stevens, R. 1994. Interseeding and transplanting to enhance species composition. In: Monsen, S.B.; Kitchen, S.G., comps. Proceedings-Ecology and management of annual rangelands; 1992 May 18-22; Boise, ID. Gen. Tech. Rep. INT-GTR-313. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Research Station: 300-306.
Stevens, R. 2004. Incorporating wildlife habitat needs into restoration and rehabilitation projects. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., eds. Restoring western ranges and wildlands. Gen. Tech. Rep. RMRS-GTR-136-vol. 1. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 155-174.
Stevens, R.; Jorgensen, K.R.; Davis, J.N. 1981. Viability of seed from thirty-two shrub and forb species through fifteen years of warehouse storage. Great Basin Naturalist. 41(3): 274-277.
Stevens, R.; McArthur, E.D.; Davis, J.N. 1992. Reevaluation of vegetative cover changes, erosion, and sedimentation on two watersheds — 1912-1983. In: Clary, W.P.; McArthur, E.D.; Bedunah, D.; Wambolt, C.L., comps. Proceedings- Symposium on ecology and management of riparian shrub communities; 1991 May 29-31; Sun Valley, ID. Gen. Tech. Rep. INT-289. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Research Station: 123-128.
Stevens, R.; Moden, W.L.; McKenzie, D.W. 1981b. Interseeding and transplanting shrubs and forbs into grass communities. Rangelands. 3(2): 55-58.
Stevens, R.; Monsen, S.B. 2004. Forbs for seeding range and wildlife habitats. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 2. Gen. Tech. Rep. RMRS-GTR-136-vol-2. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 425-466.
Stevens, R.; Monsen, S.B. 2004a. Guidelines for restoration and rehabilitation of principal plant communities. In: Monsen, S.B.; Stevens, R.; Shaw, N.L., comps. Restoring western ranges and wildlands, vol. 1. Gen. Tech. Rep. RMRS-GTR-136-vol-1. Fort Collins, CO: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 199-294.
Stevens, R.J.; Jorgensen, K.R.; Young, A.S.; Monsen, S.B. 1996. Forb and shrub seed production guide for Utah. AG 501. Logan, UT: Utah State University Extension. 52 p.
Thomson, J.D. 1980. Skewed flowering distributions and pollinator attraction. Ecology. 61(3): 572-579.
Tilley, D. 2012. Plant guide: showy goldeneye (Heliomeris multiflora). Aberdeen, ID: U.S. Department of Agriculture, Natural Resources Conservation Service, Aberdeen Plant Materials Center. 3 p.
Tilley, D. 2016. Propagation protocol for production of propagules (seeds, cuttings, poles, etc.) Heliomeris multiflora Nutt. seeds. Native Plant Network. U.S. Department of Agriculture, Forest Service, National Center for Reforestation, Nurseries, and Genetic Resources. http://NativePlantNetwork.org [Accessed 2018 January 3].
USDA Forest Service [USDA FS] 1937. Range plant handbook. Washington, DC: U.S. Department of Agriculture, Forest Service. 816 p.
USDA Forest Service, Bend Seed Extractory [USDA FS BSE]. 2017. Nursery Management Information System Version 4.1.11. Local Source Report 34-Source Received. Bend, OR: U.S. Department of Agriculture, Forest Service, Bend Seed Extractory.
USDA Forest Service, Western Wildland Environmental Threat Assessment Center [USFS WWETAC]. 2017. TRM Seed Zone Applications. Prineville, OR: U.S. Department of Agriculture, Forest Service, Western Wildland Environmental Threat Assessment Center. https://research.fs.usda.gov/pnw/products/dataandtools/seed-zone-webmap
USDA Natural Resources Conservation Service [USDA NRCS]. 2017. The PLANTS Database. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team. https://plants.sc.egov.usda.gov/
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2016. Bureau of Land Management technical protocol for the collection, study, and conservation of seeds from native plant species for Seeds of Success. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program. 37 p.
USDI Bureau of Land Management, Seeds of Success [USDI BLM SOS]. 2017. Seeds of Success collection data. Washington, DC: U.S. Department of the Interior, Bureau of Land Management, Plant Conservation and Restoration Program.
USDI Environmental Protection Agency [USDI EPA]. 2017. Ecoregions. Washington, DC: U.S. Department of the Interior, Environmental Protection Agency. https://www.epa.gov/eco-research/ecoregions
USDI Geological Survey [USDI USGS]. 2017. Biodiversity Information Serving Our Nation (BISON). U.S. Geological Survey. https://www.gbif.us/
Utah Crop Improvement Association [UCIA]. 2015. How to be a seed connoisseur. Logan, UT: UCIA, Utah Department of Agriculture and Food, Utah State University and Utah State Seed Laboratory. 16 p.
Wahlberg, N. 2001. The phylogenetics and biochemistry of host-plant specialization in Melitaeine butterflies (Lepidoptera: Nymphalidae). Evolution. 55(3): 522-537.
Wallace, M.C. 1991. Elk habitat use in the White Mountains, Arizona. Tucson, AZ: University of Arizona. Thesis. 185 p.
Wallmo, O.C. 1954. Nesting of Mearns quail in southeastern Arizona. The Condor. 56(3): 125-128.
Ward, A.L., Keith, J.O. 1962. Feeding habits of pocket gophers on mountain grasslands, Black Mesa, Colorado. Ecology. 43(4): 744-749.
Weber, W.A. 1976. Rocky Mountain flora. Boulder, CO: Colorado Associated University Press. 479 p.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C., eds. 1987. A Utah flora. The Great Basin Naturalist Memoir 9. Provo, UT: Brigham Young University. 894 p.
Wentworth, T.R. 1981. Vegetation on limestone and granite in the Mule Mountains, Arizona. Ecology. 62(2): 469-482.
Williams, M.C. 1989. Accumulation of nitrate by annual goldeneye and showy goldeneye. Journal of Range Management. 42(3): 196-198.
Young, S.A.; Schrumpf, B.; Amberson, E. 2003. The Association of Official Seed Certifying Agencies (AOSCA) native plant connection. Moline, IL: AOSCA. 9 p. Available: https://seedcert.oregonstate.edu/sites/seedcert.oregonstate.edu/files/pdfs/aoscanativeplantbrochure.pdf
How to Cite
Gucker, Corey L.; Shaw, Nancy L. 2018. Showy goldeneye (Heliomeris multiflora). In: Gucker, C.L.; Shaw, N.L., eds. Western forbs: Biology, ecology, and use in restoration. Reno, NV: Great Basin Fire Science Exchange. Online: https://westernforbs.org/species/showy-goldeneye-heliomeris-multiflora/